Review article
The role of glutamatergic pathway between septum and hippocampus in the memory formation
Fatemeh Khakpai1[*], Mohammad Reza Zarrindast2,3,4,5,6, Mohammad Nasehi7, Ali Haeri-Rohani1, Akram Eidi1
1Department of Biology, Science and Research Branch, Islamic Azad University, Tehran, Iran2Department of Neuroscience, School of Advance Medical Technologies, Tehran University of Medical Sciences, Tehran, Iran
3Department of Pharmacology School of Medicine, Tehran University of Medical Sciences, Tehran, Iran
4Institute for Cognitive Science Studies (ICSS), Tehran, Iran
5Iranian National Center for Addiction Studies, Tehran University of Medical Sciences, Tehran, Iran
6School of Cognitive Sciences, Institute for Research in Fundamental Sciences (IPM), Tehran, Iran
7Department of Biology, Faculty of Basic Sciences, Islamic Azad University, Garmsar Branch, Semnan, Iran
EXCLI J 2013;12:Doc41
Abstract
The N-methyl-D-aspartate (NMDA) receptor is a subtype of glutamate receptor that is presented in highest density in the hippocampus and septum. NMDA receptors of the septum and the hippocampus are involved in cognitive performance, especially in learning and memory processes. The septum nucleus and hippocampal formation are two regions of the limbic system. The septum and the hippocampus are anatomically and functionally connected to each other. These areas made the septo-hippocampal and hippocampo-septal pathways, which are implicated in the cognitive processes. The activity of septal and hippocampal neurons is modulated by several neurotransmitters such as glutamate. Thus, changes in the glutamatergic transmission in the septum and hippocampus may influence learning and memory processes in these pathways.
Keywords: septum, hippocampus, septo-hippocampal, hippocampo-septal, learning, memory
Introduction
Glutamate is the most frequent amino acid in the brain (Freitas-Ferrari et al., 2010[15]). The N-methyl-D-aspartate (NMDA) receptor is a subtype of glutamate receptor which is an important mediator of synaptic plasticity and plays a central role in the neurobiological mechanisms of emotionality, as well as learning and memory (Bermudo-Soriano et al., 2012[5]; Harvey and Shahid, 2012[19]; Hepp et al., 2012[21]; Kelley et al., 2010[25]; Ghafari et al., 2011[16]; Barkus et al., 2010[4]; Mahmoodi et al., 2010[35]; Ota et al., 2010[44]). The NMDA receptor is found in high density in the septum and hippocampus. NMDA receptors of the septum nucleus and the hippocampal formation are implicated in learning and memory. The medial septum/diagonal band of Broca (MS/DB) complex is projected to the hippocampal formation (Yousefi et al., 2012[56]; Lu et al., 2011[34]; Wevers, 2011[55]; Abreu-Villaca et al., 2010[1]; Deiana et al., 2010[10]; Drever et al., 2010[13]). One important and renowned efferent projection of the MS/DB complex is the septo-hippocampal pathway. This pathway is critical for learning and memory processes (Niewiadomska et al., 2009[42]). More recently, glutamatergic neurons have been suggested as a component of the neurochemical basis of the septo-hippocampal pathway (Cervera-Ferri et al., 2012[6]; Huh et al., 2010[22]).
Anatomical studies indicated that the hippocampus and the septum nucleus have reciprocal pathways (Okada and Okaichi, 2010[43]). The hippocampus receives both cholinergic and GABAergic fibers located in the medial septum/diagonal band of broca (MS/DB) complex via the fimbria-fornix (Kanju et al., 2012[24]; Zarrindast et al., 2012[57]; Robinson et al., 2011[47]; Van der Zee and Keijser 2011[53]; Hangya et al., 2009[18]; Roland and Savage, 2009[48]). On the other hand, the hippocampus is terminated on the GABAergic neurons in the medial septum nucleus, and on the glutamatergic neurons in the lateral septum nucleus (Trent and Menard, 2010[52]).
The current review provides summary experiments which have assessed connection of the septum and the hippocampus via septo-hippocampal and hippocampo-septal pathways and the role of glutamate neurotransmission that modulate learning and memory in these pathways. First, we will discuss studies in which glutamate neurotransmission has a role in the regulation of the septum functions. Next, an attempt will be made to converse the role of glutamate neurotransmitter in modulation of hippocampal function. Further, an overview will be given of the studies in which the pivotal role of glutamatergic septo-hippocampal pathway in learning and memory were examined. Finally, a summary will be made of studies investigating the glutamatergic hippocampo-septal pathway in learning and memory. We will show that changes in the glutamate neurotransmission in the septo-hippocampal and hippocampo-septal pathways may affect learning and memory processes.
