Research article
Optimization of dilute acid pretreatment of water hyacinth biomass for enzymatic hydrolysis and ethanol production
Muhammad Idrees1[*], Ahmad Adnan1, Shahzad Sheikh2, Fahim Ashraf Qureshic3
1Department of Chemistry, GC University, Katchery Road, Lahore 54000, Pakistan2Drug Control and Traditional Medicine Division, National Institute of Health, Chak Shahzad, Islamabad, Pakistan
3Office of Research, Innovation and Commercialization, COMSATS Institute of Information Technology, Chak Shahzad, Park Road, Islamabad 45600, Pakistan
EXCLI J 2013;12:Doc30
Abstract
The present study was conducted for the optimization of pretreatment process that was used for enzymatic hydrolysis of lignocellulosic biomass (Water Hyacinth, WH), which is a renewable resource for the production of bioethanol with decentralized availability. Response surface methodology has been employed for the optimization of temperature (oC), time (hr) and different concentrations of maleic acid (MA), sulfuric acid (SA) and phosphoric acid (PA) that seemed to be significant variables with P < 0.05. High F and R2 values and low P-value for hydrolysis yield indicated the model predictability. The pretreated biomass producing 39.96 g/l, 39.86 g/l and 37.9 g/l of reducing sugars during enzymatic hydrolysis with yield 79.93, 78.71 and 75.9 % from PA, MA and SA treated respectively. The order of catalytic effectiveness for hydrolysis yield was found to be phosphoric acid > maleic acid > sulfuric acid. Mixture of sugars was obtained during dilute acid pretreatment with glucose being the most prominent sugar while pure glucose was obtained during enzymatic hydrolysis. The resulting sugars, obtained during enzymatic hydrolysis were finally fermented to ethanol, with yield 0.484 g/g of reducing sugars which is 95 % of theoretical yield (0.51 g/g glucose) by using commercial baker's yeast (Sacchromyces cerveasiae).
Keywords: optimization, biomass, maleic acid, water hyacinth, Sacchroomyces cervesiae
Introduction
Bioethanol is considered as an effective fuel produced from biomass (Mukhopadhyay et al., 2008[21]). Fossil fuel with its too much use, has led to an increasing demand for alternative source for fuel (Zaldivar et al., 2001[34]). Nowadays ethanol is produced from sugar and starch for use as fuel. However, the demand of these sources will not be sufficient to meet the current need of fuel industry, due to their use as a food source (Hahn-Hägerdal et al., 2006[10]). Agricultural residue is gaining much importance in these days because of its low cost, easy and decentralized availability for use in biological production of industrial chemicals such as lactic acid, acetic acid, propionic acid and fuels (Wyman et al., 1992[33]).
Enzymatic hydrolysis of cellulosic material is the most promising approach to get high yield of the monomeric sugars which is vital for the economic success of the described method (Hinman et al., 1992[11]; Lynd et al., 1996[17]). The successful enzymatic hydrolysis of the biomass can be performed with the help of suitable pretreatment method which will enable the enzyme to hydrolyze the cellulose and hemicellulose by removing the lignin and decreasing the degree of crystallinity of the cellulose. Many kinds of physical, chemical and biological pretreatments have been reported in literature (Esteghlalian et al., 1997[5]). There is a need to optimize the pretreatment method by choosing a suitable chemical which will provide maximum enzymatic hydrolysis (Mishima et al., 2006[20]).
Water hyacinth [Eichhornia crassipes (Martius-Solms) Laubach] is an aquatic weed, which grows in wetlands, marshes, sluggish flowing water, large lakes, rivers, shallow ponds and reservoirs (Naseema et al., 2004[22]). Water hyacinth is a fast growing aquatic plant widely distributed throughout the world (Malik, 2007[18]). Many attempts have been made on the potentials and constraints of using WH for numerous applications i.e., paper, craft, ropes and furniture (Gunnarsson and Petersen 2007[9]). Much attention has been given for its use as a food product due to its high protein content and richness in vitamin A (Neogi and Rajagopal, 1949[23]). Bioconversion of water hyacinth into biogas and bioethanol using different yeast is currently investigated in a number of developing countries like India and Pakistan.
The aim of this work was to determine the optimum pretreatment condition to maximize the hydrolysis yield of cellulosic material into reducing sugars by using response surface methodology (RSM). For optimization of pretreatment condition biomass residue, amount of reducing sugars in pretreatment step and enzymatic hydrolysis yield were studied in detail. Finally, fermentation of these reducing sugars into ethanol was also evaluated by using Sacchroomyces cervesiae.
Materials and Methods
Chemicals and water hyacinth
All chemicals were of analytical grade, used without further purification. ACCELLERASE 1500 and OPTIMASH™ BG were obtained from Genencor International Inc. Water hyacinth with long stem was collected from a natural pond near Shahdhra, Lahore, Pakistan during December 2011. The WH was thoroughly washed several times with tap water to remove any adhering dirt, chopped into small pieces (1-2 cm), and dried in a hot air oven at 105 °C for 6 h. Finally, the cleaned and dried WH was ground into powdered form. The dried powder material was reserved at room temperature for further work.
