Research article
Prevalence of ESBL, AmpC β-lactamases and MRSA among uropathogens and its antibiogram
B Sasirekha1[*]
1Department of Microbiology, Center for Post Graduate Studies, Jain University, Bangalore, Karnataka- 560 011, IndiaEXCLI J 2013;12:Doc81
Abstract
Urinary tract infections (UTIs) are one of the most common bacterial infections in humans. Current knowledge on antimicrobial susceptibility pattern is essential for appropriate therapy. Therefore the aim of the present study was to determine the distribution and antibiotic susceptibility pattern of bacterial strains, with special reference to ESBL, AmpC β-lactamase and MRSA production. A total of 325 clinical isolates were collected from UTI patients from various tertiary care hospitals over a period of 6 months (June 2011 to December 2011). The antimicrobial susceptibility to various drugs was studied by the disc diffusion method as guided by CLSI guidelines. Confirmation of the extended spectrum β-lactamase (ESBL), AmpC β-lactamase and methicillin resistant Staphylococcus aureus (MRSA) production was done by an E-test respectively. Of the 325 isolates, 225 were characterized as gram negative and 77 isolates as S. aureus isolates. The majority (69.3 %) of the isolates were from females compared to males (30.7 %). Prevalence of ESBL and AmpC β-lactamase and the coexistence of the phenotype (ESBL+ AmpC β-lactamase) and MRSA in the urinary isolates were found to be 48.9 %, 20.4 %, 6.2 % and 27.5 % respectively. Ampicillin/sulbactam, norfloxacin should be no longer considered as first line of drugs for UTI, because of high resistance. Parentral drugs such as aminoglycosides, carbapenems and piperacillin/tazobactum can be the alternative choice for complicated UTI. Also, control measures such as judicious use of antibiotics, formulation of infection committee may control the spread of resistance.
Keywords: UTI, ESBL, MRSA, antibiotic resistance, AmpC ß-lactamases
Introduction
Urinary tract infections (UTIs) are among the most common bacterial infections both in the community and hospital setting (Mohammadi et al., 2010[19]). UTI has become the most common hospital-acquired infection, accounting for as many as 35 % of nosocomial infections (Kolawole et al., 2009[12]). UTI is the second most common infectious presentation in community practice. It accounts for approximately one million hospitalizations annually worldwide (Farrell et al., 2003[6]). It presents a spectrum of clinical entities upon severity ranging from asymptomatic infection to acute pyelonephritis with sepsis (Fish, 2009[7]).
In the last few decades there have been significant changes in the antimicrobial resistance patterns of uropathogens and reports on spread of bacterial resistance among uropathogens (Mohammed et al., 2007[20]). The increasing prevalence of infections caused by antibiotic-resistant bacteria makes empirical treatment of these infections difficult (Steinke et al., 2001[30]). The emergence of antibiotic resistance in the management of UTI infection is a serious public health issue, particularly in the developing countries where apart from high level of poverty, ignorance and poor hygienic practices, there is also high prevalence of fake and spurious drugs of questionable quality in circulation.
In the majority of cases, antibiotics are given empirically before the final bacteriology results are available. Antibiotic resistance varies according to geographic locations and is directly proportional to the use and misuse of antibiotics. The resistance pattern of community acquired UTI pathogens has not been studied extensively (Goldstein, 2000[8]). The etiology of UTI and the antibiotic resistance of uropathogens have been changing over the past years, both in community and nosocomial infection (Manges et al., 2006[15]). However, there is not much information available on etiology and resistance pattern of community acquired UTIs in India. Consequently, area-specific monitoring studies to document the microorganisms causing UTIs and their antimicrobial susceptibility is mandatory for helping the selection of an effective empirical treatment (Smith and Coast, 2002[29]). Therefore, the present study was aimed at gaining knowledge about the type of pathogens responsible for and their susceptibility patterns which will help the clinicians to choose the right empirical treatment.
Materials and Methods
Bacterial isolates
A total of 325 consecutive, non-repetitive, clinical isolates were collected from UTI patients from tertiary care hospitals during 2011 (June to December) were included in the study. Urine samples were cultured using a 0.001 ml calibrated loop onto blood agar and MacConkey agar plates, incubated at 37 °C for 18-24 hours and the number of colonies was counted. Significant bacteriuria was defined as greater than 105 colony forming units/ml of a single pathogen. Isolates were identified based by standard microbiological procedures (Koneman et al., 2006[13][14]).