Septum
The septum nucleus is a subcortical structure and belongs to the limbic formation (Khakpai et al., 2012[27]). The septal region is composed of two major areas: lateral (lateral septal nuclei) and medial (medial septum/diagonal band of Broca nuclei (MS/DB)), each having different neuronal populations (Lecourtier et al., 2010[32]). These subregions have been revealed to differ in their afferent and efferent innervations, their intrinsic connectivity and their functional roles (Khakpai et al., 2012[27]). The medial subdivision of the septum sends major cholinergic and GABAergic inputs to the hippocampus, while the lateral subdivision receives a strong glutamatergic input from the hippocampus and a GABAergic input from the MS/DB complex. The septal nuclei are considered as relay sites of sensory information which connect midbrain and brainstem structures, for example the ventral tegmental area and hypothalamus link to forebrain limbic structures including the hippocampus and prefrontal cortex. The functional importance of these nuclei is obvious from the complicated topographical organization of cholinergic, monoaminergic, peptidergic, and amino acid afferent and efferent systems to both medial and lateral septum. The septum is recognized as important site in processing of sensory information, memory, learning, consolidation and retrieval of passive avoidance response (PAR), reference memory and working memory, long-term potentiation (LTP), theta rhythm, fear, anxiety, stress, emotions, aggression, arousal, motivation, and vegetative function (De Paula et al., 2012[9]; Ashabi et al., 2011[2]; Gutierrez-Guzman et al., 2011[17]; Klinkenberg and Blokland, 2010[29]; Klinkenberg et al., 2010[30]; Lamprea et al., 2010[31]; Roland and Savage, 2009[49]).
Septum and Glutamate
Glutamatergic fibers have been detected in the septal area (Cervera-Ferri et al., 2012[6]). The NMDA receptor plays a key role in synaptic plasticity, neural physiology, behavioral learning and memory process (Khakpai et al., 2012[26]). NMDA receptors (NMDARs) are consisted of NR1 and NR2 subunits. The NR2 subunit mRNA is expressed in the forebrain area. Maximum levels of expression are detected in the cerebral cortex, the hippocampal formation, the septum, the caudate-putamen, the olfactory bulb, and the thalamus (Khakpai et al., 2012[27]). Memory storage may depend on the function of NR2 subunit (Day et al., 2011[8]). Past studies indicated that intra-septal infusions of AP5 (a competitive NMDA receptor antagonist) impaired memory when administered after the acquisition of a passive avoidance task. However, a report indicated that MK-801, a non-competitive NMDA receptor antagonist, can improve memory. Other investigators also revealed that NMDA receptor antagonists, D-AP7 or MK-801 facilitate retention in a step-down inhibitory avoidance task, but impair retention in place navigation and step-through dark avoidance tasks. Other evidence showed that post-training or pre-training intra-septal infusions of NMDA agonists or antagonists can improve or impair, respectively, retention of water maze or avoidance tasks. Thus, it has been argued that the effects of NMDA receptor antagonists on learning and memory are dependent on the type of the task (Khakpai et al., 2012[26]).
Hippocampus
Hippocampus is a part of the limbic system (Farahmandfar et al., 2011[14]), which is a model structure for the physiological analysis of neural systems. Its structure is relatively simple, because the principal cells, the pyramidal and granule neurons are each arranged in separate, compact layers, with dendrites emanating in a parallel manner. The hippocampus proper is divided into division CA1, CA2, CA3 and CA4 and is characterized by a narrow band of pyramidal neurons. The hippocampal formation (HCF; hippocampus, subiculum and entorhinal cortex) is a main neural substrate for learning and memory (Solati, 2011[50]; Myers et al., 2011[38]; Motevasseli et al., 2010[37]; Collins, 2009[7]). Broadly divergent projections (cholinergic, GABAergic, glutamatergic and peptidergic) from MS/DB complex innervate the entire hippocampal formation. These inputs regulate hippocampal formation physiology and memory function. The CA1 region of the dorsal hippocampus mediates neural plasticity processes involved in the acquisition, storage and retrieval of memory in the hippocampus. The CA3 is critical in organizing information in sequential order. The hippocampus processes information not only from several cortical areas, but also from a number of subcortical structures for example the medial septum, hypothalamus and the brain stem. These structures regulate input to hippocampal formation and provide information about the “behavioral state” of the animal (Khakpai et al., 2012[26]).