Optimization of pretreatment process
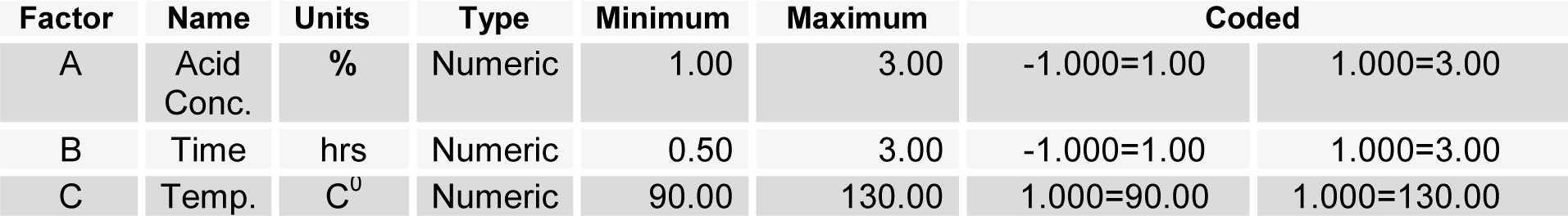
The acid concentration (X1), time (X2) and temperature (X3) for pretreatment, was optimized by using central composite design experiment to enhance the enzymatic hydrolysis yield. The design matrix with eighteen experimental runs in two blocks with four replicates of the midpoint was used. Coded values of independent variable along with their minimum and maximum values are shown in Table 1(Tab. 1). The model used to enhance the response by optimizing the pretreatment factors, was a second-order polynomial as follows:
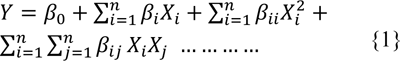
where Y is the measured response (Hydrolysis yield) and i, j are linear and quadratic coefficients respectively, β0 is the regression coefficient, X1-X3 are the coded factors under study. Regression analysis and estimation of the coefficient were performed using Design Expert Software 8.1.07.
Enzymatic hydrolysis of pretreated material
Accellerase 1500 has multiple enzyme activities and is effective for cellulose, hemicellulose and ß-glucans. Optimash™ BG is a mixture of xylanase and ß-glucanase which was used together with Accellerase 1500 to enhance the enzymatic hydrolysis efficiency of hemicellulose. The enzymatic hydrolysis was carried out in 250 ml Erlenmeyer flasks. Accellrase 1500, 0.2 ml/g dry weight of biomass and 0.1 ml/g Optimesh BG were used. Five grams of pretreated dry mass of WH was added in each flask separately. pH of the reaction mixture was set at 4.8 by adding 100 ml of 0.1M acetate buffer solution. The flasks were kept in orbital shaker for 48 h at 50 °C at 160 rpm. After regular time intervals, samples were taken from each flask and kept in boiling water to inactivate the enzyme. Each sample was filtered on a Whatman filter paper and subsequently analyzed. Each experiment was performed in duplicate.
Ethanol fermentation
Saccharomyces cerevisiae (commercial Baker's yeast) was used for the ethanol fermentation. Inoculum was prepared by transferring some cells into 250 ml flask containing 50 ml of culture medium containing 10 g/l yeast extract, 20 g/l peptone, and 20 g/l glucose and was subsequently incubated at 30 oC for 24 h. This was used to inoculate the fermentation medium. Cellulosic hydrolyzate, obtained from enzymatic hydrolysis of acid treated WH, was supplemented with 1 g/l yeast extract, 2 g/l (NH4)2SO4 and 1 g of MgSO4. The inoculum to solution ratio of 1:10 was used for fermentation purposes. Samples, for glucose and ethanol analysis, were taken at the beginning and end of a 24 h fermentation process.
Analysis of reducing sugars and ethanol
Reducing sugars produced during hydrolysis were determined by using Ghose (Ghose, 1987[7]) method. 3,5-dinitrosalicylic acid (DNS) was used as a coloring reagent, the absorbance for each sample was recorded at λ=546 nm with double beam spectrophotometer (7200). Identification of monosaccharide contents was determined with the help of thin layer chromatography (TLC) in pretreated, enzymatic hydrolyzate and in fermentation media (Hyeon et al., 2009[12]). The mobile phase for TLC analysis consisted of acetonitrile solution (acetonitrile: water, 85:15 v/v), using a 20×20 cm Kieselgel 60F 254 (Merck) as a TLC plate, and visualization of band was done with the help of α-naphthol solution. The TLC plates were soaked in 0.5 % α-naphthol and 5 % H2SO4 in ethanol and then dried in oven at 80 oC for 5 min. Sugar yield was calculated on solid pretreated biomass, using the following equation (Dedsuksophon et al., 2010[4]).