Antibiotic susceptibility testing
The isolates were tested for their antimicrobial susceptibility and the results were interpreted according to the guidelines of Clinical and Laboratory Standards Institute (CLSI, 2010[4]). Gram negative isolates were tested against ampicillin (10 μg), ampicillin/Sulbactam (20 µg/10 μg), piperacillin (100 µg); piperacillin-tazobactam (100 µg/10 μg), cephotaxime (30 μg), ceftriaxone (30 μg), ceftazidime (30 μg), cefoxitin (30 µg), amikacin (30 μg), norfloxacin (5 μg), nalidixic acid (30 μg), nitrofurantoin (300 μg), gentamicin (15 μg), ciprofloxacin (5 μg), trimethoprim-sulfamethoxazole (1.25/23.75 μg), and imipenem (10 μg). S. aureus isolates were tested with oxacillin (1 µg); cefoxitin (30 µg); erythromycin (15 μg); mupirocin (5 μg); fusidic acid (30 μg); pristinomycin (15 μg); linezolid (30 μg); vancomycin (30 μg); teicoplanin (30 μg); rifampin (5 μg); chloramphenicol (30 μg); cotrimoxazole (30 μg); ciprofloxacin (5 μg); gentamicin (30 μg); amikacin (30 μg), and tetracycline (30 μg) (Hi Media, India). Multi-drug resistance was defined as resistance to three or more different antimicrobial agents. E. coli ATCC 25922 and S. aureus ATCC 25923 was used as a quality control strain.
Detection of ESBL
According to the CLSI guidelines (CLSI 2010[4]), isolates showing inhibition zone size of ≤ 22 mm with ceftazidime (30 μg), ≤ 25 mm with ceftriaxone (30 μg), and ≤ 27 mm with cefotaxime (30 μg) were interpreted as positive for ESBL production and shortlisted for confirmation of ESBL production. E. coli ATCC 25922 was used as the control strain.
Confirmation of ESBL production by E-test
Confirmation of ESBL was also done by E-test ESBL strips (Hi Media, India), and the test was performed in accordance with the manufacturer's guidelines. Double ended strips containing gradient of cefotaxime or ceftazidime at one end and cefotaxime or ceftazidime plus clavulanic acid at the other end were tested. The presence of ESBL was confirmed if the ratio of the MIC of cefotaxime or ceftazidime to the MIC of cefotaxime or ceftazidime plus clavulanic acid was ≥ 8 (CLSI, 2011[3]).
Phenotypic AmpC detection
Since there are no published CLSI criteria for phenotypic screening or confirmatory test for AmpC β-lactamases, a modified three dimensional test was used for the detection of AmpC enzymes in cefoxitin resistant isolates (Singhal et al., 2005[28]).
Oxacillin E-test
MICs of oxacillin were determined by the E-test (Hi Media, India) according to the instructions of the manufacturer. The plates were inoculated by swabbing the surfaces with a 0.5 McFarland's standard bacterial suspension on the MHA medium which was supplemented with 2 % NaCl. The E-test strips were placed on the medium, and the plates were then incubated at 35°C for 24 hours. The results were analyzed on the basis of the CLSI guidelines (CLSI, 2010[4]).
Statistical analysis
Statistical package for social sciences (SPSS) version 18 was used to analyze our data. Clinical outcomes were compared statistically using Student's t- test. P <0.05 was taken as “statistically significant”.
Results
During the study period, a total of 325 isolates were collected from various healthcare centers, out of which 225 isolates were characterized as gram negative and 77 isolates as S. aureus isolates. These isolates were identified up to species level by conventional methods. The uropathogens isolates include Escherichia coli (91), Klebsiella pneumoniae (82), Pseudomonas aeruginosa (26), Proteus mirabilis (21), Enterobacter aerogenes (4), Citrobacter freundii (1) and Staphylococcus aureus (77). The number of uropathogens was larger in females (69.3 %) than in males (30.7 %). The difference was statistically significant (p < 0.0001). Antibiogram patterns of 225 gram negative isolates revealed that 78.7 % (177/225) isolates were found to be resistant to more than 3 antibiotics.