Two main pathways projecting to the hippocampus originate from the medial septum, and the median raphe. The septo-hippocampal pathway contains a cholinergic component, a GABAergic component, and a glutamatergic component (Cervera-Ferri et al., 2012[6]), whereas the raphe-hippocampal projection consists of both serotonergic and non-serotonergic fibers. The hippocampal neurons are innervated by cholinergic, GABAergic, glutamatergic, noradrenergic and serotonergic axon terminals, and the release of these neurotransmitters plays a modulatory role. The hippocampus is known as a main area for learning and memory, associative learning and memory, spatial learning, spatial memory, declarative memories, working memory, episodic memory, formation of memory, synaptic plasticity, long-term potentiation (LTP), integration of information arriving from different sensory and associational cortical areas, encoding, short-term and long-term memory, acquisition, consolidation and retrieval cognition-related processes such as arousal, attention, anxiety, emotional processing, and time-limited role in the permanent storage of memory (Dong et al., 2012[11]; Lee and Hynds, 2012[33]; Drago et al., 2011[12]; Farahmandfar et al., 2011[14]; Khan and Muly, 2011[28]; Zarrindast et al., 2011[58]; Nasehi et al., 2009[40], 2010[39]; Okada and Okaichi, 2010[43]; Watson and Stanton, 2009[54]). It is well known that the dorsal hippocampus plays a critical role in the learning and memory of spatial tasks while the ventral hippocampus is principally involved in the modulation of fear and anxiety (Lu et al., 2011[34]; Morgado-Bernal, 2011[36]; Nazari-Serenjeh et al., 2011[41]).
Hippocampus and Glutamate
The major glutamatergic input to the hippocampus is provided by pyramidal neurons in layer II and III of the entorhinal cortex. NMDA glutamate receptors are widely dispensed in the brain. Although the maximum concentrations of NMDA receptors are present in the hippocampal CA1 region (Jafari-Sabet, 2011[23]; Watson and Stanton, 2009[54]), these receptors are also present in many brain areas, including septum, thalamus, cortex, amygdala, caudate nucleus, nucleus accumbens, basal ganglia, cerebellum and spinal cord. NMDA receptors in the hippocampus play an important role in the regulation of synaptic plasticity for example LTP, short- and long-term memory, learning, spatial and non-spatial learning, working memory, formation of aversive memory, object recognition memory, and cognitive processes (Jafari-Sabet, 2011[23]; Stephens et al., 2011[51]; Rezayof et al., 2010[46]).
Decreasing of NMDA receptors in the hippocampus are associated with deficits of LTP and spatial learning. So, the NMDA receptor antagonists abolish hippocampal LTP and severely disrupt the hippocampal-dependent learning. Some forms of hippocampal-dependent learning depend on activation of the NMDA receptor. Experimental evidence implicated that blockade of NMDA receptors with AP5 or MK-801 decreased acquisition of hippocampal-dependent tasks. Past studies indicated that intra-hippocampal infusions of AP5 impaired memory when administered after the acquisition, and after training of a passive avoidance task. Taken together, it is reasonable to argue that glutamatergic neurotransmission via NMDA receptors as an essential component of memory processing in the hippocampus (Khakpai et al., 2012[26]).
As for NMDA receptors, AMPA receptors are widely but discretely distributed in the rat brain with high densities found in the hippocampal formation (i.e. stratum radiatum of the CA1 subfield and molecular layer of the dentate gyrus). Moreover, there are many evidences that mGlu1 receptors are critical for learning tasks associated with hippocampal function (Khakpai et al., 2012[27]).
Septo-Hippocampal Pathway
The septo-hippocampal pathway is the important and renowned efferent projection of the MS/DB complex that plays a key role in the learning and memory processes (Khakpai et al., 2012[27]; Roland and Savage, 2009[48]) (Figure 1(Fig. 1); Reference in Figure 1: Khakpai et al., 2012[27]). Activation of the medial septum nucleus enhances the synchronized firing of pyramidal fibers in the hippocampus. Septal afferent neurons are found in all regions of the hippocampus (dentate gyrus, CA3 and CA1) but, they are especially dense in the dentate gyrus. The afferent neurons from the medial septum nucleus to the dentate gyrus of the hippocampus is not chemically homogenous. In the hippocampal formation, septal afferent neurons are topographically arranged, with the most fibers terminating in the hilus of the dentate gyrus, strata oriens, radiatum of the CA3 area and subiculum. A few septal afferents terminate within the molecular layer of the dentate gyrus, presubiculum, parasubiculum and entorhinal cortex (Khakpai et al., 2012[26][27]).
