Review article
Current developments in toxicological research on arsenic
Hermann M. Bolt1
1Leibniz Research Centre for Working Environment and Human Factors (IfADo)
EXCLI J 2013;12:Doc64
Abstract
There is a plethora of recent publications on all aspects relevant to the toxicology of arsenic (As). Over centuries exposures to arsenic continue to be a major public health problem in many countries. In particular, the occurrence of high As concentrations in groundwater of Southeast Asia receives now much attention. Therefore, arsenic is a high-priority matter for toxicological research. Key exposure to As are (traditional) medicines, combustion of As-rich coal, presence of As in groundwater, and pollution due to mining activities. As-induced cardiovascular disorders and carcinogenesis present themselves as a major research focus. The high priority of this issue is now recognized politically in a number of countries, research funds have been made available. Also experimental research on toxicokinetics and toxicodynamics and on modes of toxic action is moving very rapidly. The matter is of high regulatory concern, and effective preventive measures are required in a number of countries.
Keywords: arsenic, research focus, history, toxicology
Introduction
Toxicological research on arsenic is of increased importance in recent years, world-wide, and recent publications cover all aspects of toxicology. Trends are visible in these publications, which are analysed in this review against the historical background. This review describes recent trends, as opposed to classical knowledge. It is an updated extension of an editorial published in Archives of Toxicology (Bolt, 2012[8]).
Inorganic arsenic is acutely toxic, and ingestion of large doses leads to gastrointestinal symptoms, disturbances of cardiovascular and nervous system functions, and eventually death. Bone marrow depression, haemolysis, hepatomegaly, melanosis, polyneuropathy and encephalopathy may be observed. Chronic human exposure has been linked to a variety of dermal symptoms (exfoliative dermatitis, keratosis, vitiligo, skin cancer), peripheral neuropathy, encephalopathy, bronchitis, pulmonary fibrosis, hepato-splenomegaly, portal hypertension, peripheral vascular disease/ “blackfoot disease”, atherosclerosis, cancer and diabetes mellitus (Pimparker and Bhave, 2010[67]). According to WHO, long-term exposure to arsenic in drinking water is causally related to increased risks of cancer in the skin, lungs, bladder and kidney, as well as to skin changes such as hyperkeratosis and pigmentation changes. The current WHO drinking water guideline value is 10 µg As/L. Exposure-response relationships have been observed for a variety of end-points, and increased risks of lung and bladder cancer and of arsenic-associated skin lesions are reported to be associated with drinking water concentrations ≥ 50 µg As/L.
Historical Remarks
Early knowledge on the toxicity of arsenicals has accumulated in Asian civilizations, where especially arsenic sulphide (realgar) had been traditionally used, both as a medicine and a poison. According to Lewin (1920[48]) such knowledge arrived at the Hellenic Mediterranean world following the conquests of Alexander the Great. Since then, there has been a continuous European history of both use of arsenic as a medicine and abuse as a homicide, until recently (Lewin, 1920[48]). For instance, Napoleon's health began rapidly to fail in February 1821. He suffered from chronic As intoxication when he died on May 5, 1821, as evidenced by analysis of his hair (Kintz et al., 2007[44]; see Table 1(Tab. 1)) (References in Table 1: (Pre-)Historical: Brothwell and Grime (2002[13]); Kintz et al., 2007[44]; Coal combustion: Baoshan et al., 2005[6]; Bencko et al., 2009[7]; Mining: Wu et al., 2011[84]; Liu et al., 2009[52]; Liu et al., 2010[53]; Food and drinking water: Shah et al., 2011[71]; Roychowdhury, 2010[68]; Uchino et al., 2006[78]; Maity et al., 2012[56]; Gault et al., 2008[30]; Hanh et al., 2011[34]; Concha et al., 2006[21]; Yanez et al., 2005[87]; de Fátima Pinheiro Pereira et al., 2010[22]; Controls: Baoshan et al. (2005[6]); de Fátima Pinheiro Pereira et al., 2010[22]; Moreno-Santini et al., 2012[61]; Brima et al., 2006[12]). Biomonitoring of As in human hair is regarded as a useful tool in field research evaluating chronically elevated As exposures (Pandey et al., 2007[65]; Orloff et al., 2009[64]; Kazi et al., 2011[42]), although being less indicative at low (non-elevated) exposures (Cleland et al., 2009[19]). A number of very recent environmental studies are making use of this parameter (Geier et al., 2012[31]; Evrenoglou et al., 2012[28]; Marchiset-Ferlay et al., 2012[58]; Savabieasfahani et al., 2012[70]; Miklavčič et al., 2013[60]).