Sugar Yield (%) = 100 (sugar produced during hydrolysis/gram of biomass feedstock)
After centrifuging sample for 10 min, ethanol was quantified with the help of GC-MS (GCMS-QP2010, Shimadzu) using capillary column (DB-5, diameter 0.25 mm, length 30.0 m and thickness 0.25 µm). Nitrogen was used as a carrier gas with flow rate of 1.41 ml/min. Temperature was programmed as: maintained the temperature at 40 °C for 1min, increased at 44 °C with 15 °C/min, rose with 1 °C/min up to 50 °C and then continuously increased to 250 °C with rate of 25 °C/min, finally stayed at 250 °C for 2 min and ion source temperature was 200 °C. Injection volume was 2.0 µl and data was obtained in a scan mode in the mass range of 30-120 m/z. Fragment ions, 31 m/z and 45 m/z were used for identification and quantification of ethanol respectively. Calibration curve was obtained from 0.1, 0.2, 0.3 up to 1 % v/v ethanol in HPLC grade water and their peak areas. From this, concentration of ethanol (v/v) in sample was determined which converted to w/v by multiplying it with 0.79 (specific gravity of ethanol at 20 °C).
Results and Discussion
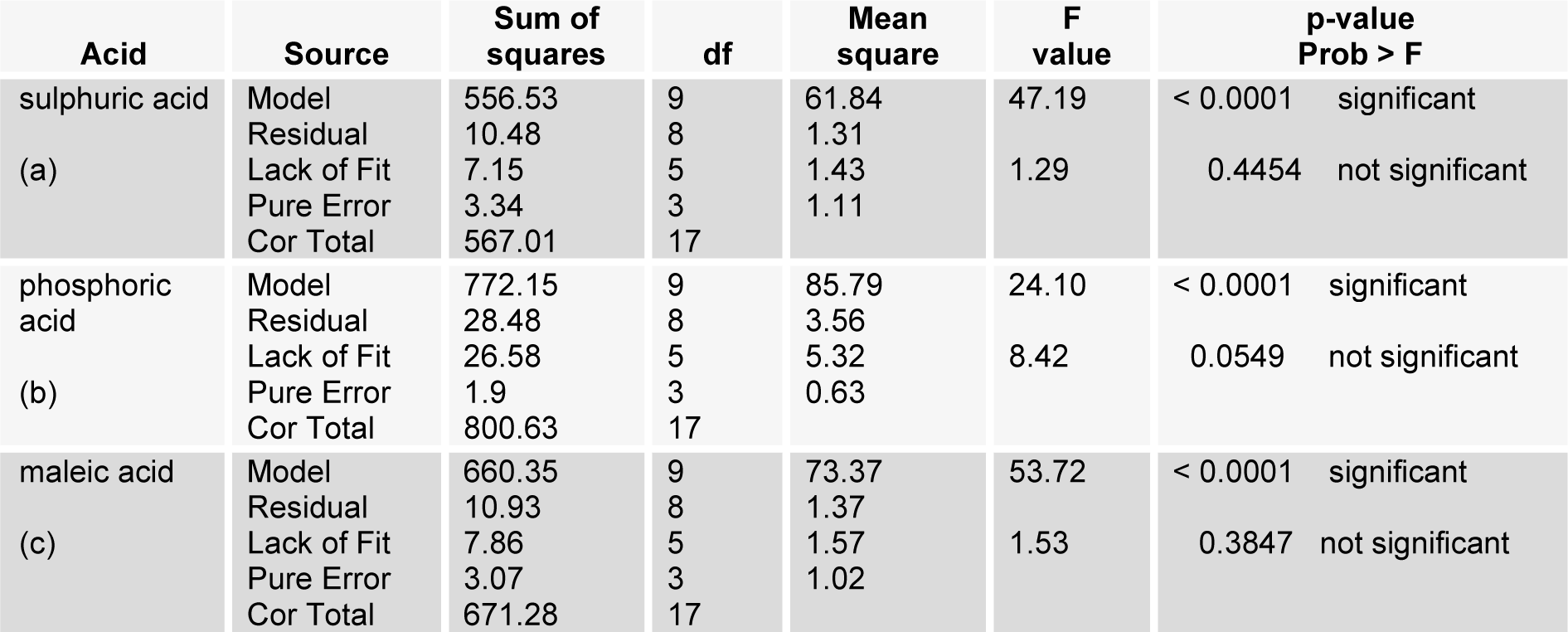
The high cellulosic content of (40-65 %) (Malik, 2007[18]; Nigam, 2002[24]) water hyacinth was considered as a potential source for production of ethanol and other fuels (Nigam, 2002[24]; Abraham & Kurup, 1996[1]; Sharma et al., 1999[28]; Singhal and Rai, 2003[29]). The maximum enzymatic hydrolysis was obtained by decreasing the crystallinity of the cellulose and hemicellulose present in the WH biomass with the help of different chemical pretreatments. Different physico-chemical pretreatments are in practice, among them dilute acid pretreatment using sulphuric acid concentration below 4 % (w/v) was economical and provide higher hydrolysis rate with cellulase enzyme (Esteghlalian et al., 1997[5]). The dilute acid pretreatment process has to be conducted under temperature range of 100-200 °C with pressure 103 KPa (15 Psi) to 517 KPa (75 Psi) for different time intervals (Wen et al., 2004[32]). Comparative pretreatments have been performed with sulphuric acid, phosphoric acid and maleic acid to enhance the hydrolysis yield at different conditions of temperature, time and acid concentrations specified through RSM. Second order polynomial equations, giving hydrolysis yield (Y, g/l) as a function of time (h), temperature (C) and acid concentrations (%) were obtained as: (equation 2 for model 'a' (sulphuric acid), equation 3 for model 'b' (phosphoric acid), equation 4 for model 'c' (maleic acid).