By the screening test ESBL production was noticed in 145 (64.4 %) isolates with maximal incidence in E. coli (71.42 %) followed by K. pneumoniae (65.9 %), P. aeruginosa (50 %), Proteus mirabilis (47.61 %) and Enterobacter aerogenes (75 %). ESBL production was not noticed in Citrobacter freundii. Of the 145 ESBL screen positive isolates, 110 isolates were found to be ESBL producers by MIC using the E-test strip. MIC values of ESBL positive isolates towards cefotaxime alone were ≥ 64 µg/ml and for cefotaxime/clavulanate combination was ≥ 1 µg/ml.
Overall, the prevalence of ESBL and AmpC β-lactamase and the coexistence of the phenotype (ESBL+ AmpC β-lactamase) and MRSA in the urinary isolates were found to be 48.9 %, 20.4 %, 6.2 % (Table 1(Tab. 1)), and 27.5 % respectively. Three isolates of Klebsiella pneumoniae and 8 isolates of Escherichia coli showed coexistence of their phenotypes which showed a significant correlation (p value < 0.05) along with the production of ESBL. All the cefoxitin resistant isolates were AmpC β-lactamase among gram negative isolates, indicating that cefoxitin was a good drug for the detection of AmpC β-lactamase.
The maximum ESBL activity was seen in Escherichia coli (52.74 %), followed by P. aeruginosa (53.9 %) and Klebsiella pneumoniae (45.1 %). The maximum AmpC β-lactamase production was found in P. aeruginosa (34.7 %) followed by E. coli (19.8 %) and K. pneumoniae (18.2 %) and P. mirabilis (14.2 %).