By contrast to its intentional use, arsenic as a chronic human environmental toxicant has an even much longer history by a couple of thousand years. At present, the oldest example of environmental exposure to arsenic is testified by hair analysis of the Tyrolean Neolithic mummy called “Ötzi”, who died of homicide between 3359 and 3105 BC (Oeggl, 2009[63]). In his case, copper enrichment near the hair surface and high arsenic contents in keratin cells of the interior of the hair shaft strongly argue in favour of a long-term involvement in copper working, accompanied by high arsenic exposure (Brothwell and Grime, 2002[13]). Table 1(Tab. 1) compares these data with contemporary concentrations of arsenic in hair. Regarding historical aspects, a challenging theory is being advanced that desertion of several important Etruscan settlements at the end of the Archaic period in Italy had been a consequence of man-made pollution and associated arsenic environmental poisoning (Harrison et al., 2010[35]).
The textbook-relevant case of arsenic-related peripheral vascular disease was found in the early 20th century, as an endemic along the Southwestern coast of Taiwan. This disease involved the lower extremities, characterised by typical clinical symptoms of progressive arterial occlusion; it was called "blackfoot disease" because of the gangrenous appearance of feet of patients. The prevalence of the disease ranged from 6.51 to 18.85 per 1,000 capita in different villages. Epidemiologic studies revealed that blackfoot disease was associated with the consumption of fossil Artesian well water containing high levels of arsenic. Co-occurrence of blackfoot disease and arsenic-related skin lesions such as hyperpigmentation, hyperkeratosis, and skin cancer was also observed. Recent studies confirmed the association also of preclinical peripheral vascular disease with arsenic exposure. The incidence of clinical manifestation of blackfoot disease decreased dramatically after the installation of tap water in these villages over the last 2-3 decades of the 20th century (Tseng, 2002[76]). Thus, the number of publications on this classical disease has now decreased.
In essence, it is evident that over centuries exposures to arsenic continue to be a public major health problem in many countries (Golka et al., 2010[32]). In particular, the occurrence of high As concentrations in groundwaters of Southeast Asia has received much attention in the past decade (Kim et al., 2011[43]). Therefore, arsenic is a high-priority matter for toxicological research in a number of countries (Bolt and Hengstler, 2011[10]).
Contemporary Arsenic Exposures
An analysis of the literature reveals the following key exposures to arsenic:
- Heavy metals, including arsenic, are being used in traditional medicines in Asia, which is being addressed as a major toxicological problem, for instance in China and India (Liu et al., 2008[54]; Kamath et al., 2012[41]). Recently, arsenic trioxide is being recommended for a very special medicinal use, i.e. in the treatment of acute promyelocytic leukaemia (Emadi and Gore, 2010[26]).
- As can be seen from Table 1(Tab. 1), the combustion of As-rich coal is a major reason for chronic intoxication, especially in distinct areas of China (Baoshan et al., 2005[6]; Wang et al., 2007[80]; Lin et al., 2010[50]). As a worst-case reported from the Chinese Southwest Province of Guizhou, coal with an As content of up to 35,000 ppm is burned indoors in open pits for daily cooking and crop drying. In a few villages, approximately 3,000 patients with skin lesions indicative of chronic arseniosis had been detected by 1998 (Baoshan et al., 2005[6]). Genetic polymorphisms at XPD/ERCC2 appear to modulate the risk for arsenic-related skin lesions in such patients (Lin et al., 2010[50]).