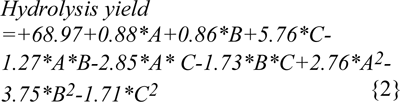
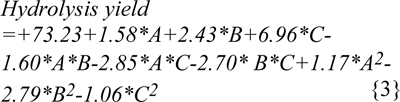
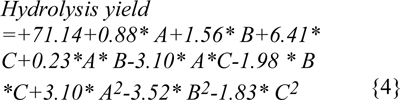
Effect of acid and its concentration
The pretreatment has shown greater influence on the sugar release through enzymatic hydrolysis of the polysaccharides present in WH. The conversion of hemicellulose during dilute acid pretreatment is predictable and has been reported before by several authors when examining hemicellulose hydrolysis of corn stover and wheat straw (Lu and Mosier, 2007[16]; Kootstra et al., 2009[15]; Radecki et al., 1988[27]; Partanen and Mroz, 1999[26]; Kabel et al., 2007[13]). Mineral and organic acids hydrolyzed the hemicellulose which results in the reducing sugars being available in the pretreated hydrolyzate (Kootstra et al., 2006[14]) and weight loss in biomass. The amount of cellulosic residue after pretreatment was found to be different for different acids at varying conditions (Table 2(Tab. 2)). The mineral acids showed more decrease in the mass of the WH biomass ranging from 20 % to 60 %, while organic acids showed 5-10 % less decrease in the mass. Sulphuric acid at higher temperature degrades the xylose and glucose into furfural more while maleic acid degrades these sugars less (Kootstra et al., 2006[14]). Approximately 36+1 % reducing sugars are available for fermentation (Ogawa et al., 2008[25]) during pretreatment step at 110-130 °C. The amount of the reducing sugars obtained after enzymatic hydrolysis, ranging from 52 % to 78 % of the WH pretreated mass. The biomass treated with phosphoric acid and maleic acid provided greater hydrolysis yield as compared to sulphuric acid. It is cleared from the results of enzymatic hydrolysis (Table 2(Tab. 2)), high concentration of acid at higher temperature of pretreatment with short time produced better hydrolysis yield (Ogawa et al., 2008[25]; Chartchalerm et al., 2007[3]). Pure glucose was obtained after enzymatic hydrolysis from the dilute acid pretreated WH biomass (Figure 1(Fig. 1)).
Effect of time on pretreatment
The cellulosic residue and the byproducts formed during pretreatment steps were different for varying time at different temperature. In conventional practice high temperature and long time for pretreatment hydrolyzed more hemicellulose for production of reducing sugar (Ogawa et al., 2008[25]). The amount of reducing sugars was less at short time at low temperature and high at high temperature and vice versa during pretreatment (Sirikarn et al., 2012[30]). The quantity of reducing sugars at higher temperature and long time was less due to degradation of the xylose and arabinose into furfural at higher temperature and long time (Ezeji et al., 2007[6]). Sulphuric acid with 1 % concentration produced 33.35 g/l of reducing sugars at 130 °C when treated for one hour while phosphoric acid produced maximum of 35.21 g/l with 3 % concentration at the same temperature and time. Maleic acid produced 36.72 g/l of reducing sugar when 1 % was used at 130 °C for one hour. The greater amount of the reducing sugars in the acid pre-hydrolyzate of the maleic acid and phosphoric acid corresponds to the non degrading nature of these acids as compared to sulphuric acid (Kootstra et al., 2006[14]). The enzymatic hydrolysis yield was influenced by the duration of the pretreatment time, low acid concentration for long time of pretreatment provided higher yield and high acid concentration with short time of pretreatment, gave higher amount of reducing sugars. Time of pretreatment significantly influences the hydrolysis yield.
Effect of temperature on pretreatment
At low temperature conditions, the solid residue available for hydrolysis has high hemicellulose content while at higher temperature maximum hemicellulose was hydrolyzed into component sugars. During acid pretreatment, hemicellulose material was converted into glucose, arabinose and xylose. With the increase in pretreatment temperature, the reducing sugars produced from hemicellulose (Ackerson et al., 1981[2]; Taherzadeh and Karimi, 2007[31]) were converted into furfural. The amount of furfural depends on the acid concentration and temperature (Gonzales et al., 1986[8]). At high temperature and time, the maximum hydrolysis of the hemicellulose was observed in pretreatment steps which results in decreasing the remaining polysaccharides or biomass (Table 1(Tab. 1)). At low temperature, the solid residue left for hydrolysis showed higher hemicellulose content while at high temperature conditions maximum hemicellulose was depicted to be hydrolyzed into component sugars. As shown in surface plots (Figures 1(Fig. 1) and 2(Fig. 2)) temperature significantly affects the hydrolysis yield. At low temperature with short time of pretreatment, hydrolysis yield was low while at high temperature hydrolysis yield was greater.