Septo-Hippocampal Glutamatergic Pathway
A glutamatergic septo-hippocampal connection has also been recognized to regulate the activity of septal neurons projecting to the hippocampus (Cervera-Ferri et al., 2012[6]; Khakpai et al., 2012[26]; Ransome and Hannan, 2012[45]; Roland and Savage, 2009[48]). The different subtypes of glutamate receptors present in high density in the hippocampus and septum (Khakpai et al., 2012[27]). Glutamate receptors of the septum nucleus and the hippocampal formation play a critical role in learning and memory (Khakpai et al., 2012[26]). Some studies have proven that medial septal NMDA receptors contribute to hippocampal-dependent learning in the rat. Glutamate has been indicated to activate ionotropic (AMPA and NMDA receptors) and metabotropic (type I) receptors, possibly locating on both cholinergic and GABAergic neurons. Glutamate is in the site to regulate the activity of septal neurons projecting to the hippocampus, because the MS/DB cholinergic and GABAergic fibers are synaptically innervated by vesicular glutamate transporter 2-immunoreactive glutamatergic terminals (Khakpai et al., 2012[27]; Banerjee et al., 2010[3]).
Memory Functions of Septo-Hippocampal Pathway
The medial septum nucleus and diagonal band of Broca (MS/DB) complex are mutually connected with the hippocampus which made a main input for spatial learning and hippocampal theta rhythm. Although cholinergic septo-hippocampal projections have been well described, but the functional significance of the glutamatergic projection has not been proven. Furthermore, about 4-23 % of the septo-hippocampal projection is glutamatergic. The glutamatergic system in the (MS/DB) complex, probably via the NMDA receptors is involved in hippocampal-dependent learning and memory. Some evidence is shown that intra-septal administration of glutamate receptor antagonists decreased hippocampal learning and memory (Huh et al., 2010[22]; Henderson et al., 2010[20]).
Hippocampo-Septal Pathway
The hippocampus has anatomical connections with various subcortical areas, for instance the medial septum nucleus and the lateral septum nucleus (Figure 2(Fig. 2); Reference in Figure 2: Okada and Okaichi, 2010[43]). The hippocampus uses this connection to self-regulate cholinergic input. Specifically, high novel in the input pattern should cause activity in the hippocampo-septal pathway, which should in turn activate cholinergic septo-hippocampal pathway and drive storage in the hippocampus. The medial septum nucleus projects into the hippocampus almost entirely ipsilaterally through the fimbria-fornix. In return, the hippocampus projects into the medial septum nucleus directly via the fimbria-fornix and indirectly via the lateral septum nucleus. The main hippocampus subregions, CA1, CA3 and the dentate gyrus have diverse anatomical relationships with the medial septum nucleus. While each of these subregions receives ipsilaterally projection from the medial septum nucleus, only CA1 and CA3 project to the cholinergic and non-cholinergic fibers in the medial septum nucleus. Reciprocal connections between the medial septum nucleus and the CA1 and CA3 might be important components of the septo-hippocampal pathway. In particular, the CA3 and CA1 pyramidal neurons innervate topographically distinct areas of the lateral septum nucleus. The CA3 pyramidal fibers have confined extra-hippocampal projections, predominantly innervating caudodorsal lateral septum nucleus. In contrast, the CA1 gives rise to main projections to the subiculum, the retrohippocampal region, the medial/limbic striatum including the nucleus accumbens, and the rostroventral septum (Khakpai et al., 2012[26][27]; Okada and Okaichi, 2010[43]).