- High As concentrations in groundwater, especially in Southeast Asia, have received much attention. This refers to parts of India and Bangladesh, with groundwater levels equal or greater than 200 µg/L (Guha Mazumder and Dasgupta, 2011[33]), and especially the floodplain areas along the Mekong river (Laos, Cambodia, Vietnam). The source of elevated As concentrations in these groundwaters is connected with the release of As from river sediments, and it is estimated that more than 10 million residents in Southeast Asia are presently at risk from consuming As-contaminated groundwater (Kim et al., 2011[43]). Data from Latin America show that this problem is not restricted to Asia. It has been estimated that some 4.5 million people in Latin America are chronically exposed to high levels of As (> 50 µg/L drinking water), with extremes up to 2000 µg/L (McClintock et al., 2012[59]). Such situations are being further investigated (de Fátima Pinheiro Pereira et al., 2010[22]). As mentioned initially, the World Health Organization has recommended a provisional guideline value of 10 µg/L for arsenic in drinking water (WHO, 1996[83]).
- High groundwater levels of As and increased exposure of the general population occur also in conjunction with mining activities. In 2012, examples are being reported from Portugal (Coelho et al., 2012[20]), India (Chakraborti et al., 2012[14]) and China (Li et al., 2012[49]).
- Co-exposure and possible interaction of As with other environmental toxicants is also a field of public concern. Most important appears to be co-exposure to fluoride, both by ground water pollution (Chouhan and Flora, 2010[18]) and by indoor combustion of coal (Lin et al., 2012[51]). Also, interactions with iron (Kumasaka et al., 2012[46]) and with nutritional deficiencies (Deb et al., 2012[23]) are a matter of present research.
Current Research Focus
The environmental situation of exposures to As has triggered the main avenues of present research. So far, only few environmental studies include a speciation of As (e.g., inorganic trivalent/pentavalent forms), and the present results are not entirely uniform (Sanz et al., 2007[69]; de Fátima Pinheiro Pereira et al., 2010[22]). Therefore, it ought to be expected that As speciation research will further develop.
Connected with this field, a focal research point of the last years has been adsorption, distribution, metabolism and excretion (ADME) of As species/compounds (Dopp et al., 2008[25], 2010[24]; Bolt and Stewart, 2010[11]; Chang et al., 2012[15]; Watanabe and Hirano, 2012[81]). It was soon recognized that the toxicities of As(III) and As(V) are different (Laib and Moritz, 1989[47]), and that the toxicity of As(V) results in part from its reduction to As(III) (Huang and Lee, 1996[37]). Metabolism of inorganic As proceeds mainly by a sequence of repetitive reduction and oxidative methylation steps, the latter mediated by arsenic methyl transferase (CYT19). A highly cited key publication by Hayakawa et al. (2005[36]) has demonstrated that arsenic-glutathione complexes are substrates for the human CYT19. In general, toxicokinetics of As species remain an important experimental research focus (Kobayashi et al., 2008[45]; Naraharisetti et al., 2008[62]; Juárez-Reyes et al., 2009[40]).
As mentioned above, there may be relevant environmental co-exposures of As with other inorganic compounds that lead to combined action, with questions of the mechanisms involved. For instance, Yajima et al. (2012[86]) address apoptotic mechanisms induced by co-exposure with barium, which can additionally be present in As-containing drinking-water wells. Other publications address co-exposures of arsenic and fluoride. Thus, Lin et al. (2012[51]) describe a population-based study in a rural area in Northwest China with a large number of cases diseased with a combination of arseniasis and fluorosis. The causal factor was again indoor combustion of coal rich in both As and F, which led to extremely high co-exposures via the inhalation route. In an experimental study on rabbit aorta as a cardiovascular target, Ma et al. (2012[55]) show that inflammatory responses play a critical role in the combined cardiovascular toxicity of As and F.
Indeed, with regard to the different manifestations of toxicity of arsenic (Singh et al., 2011[73]), cardiovascular disorders, such as hypertension, atherosclerosis and myocardial injury, are receiving increased interest (Manna et al., 2008[57]; Balakumar and Kaur, 2009[5]; Chen et al., 2012[16]; Wang et al., 2012[79]). A mechanism likely to be involved is oxidative stress, connected with activation of eNOS and enhanced the phosphorylation of MLCK (Singh et al., 2011[73]). The entire field of As-induced oxidative stress and related signalling pathways is clearly of increasing scientific relevance (Flora, 2011[29]; Jomova and Valko, 2011[39]; Jomova et al., 2011[38]; Tseng et al., 2012[77]; Sinha et al., 2012[74])
Mechanisms of As-induced carcinogenesis are an important area of research (Pastoret et al., 2012[66]; Anwar-Mohamed et al., 2012[4]). Oxidative stress plays a pivotal role (Shi et al., 2004[72]). The public health impact of malignancies caused by arsenic is obvious (Bolt, 1991[9]). Authors from leading governmental U.S. institutions (NTP, NIEHS, NCI) raised serious concern of transplacental carcinogenesis by a specific As compound, methylarsenous acid (Tokar et al., 2012[75]). Against such a background, the priority of current research into mechanisms of As-induced malignant transformation is evident (Xu et al., 2012[85]).