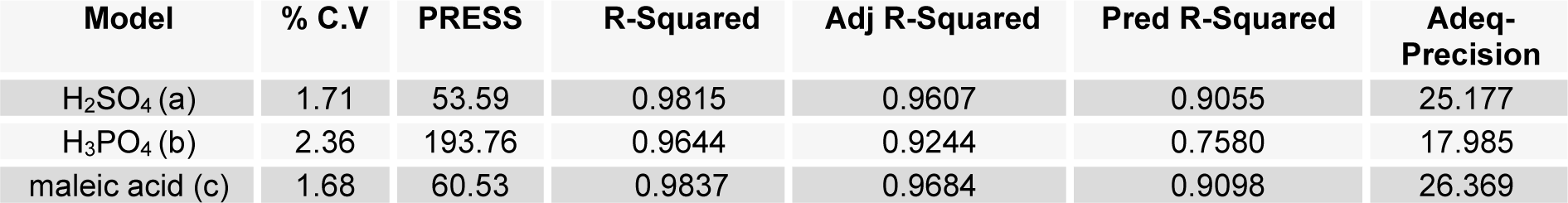
Most effective pretreatment condition
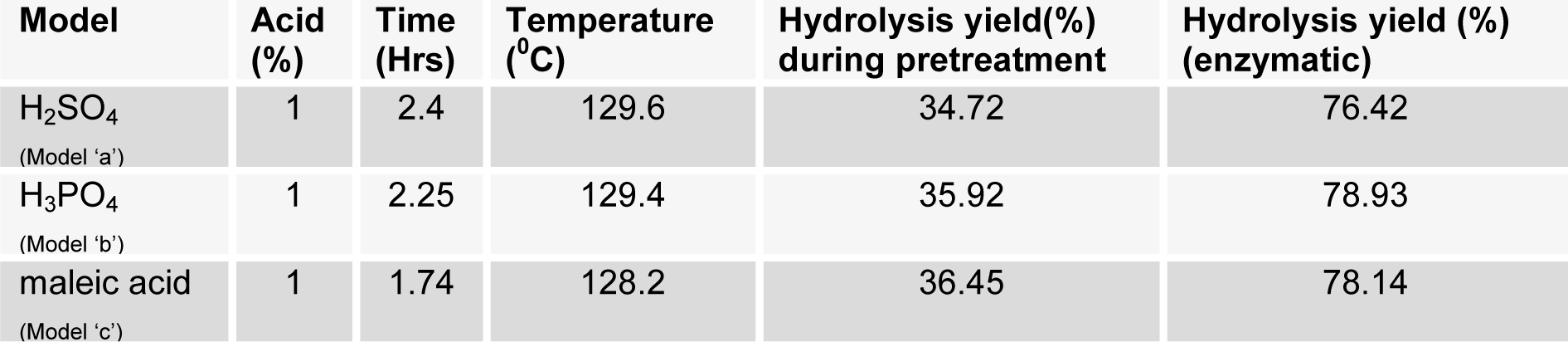
Pretreatment of biomass was executed with acids like sulphuric acid, hydrochloric acid or nitric acid (Abraham and Kurup, 1996[1]) and alkalies like NaOH or NH3 solution (Sharma et al., 1999[28]; Singhal and Rai, 2003[29]) at different temperature which efficiently improved the enzymatic hydrolysis. The pretreatment of WH biomass, either with acids or alkalies cause the loss of biomass weight by removing the lignin and hydrolysis of the hemicellulose (Table 2(Tab. 2)). At low temperature this loss is less and at high temperature more mass was lost. Mineral acids lost more mass (<50 %) as compared to maleic acid (organic acid; >50 %). To assess the effect of different reaction parameters of pretreatment on the hydrolysis yield, quadratic models were selected out of linear, 2FI, quadratic and cubic based on sequential model testing, lack of fit test and model summery statistic (Table 3(Tab. 3)). The aptness of the model was verified through different diagnostic checks. Residuals were found to follow the normality and the plots of predicted versus actual yield (Figure 3(Fig. 3)) also ascertained the overall appropriateness of the suggested model. Analysis of variance (ANOVA) for the quadratic models (a, b, c), describe significance of linear terms i.e., acid concentration, time and temperature of pretreatment, were found to be significant at 5 % level. First order interaction terms for model a and b are significant while in model 'c' acid concentration* pretreatment time was not at 5 % level. All the quadratic terms show significant contribution. The optimum hydrolysis yield (predicted) 76.42 % for model 'a', 81.2 % for model 'b' and 80.3 % for model 'c' were obtained from optimized parameters (Table 4(Tab. 4)). To validate optimum yield, experiments with specified conditions were performed. Resultant hydrolysis yield (Table 5(Tab. 5)) 75.9 % for model 'a', 79.59 % for model 'b' and 78.78 % for model 'c' showed that model was predictive and useful for the optimization of the pretreatment conditions.
Ethanol production from enzymatic hydrolyzate
Ethanol fermentation was conducted on the hydrolyzate obtained from the enzymatic hydrolysis of WH with the most effective pretreatment conditions. In 24 hr, 39.7 g/l, 39.1 g/l and 37.01 g/l fermentable sugars were converted into 19.23 g/l, 18.94 g/l and 17.93 g/l of ethanol, equivalent to 95 % (0.484 g/g) of the theoretical yield of the glucose which was 0.51 g ethanol/g of glucose. Previously 94 % ethanol yield was obtained from corn cob enzymatic hydrolyzate (Ming et al., 2007[19]) and 18 g/l of ethanol from acid hydrolyzate of water hyacinth has been reported by Nigam (2002[24]). DNS analysis and TLC images show that there were no sugars (glucose) left in the fermenting media after 24 h. The acid pretreatment completely hydrolyzed the hemicellulose present in WH plant body, resulting in pure cellulose for enzymatic saccharification, produced pure glucose for fermentation into ethanol by common Baker's yeast.