The hippocampus sends a GABAergic projection to the medial septum nucleus and a glutamatergic projection to the lateral septum nucleus (Figure 3(Fig. 3); Reference in Figure 3: Trent and Menard, 2010[52]). The GABAergic pathway projects from non-pyramidal neurons in the stratum oriens of the CA1-CA3 area and innervates cholinergic and non-cholinergic fibers. The glutamatergic projection originates from pyramidal neurons and terminates on the GABAergic fibers of the lateral septum nucleus. Stimulating of cholinergic receptors in the hippocampus could excite both the GABAergic and the glutamatergic projections. Exciting of the GABAergic projection would possibly result in a reduction of septal activity. Likewise, stimulating the glutamatergic pathway would stimulate the GABAergic neurons of the lateral septum nucleus and also result in a decrease of septal activity. So, the hippocampus could prevent the medial septum nucleus via either a direct or an indirect pathway. The direct pathway is the GABAergic projection from the hippocampus to the medial septum nucleus and the indirect pathway is the excitatory glutamatergic projection from the hippocampus to the lateral septum nucleus, which in turn stimulates an inhibitory GABAergic projection to the medial septum nucleus. Therefore, exciting hippocampal cholinergic receptors could excite the direct hippocampal GABAergic projection, or the indirect glutamatergic projection (Khakpai et al., 2012[27]; Trent and Menard, 2010[52]).
Memory Functions of Hippocampo-Septal Pathway
The hippocampus cooperates with the medial septum nucleus in the acquisition of information. Several reports show that electrolytic and some neurotoxic lesions of the medial septum nucleus result in extreme impairments in learning and memory for tasks also impaired by hippocampal damage. Exciting of the glutamatergic hippocampo-septal pathway would stimulate the GABAergic neurons of the lateral septum nucleus and also result in a reduction of septal activity. Glutamatergic system in the hippocampo-septal pathway has a role in modulation of memory. However, many experiments are required to clarify the exact mechanism(s) involved (Khakpai et al., 2012[27]; Okada and Okaichi, 2010[43]; Niewiadomska et al., 2009[42]).
Conclusion
The current review article described studies in the septo-hippocampal and hippocamo-septal pathways which have been investigated the role of these pathways in the learning and memory processes. An attempt has been made to define glutamate neurotransmitter in the septum nucleus and the hippocampus region that modulated learning and memory in the septo-hippocampal and hippocampo-septal pathways. To this purpose, various experimental studies were used.
Anatomical connections between the medial septum nucleus and hippocampus by the way of the fimbria-fornix suggest that functional cooperation between these compartments may be important for the learning and memory-related behaviors, such as acquisition of information, spatial memory and extent of voluntariness of behavior (Okada and Okaichi, 2010[43]).
The glutamatergic system in the septum is involved in hippocampal-dependent learning and memory. Furthermore, stimulating of the glutamatergic receptors in the hippocampus would excite the GABAergic fibers of the lateral septum which result in a decrease of septal activity. Some studies revealed that blockade of NMDA receptors with glutamate receptor antagonist's decreased acquisition of hippocampal-dependent tasks. Also, there are many studies that metabotropic receptors of glutamate (mGlu1 receptors) are important for learning processes associated with hippocampal function.
Numerous evidences indicated that glutamatergic inputs to the septum exerted a tonic excitatory influence on septal GABAergic activity, so they might inhibit indirectly septo-hippocampal cholinergic neurons. Notably, investigators have implicated that medial septal glutamatergic NMDA receptors contribute to hippocampal-dependent learning in the rat (Khakpai et al., 2012[27]; Cervera-Ferri et al., 2012[6]; Trent and Menard, 2010[52]).
The hippocampo-septal fibers receive local axon collaterals from pyramidal neurons, enabling them to effectively transfer the pyramidal neuron synchrony to the septum. On the other hand, hippocampal GABAergic inhibition of MS/DB complex fibers could be strong enough to suppress MS/DB complex fibers firing, e.g. during hippocampal sharp waves or theta oscillation. Therefore, changes in hippocampal oscillatory activity can shift oscillatory activity of MS/DB complex neurons to the same direction without impacting significantly on their firing rate (Khakpai et al., 2012[27]).