There is a remarkable development of genetic polymorphism studies, in conjunction with epidemiological research in As-exposed populations. A diversity of polymorphic enzymes is presently being investigated, including XPD/ERCC2 (Lin et al., 2010[50]), arsenic methyl transferase (Agusa et al., 2009[2], 2010[3]), glutathione S-transferases (Agusa et al., 2010[1]), Mn-superoxide dismutase and 8-oxoguanine DNA glycosylase (Chen et al., 2012[17]). A study by Escobar-García et al. (2012[27]) suggests a specific role of the glutathione S-transferase GST01-1 in As-mediated inflammatory response and apoptotic processes, and the idea is being put forward that A140D and E208K polymorphisms increase the risk for inflammatory and apoptosis-related diseases in As-exposed populations. The role of GST01 polymorphisms as a modifying factor of As toxicity has been confirmed in another study (Chen et al., 2012[16]). Gene-environmental interactions focussing on human As exposures and As-induced effects are a very attractive and rapidly developing research area.
Conclusion
In essence, there is a very rapid recent development of research into human environmental exposures to arsenic and related As-induced diseases, which are highly relevant to public health in a number of countries. It appears that the high priority of this issue is being recognized politically in a number of countries, and that research funds have been made available. In this context, experimental research on As toxicokinetics and toxicodynamics and on modes of toxic action is moving very rapidly. The matter is of high regulatory concern, and preventive measures are urgently required in a number of countries.
References
1.
Agusa T, Iwata H, Fujihara J, Kunito T, Takeshita H, Minh TB et al. Genetic polymorphisms in glutathione S-transferase (GST) superfamily and arsenic metabolism in residets of the Red River Delta, Vietnam. Toxicol Appl Pharmacol. 2010;242:352-62.2.
Agusa T, Iwata H, Jujihara J, Kunito T, Takeshita H, Minh TB et al. Genetic polymorphisms in AS3MT and arsenic metabolism in residents of the Red River Delta, Vietnam. Toxicol Appl Pharmacol. 2009;236:131-41.3.
Agusa T, Kunito T, Kubota R, Inoue S, Fujihara JU, Minh TB et al. Exposure, metabolism, and health effects of arsenic in residents from arsenic-contaminated groundwater areas of Vietnam and Cambodia: a review. Rev Environ Health. 2010;25:193-220.4.
Anwar-Mohamed A, Abdelhamid G, Amara IEA, E-Kadi AOS. Differential modulation of aryl hydrocarbon receptor regulated enzymes by arsenite in the kidney, lung, and heart of C57BL/6 mice. Arch Toxicol. 2012;86:897-910.5.
Balakumar P, Kaur J. Arsenic exposure and cardiovascular disorders: an overview. Cardiovasc Toxicol. 2009;9:169-76.6.
Baoshan Z, Binbin W, Zhenhua D, Daixing Z, Yunshu Z, Chen Z et al. Endemic arseniosis caused by indoor combustion of high-As coal in Guizhou Province, P.R. China. Environ Geochem Health. 2005;27:521-8.7.
Bencko V, Rames J, Fabiánová E, Pesek J, Jakubis M. Ecological and human health risk aspects of burning arsenic-rich coal. Environ Geochem Health. 2009;31:339-43.8.
Bolt HM. Arsenic: an ancient toxicant of continuous public health impact, from Iseman Ötzi until now. Arch Toxicol. 2012;86:825-30.9.
Bolt HM. Carcinogens relevant to the bronchopulmonary system [Article in German]. Pneumologie. 1991;45:407-9.10.
Bolt HM, Hengstler JG. Most cited articles: ethanol-induced hepatotoxicity, anticarcinogenic effects of polyphenolic compounds in tear, dose-response modelling, novel roles of epoxide hydrolase and arsenic-induced suicidal erythrocyte death. Arch Toxicol. 2011;85:1485-9.11.