Conclusions
Water hyacinth could be hydrolyzed chemo-enzymatically in two steps i.e., pretreatment and enzymatic hydrolysis. This study shows that response surface methodology is predictive in nature for optimizing the pretreatment of biomass for ethanol production with high F and R2 values and low p-value. Phosphoric and maleic acid credited higher enzymatic (79 %) hydrolysis as compared to sulphuric acid. These sugars successfully converted into ethanol with 95 % yield. Water hyacinth, through chemo-enzymatic hydrolysis seems to be a potential source for fermentable sugars and bioethanol production at industrial level.
Acknowledgements
We gratefully acknowledge the financial support from Higher Education Commission Islamabad, Pakistan and Genencor International Inc.(Singapore) for providing enzyme samples.
References
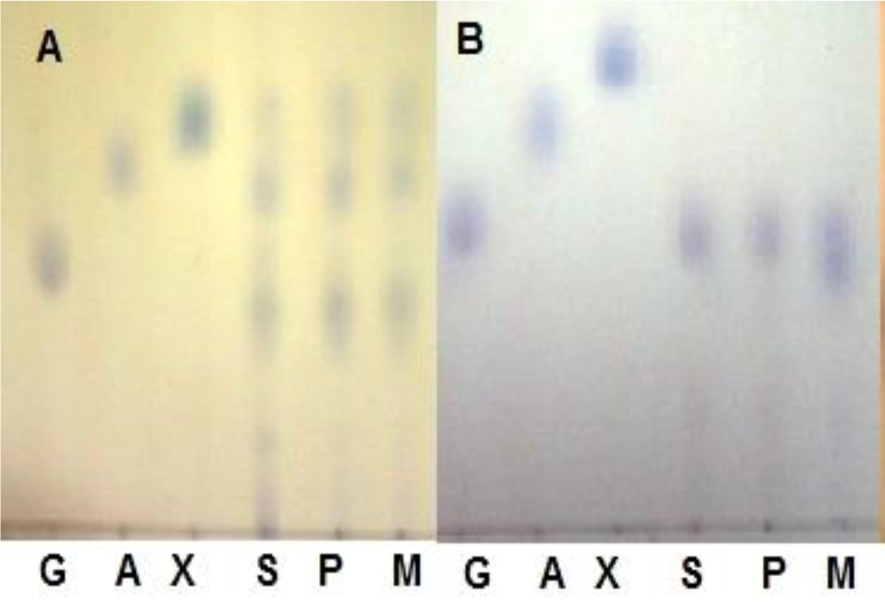
Figure 1: A: TLC images showing reducing sugars in acid hydrolyzate (pretreatment step), B: In enzymatic hydrolyzate (G: glucose, X: xylose, A: arabinose, S: sulphuric acid, P: phosphoric acid, M: maleic acid)
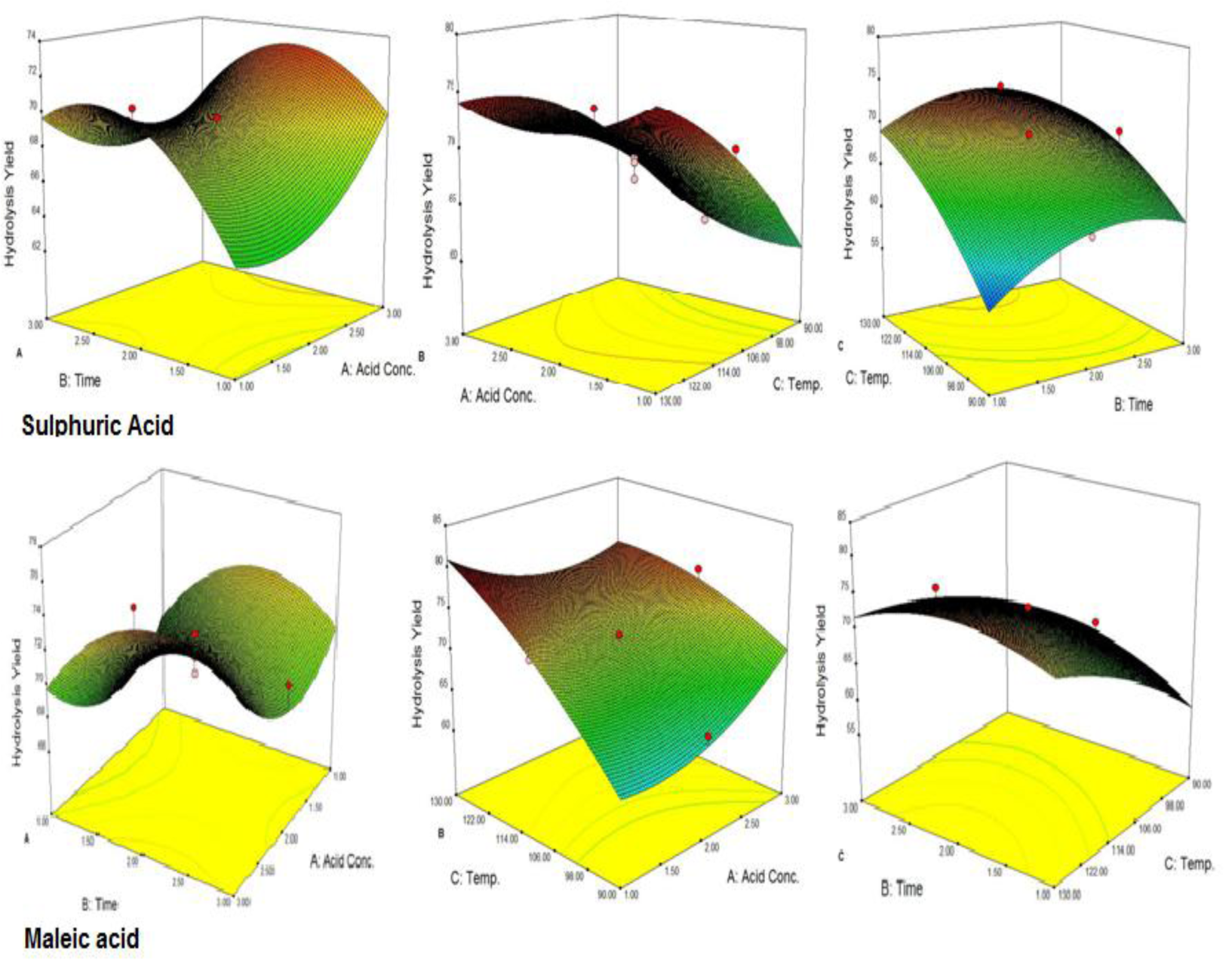
Figure 2: Interaction effect of independent variables on hydrolysis yield from sulphuric acid and maleic acid treated WH; A: (acid vs. time), B: (acid vs. temp.), C: (time vs. temp.).
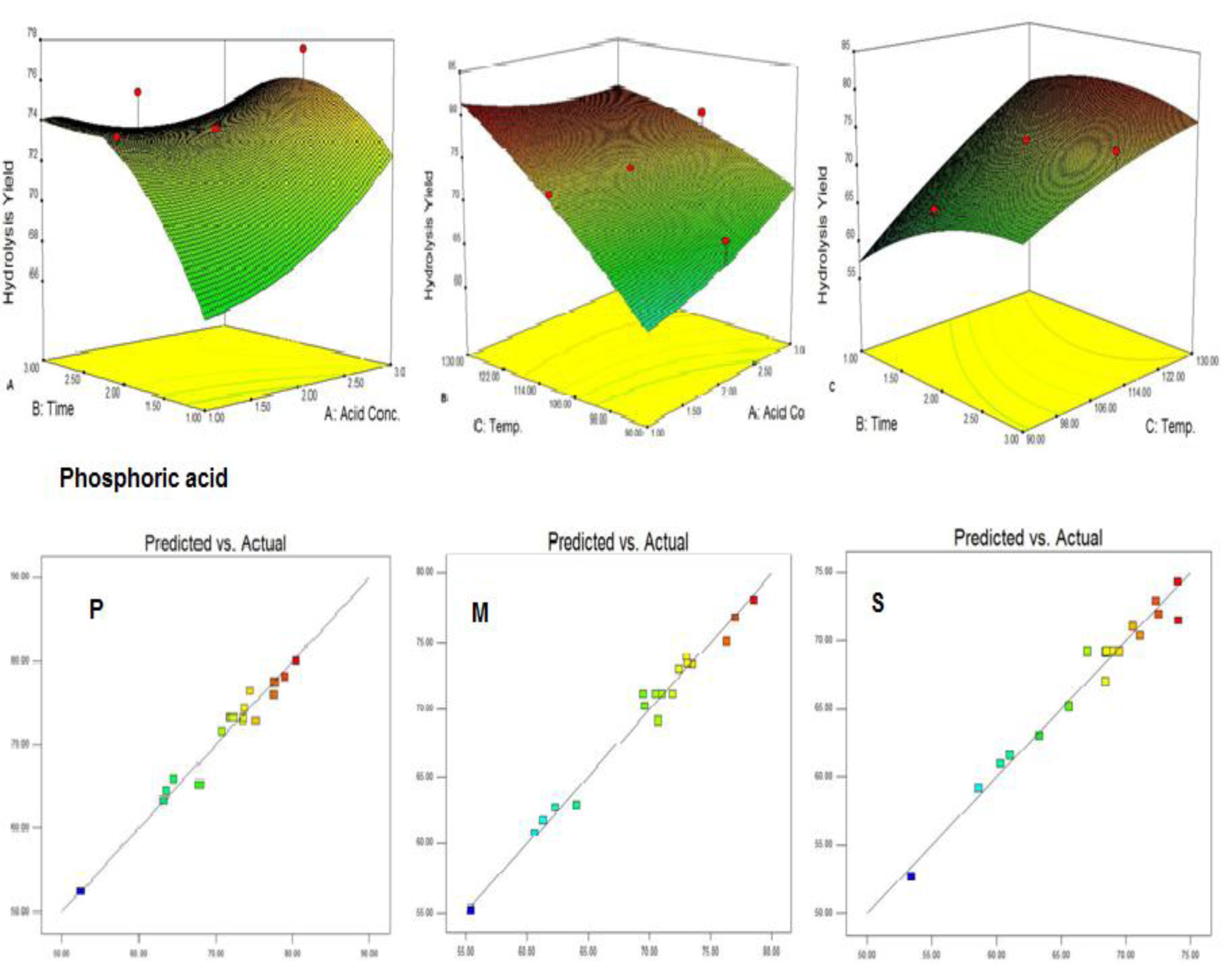
Figure 3: Interaction effect of independent variables on hydrolysis yield from phosphoric acid treated WH; A: (acid vs. time), B: (acid vs. temp.), C: (time vs. temp.), predicted vs. actual (P: phosphoric acids, M: maleic acid, S: sulphuric acid).