Out of 58 oxacillin resistant isolates 42 isolates were resistant to cefoxitin, which were further confirmed as MRSA strains by the oxacillin E-test (p < 0.0001). Out of 42 MRSA, 90.4 % MRSA isolates were from coagulase positive S. aureus whereas 9.52 % MRSA isolates were from coagulase negative S. aureus.
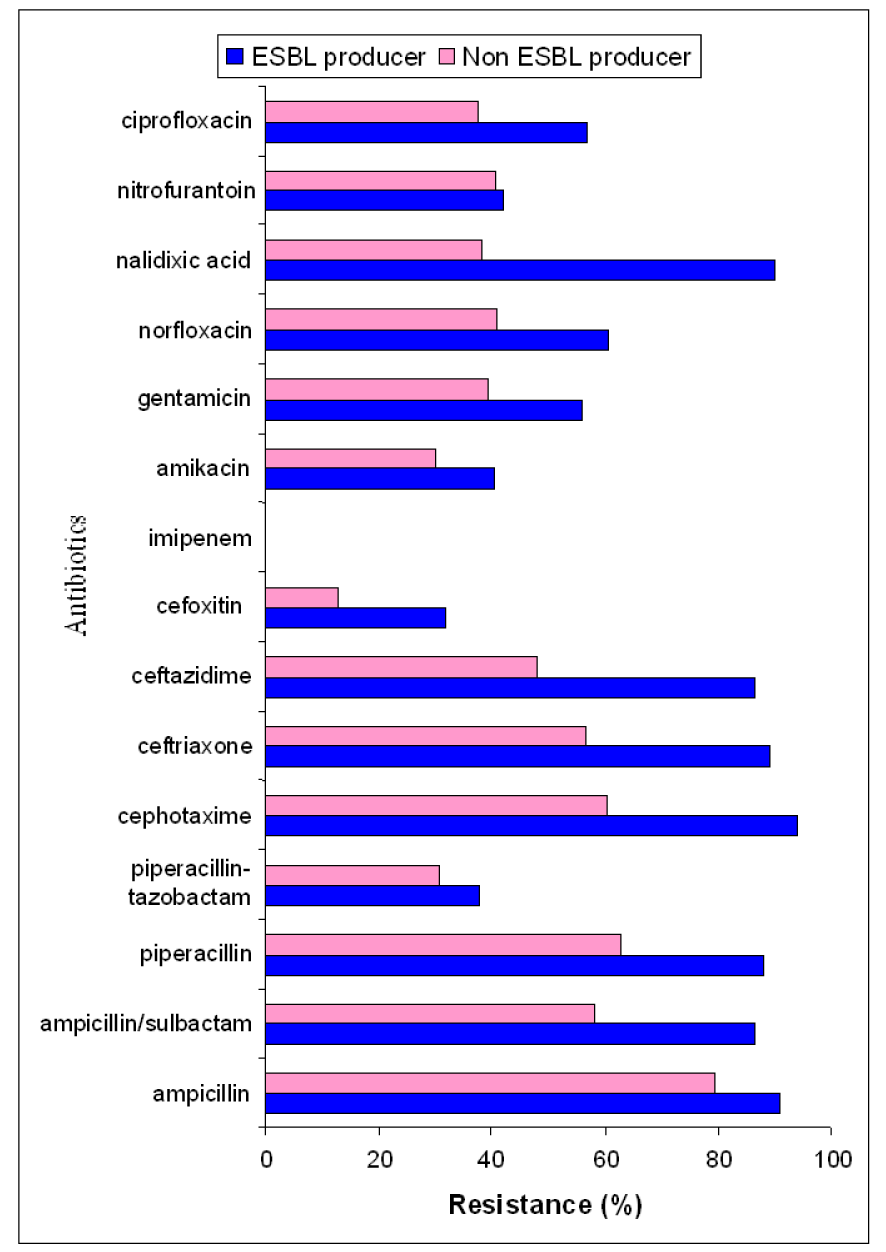
Antibiogram patterns for the gram negative isolates analyzed showed ESBL-producing isolates to possess a higher degree of resistance towards antibiotics that are routinely prescribed against urinary tract infections as compared to non-ESBL-producing isolates (Figure 1(Fig. 1)). Cephalosporin resistance was higher in ESBL producing (87-94 %) when compared to ESBL non producers. Combination of β-lactam/β-lactamase inhibitors showed greater activity in both ESBL producers and non-producers. However, all isolates were sensitive to imipenam. Among aminoglycosides, resistance to amikacin was found to be comparatively lower in both ESBL and non-ESBL producers.
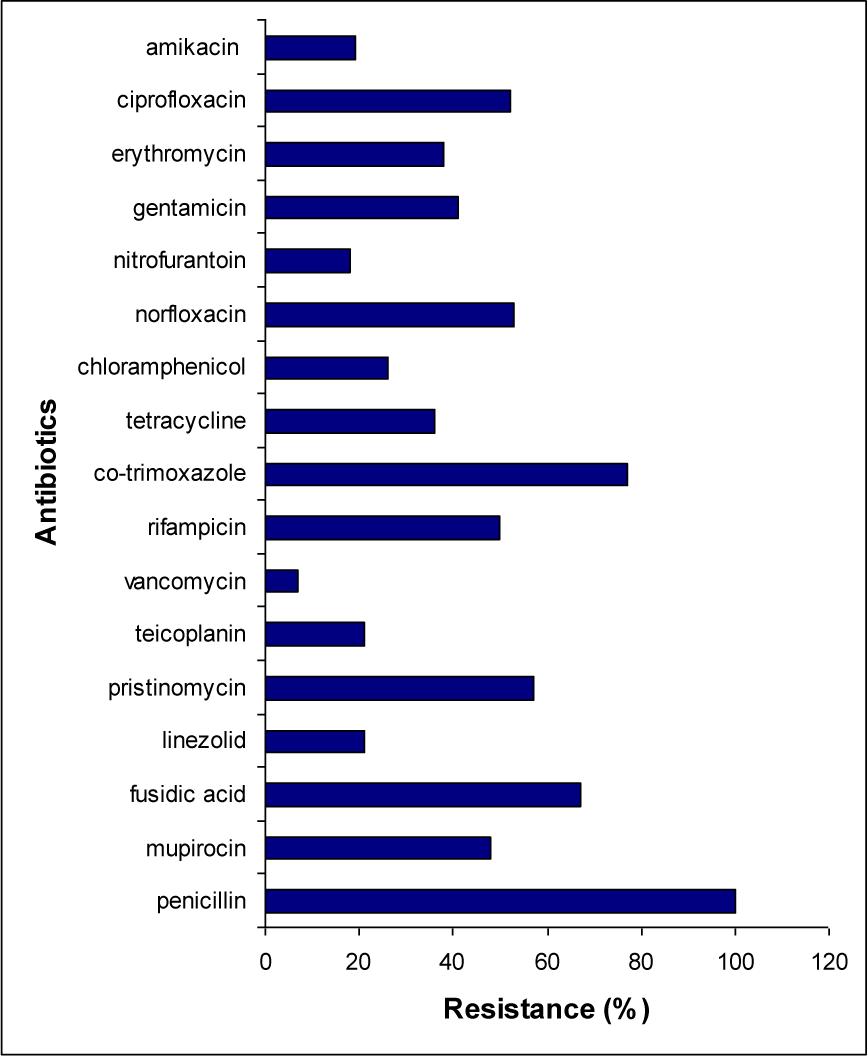
S. aureus were resistant to two to fourteen antibiotics out of fifteen antibiotics tested corresponding to a multiple antibiotic resistance index (MAR) range from 0.27 to 0.93. Antibiotic sensitivity pattern in MRSA isolates revealed 92.9 % sensitivity for vancomycin, 81 % to amikacin and 79 % for linezolid and teicoplanin. The maximum resistance was seen against penicillin, fusidic acid, cotrimoxazole, pristinomycin, rifampicin and mupirocin (47.7 %) (Figure 2(Fig. 2)).