References
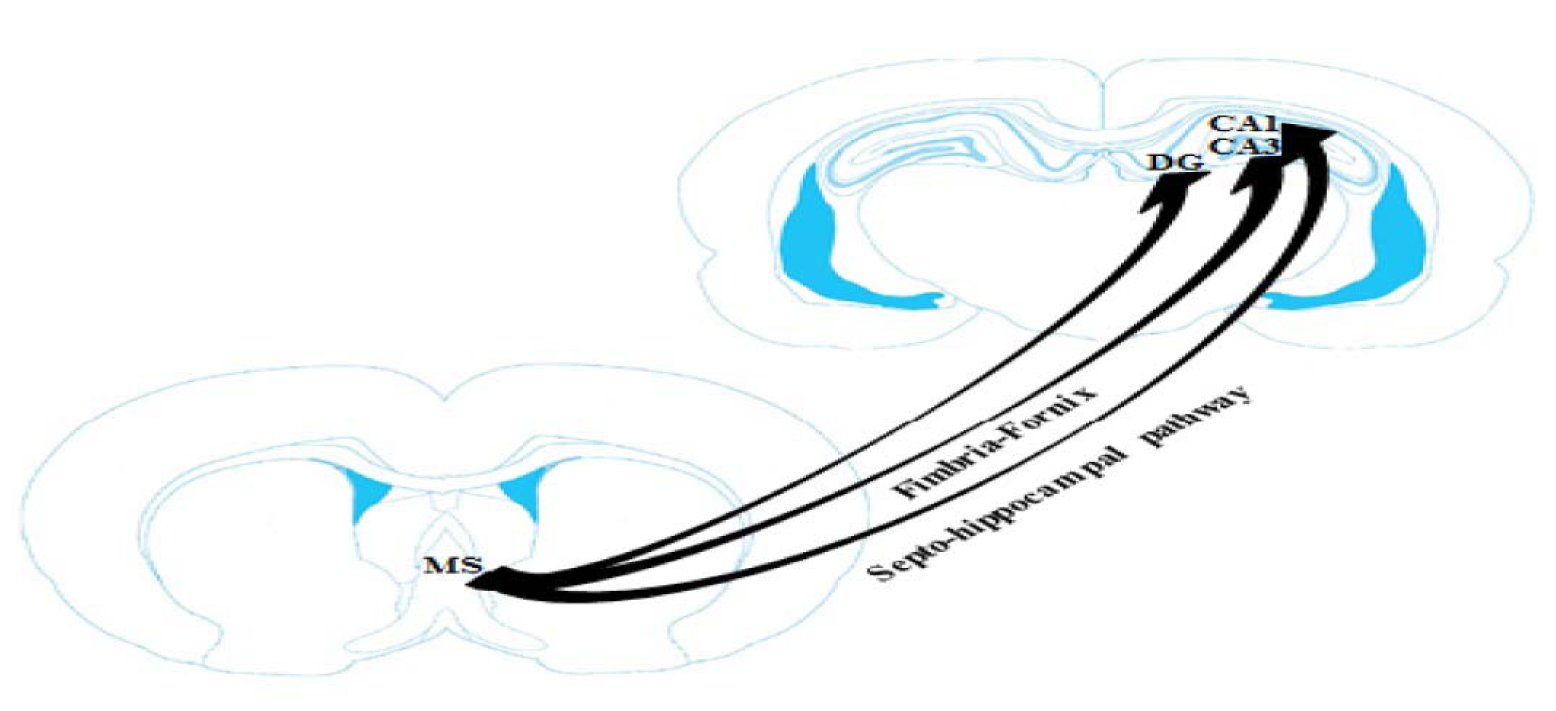
Figure 1: Schematic illustration of septo-hippocampal pathway. This pathway plays an important role in memory processes (Khakpai et al., 2012). For details see text in Section “Septo-hippocampal pathway” and “Memory functions of septo-hippocampal pathway”. MS: medial septum and DG: dentate gyrus
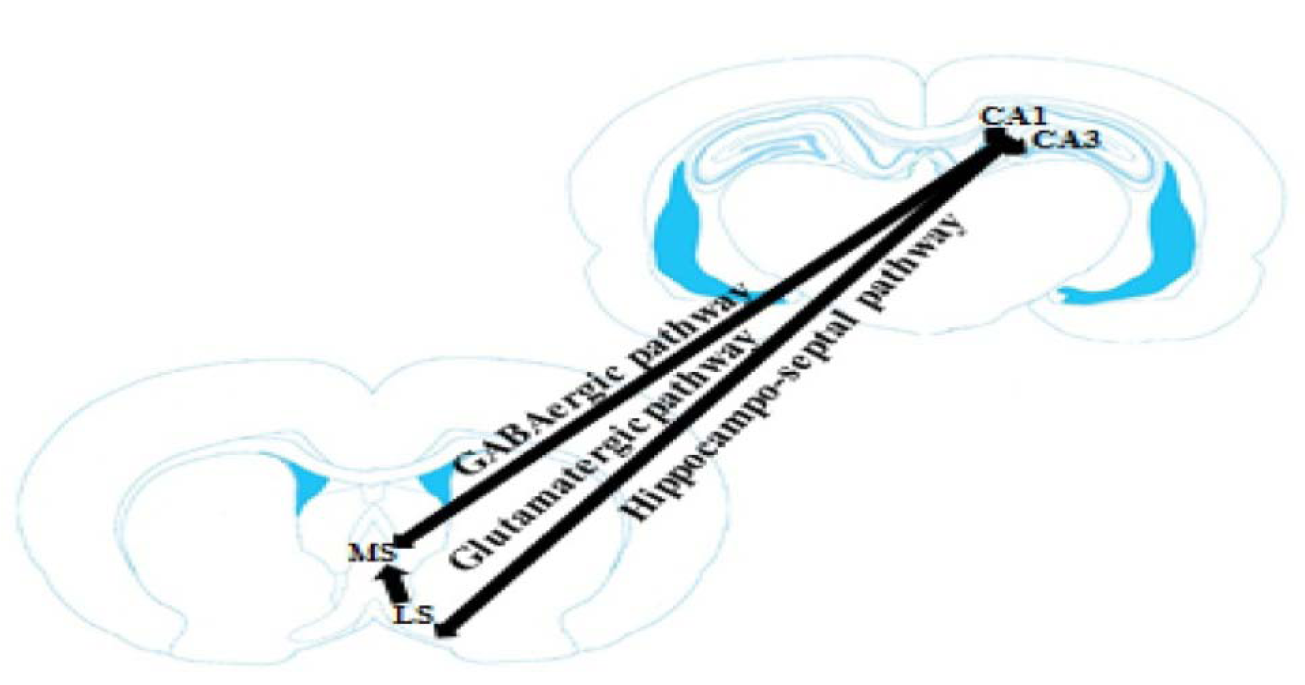
Figure 2: Schematic illustration of hippocampo-septal pathway. This pathway plays an important role in cognitive processes (Okada and Okaichi, 2010). For details see