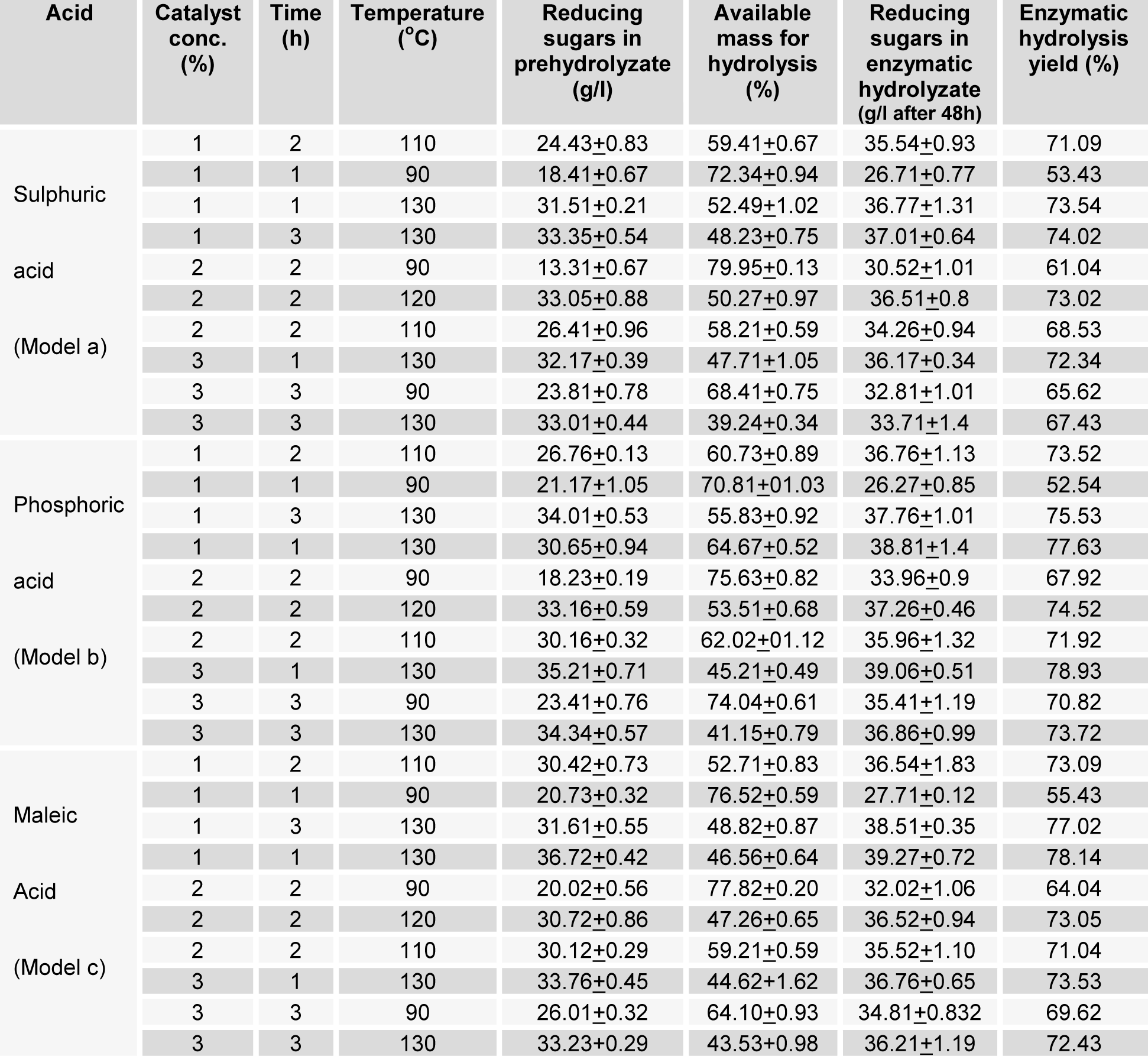
Table 2: Showing the residue of pretreatment, reducing sugars in pretreated and enzymatic hydrolyzate
[*] Corresponding Author:
Muhammad Idrees, Department of Chemistry, GC University, Katchery Road, Lahore 54000, Pakistan, Tel no: +92-03214230056, eMail: m.idrees20@yahoo.com