Discussion
Knowledge on local antimicrobial resistance trends among urinary isolates is important not only in guiding clinicians to prescribe appropriate antibiotics but also for evidence based recommendations in empirical antibiotic treatment of UTI. The current study describes the antimicrobial resistance rates including detection of ESBL, AmpC β-lactamase and MRSA among urinary isolates.
The study revealed E. coli and Klebsiella species to be the dominant organisms among other uropathogens. This result coincides with the previous report on the predominance of these pathogens (Karou et al., 2009[10]; Moyo et al., 2010[21]). Some studies, however, have shown a decline of E. coli, being replaced by other members of the Enterobacteriaceae. The UTIs are more frequent in women than in men, principally owing to anatomic and physical factors (Muvunyi et al., 2011[22]; Moyo et al., 2010[21]).
Antimicrobial susceptibility patterns of uropathogens vary widely by region. In the current study the antimicrobial resistance rate of gram negative isolates was high to the first line antimicrobial agents such as ampicillin, cotrimoxazole, and amoxicillin/clavulanic acid. This may be due to wide use of these drugs empirically because they are relatively cheap and also by being oral antibiotics they are easy to administer.
In India, the prevalence of ESBLs has been reported from 1990s. The occurrence of ESBL producers among the gram negative bacilli in the current study was 48.9 %. This number is less than that previously reported (Dalela et al., 2012[5]; Narayanaswamy and Mallika, 2011[23]). The present study reports 27.4 % MRSA among S. aureus isolates. Few earlier studies have reported comparable prevalence of 24 % in Chandigarh (Mehta et al., 2007[18]), 31.1 % in a multicenter study in Tamilnadu (Rajaduraipandi et al., 2006[26]). In contrast, variable prevalence of 80.89 % in Indore (Verma et al., 2000[31]) and 54.9 % in eastern Uttar Pradesh (Anupurba et al., 2003[2]) has also been reported. This variability could be due to difference in the study design, population and geographical distribution and the variation is probably due to differential clonal expansion and drug pressure in community. In the current study, 52.8 % and 45.1 % of E. coli and K. pneumoniae were identified as ESBL-producers.
Recently, the co-existence of both AmpC β-lactamase and ESBL in some gram negative bacilli has also been reported. This could be because plasmid mediated AmpC β-lactamase has been disseminated among the Enterobacteriaceae. These strains in combination with ESBL may give false negative tests in the detection of ESBL.
ESBL-producing strains showed a significantly high rate of resistance to non β- lactam antibiotics. These findings are similar to those reported by others (Ndugulile et al., 2005[24]; Mehrgan and Arab-Halvaii, 2010[17]). This observation may be explained by the fact that ESBL are plasmid mediated enzymes which are transferable between one bacterium to another and such transferable plasmids also code for resistance determinants to antimicrobial agents other than β-lactams (Paterson, 2006[25]).
Among the oral antibiotics, norfloxacin a commonly prescribed for the treatment of UTI in India and also in other countries (Karlowsky et al., 2003[9]) showed a high resistance rate against uropathogens in our study which indicated an increased quinolone resistance in our area, the findings are consistent with those of the previous studies (Akram et al., 2007[1]; Manjunath et al., 2011[16]). Another oral antibiotic nitrofurantoin was found to be more effective in the treatment of UTI, these findings are in agreement with similar surveillance studies (Khameneh and Afshar, 2009[11]). A low degree of resistance to amikacin was observed for both ESBL and non-ESBL producers and hence may be helpful in combating severe infections. In our study, imipenem was the only drug found unaffected by the action of these enzymes. These findings are in concordance with the studies conducted by other authors, who also reported a rate of 100 % sensitivity to imipenam (Sasirekha and Srividya, 2012[27]).