text in Section “Hippocampo-septal pathway” and “Memory functions of hippocampo-septal pathway”. MS: medial septum and lateral septum: LS
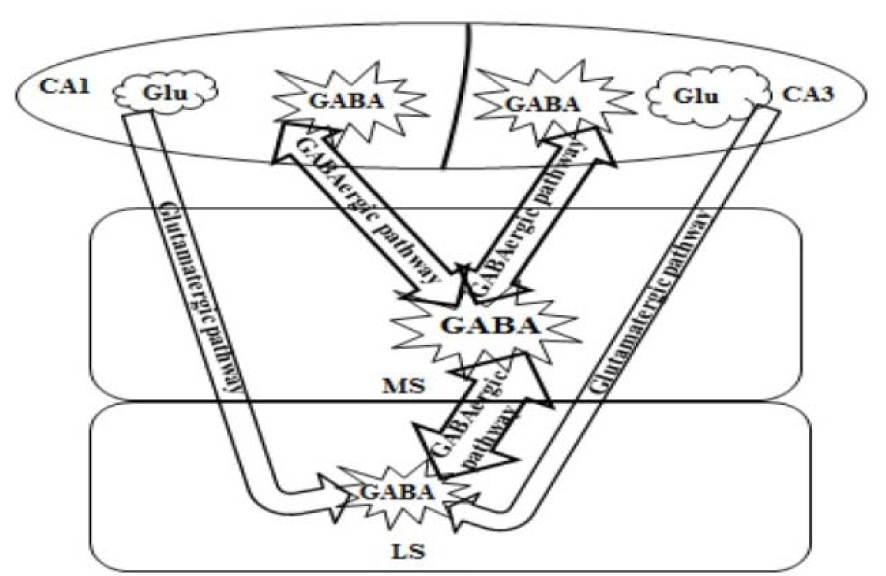
Figure 3: Schematic diagram summarizing the organization of the septo-hippocampal and hippocampo-septal pathways (Trent and Menard, 2010). Medial septum: MS and lateral septum: LS
[*] Corresponding Author:
Fatemeh Khakpai, Department of Biology, Science and Research Branch, Islamic Azad University, Tehran, Iran, P.O.Box 1637679813, Tel: +9821-88421475, eMail: Khakpai@gmail.com