The current study, which examines the spread of drug resistance among uropathogens, is perhaps first of its kind as it is not restricted to a specific health care center. On the basis of our findings, we suggest urine culture and antimicrobial susceptibility testing for UTI patients. Continuous analysis of the antibiotic resistance pattern acts as a guide in initiating the empirical treatment of UTI. Development of regional surveillance programs is necessary to provide information which would then enable the development of Indian UTI guidelines.
References
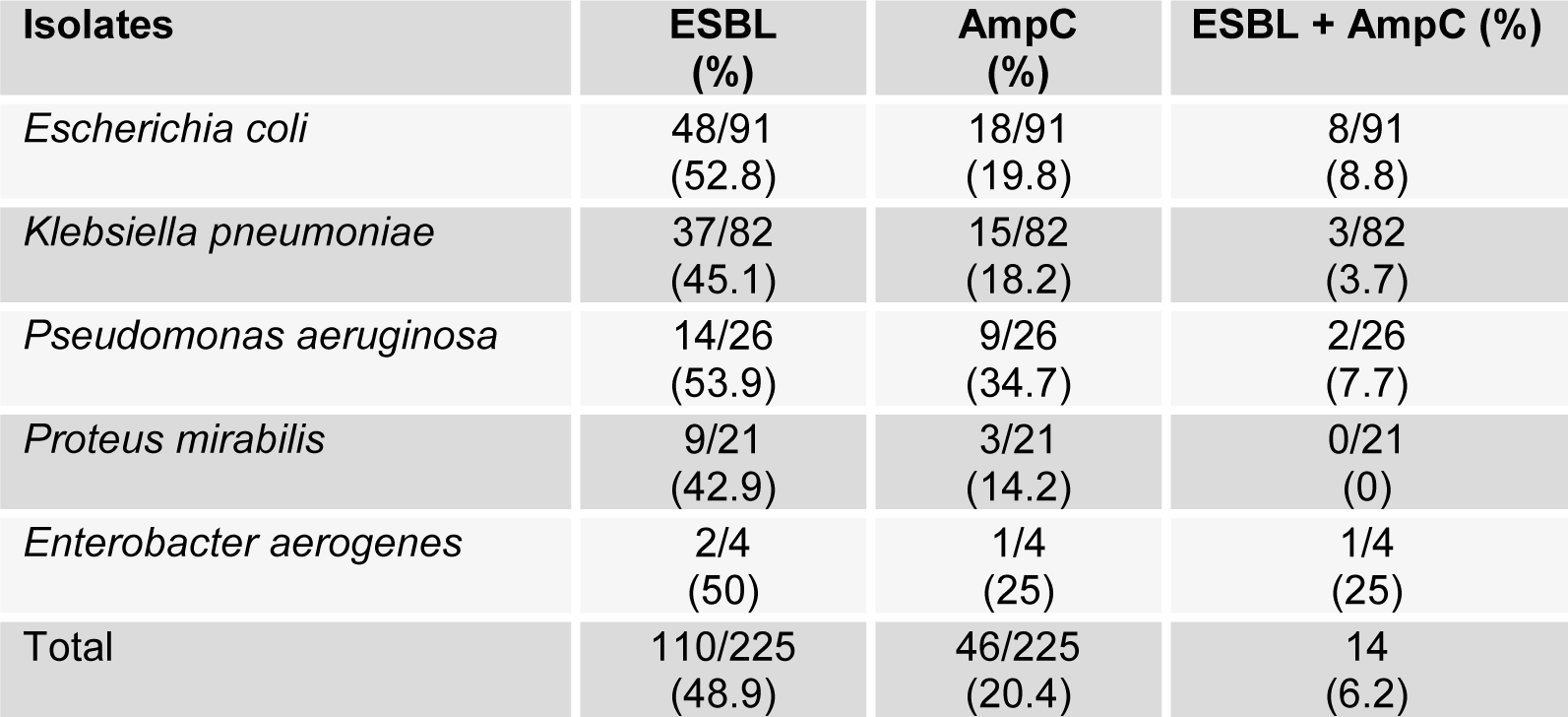
Table 1: Prevalence of ESBL, AmpC β-lactamases and the coexistence of resistance (ESBL + AmpC β-lactamases) among gram negative bacilli in various clinical isolates
[*] Corresponding Author:
B Sasirekha, Department of Microbiology, Center for Post Graduate Studies, Jain University, Bangalore, Karnataka- 560 011, India, Phone: +91 080 43226500, eMail: nagsrani@yahoo.co.in