Bolt HM, Stewart JD. Arsenic: metabolism and transport mechanisms in human hepatocytes. Arch Toxicol. 2010;84:1-2.12.
Brima EI, Haris PI, Jenkins RO, Polya DA, Gault AG, Harrington CF. Understanding arsenic metabolism through a comparative study of arsenic levels in the urine, hair and fingernails of healthy volunteers from three unexposed ethnic groups in the United Kingdom. Toxicol Appl Pharmacol. 2006;216:122-30.13.
Brothwell D, Grime G. The analysis of the hair of the Neolithic Iceman. In: Lynnerup N, Andreasen C, Berglund J (eds): Mummies in a new millenium. Proceedings of the 4th World Congress on Mummy Studies. Nuuk, Greenland, Sept 4-10, 2001 (pp 66-9). Greenland National Museum and Archives and Danish Polar Center, 2002.14.
Chakraborti D, Rahman MM, Murrill M, Das R, Siddayya, Patil SG et al. Environmental arsenic contamination and its health effects in a historic gold mining area of the Mangalur greenstone belt of Northeastern Karnataka, India. J Harzard Mater. 2012. 2012, pii:S0304-3894(12)01007-2. doi: 10.1016/j.jhazmat.2012.10.00215.
Chang YY, Kuo TC, Hsu CH, Hou DR, Kao YH, Huang RN. Characterization of the role of protein-cysteine residues in the binding with sodium arsenite. Arch Toxicol. 2012;86:911-22.16.
Chen JW, Wang SL, Wang YH, Sun CW, Huang YL, Chen CJ, Li WF. Arsenic methylation, GST01 polymorphisms, and metabolis syndrome in an arseniasis endemic area of Southwestern Taiwan. Chemosphere. 2012;88:432-8.17.
Chen SC, Chen CC, Kuo CY, Huang CH, Lin CH, Lu ZY et al. Elevated risk of hypertension induced by arsenic exposure in Taiwanese rural residents: possible effects of manganese superoxide dismutase (MnSOD) and 8-oxoguanine DNA glycolylase (OGG1) genes. Arch Toxicol. 2012;86:869-78.18.
Chouhan S, Flora SJ. Arsenic and fluoride: two major ground water pollutants. Indian J Exp Biol. 2010;48:666-78.19.
Cleland B, Tsuchiya A, Kalman DA, Dills R, Burbacher TM, White JW et al. Arsenic exposure within the Korean community (United States) based on dietary behavior and arsenic levels in hair, urine, air, and water. Environ Health Perspect. 2009;117:632-8.20.
Coelho P, Costa S, Silva S, Walter A, Ranville J, Sousa AC et al. Metal(loid) levels in biological matrices from human populations exposed to mining contamination – Panasqueira Mine (Potugal). J Toxicol Environ Health. 2012;75A:893-908.21.
Concha G, Nermell B, Vahter M. Spatial and temporal variations in arsenic exposure via drinking-water in Northern Argentina. J Health Popul Nutr. 2006;24:317-26.22.
de Fátima Pinheiro Pereira S, Saraiva AF, de Alencar MI, Ronan DE, de Alencar WS, Oliveira GR et al. Arsenic in the hair of individuals in Santana-AP-Brazil: significance of residence location. Bull Environ Contam Toxicol. 2010;84:368-72.23.
Deb D, Biswas A, Ghose A, Das A, Majumdar KK, Guha Mazumder DN. Nutritional deficiency and arsenical manifestations: a perspective study in an arsenic-endemic region of West Bengal, India. Public Health Nutr. 2012;27:1-12.24.
Dopp E, Klingerman AD, Diaz-Bone RA. Organoarsenicals. Uptake, metabolism, and toxicity. Met Ions Life Sci. 2010;7:231-65.25.
Dopp E, von Recklinghausen U, Hartmann LM, Stueckradt I, Pollok I, Rabieh S et al. Subcellular distribution of inorganic and methylated arsenic compounds in human urothelial cells and human hepatocytes. Drug Metab Dispos. 2008;36:971-9.26.
Emadi A, Gore SD. Arsenic trioxide – an old drug rediscovered. Blood Rev. 2010;24:191-9.27.
Escobar-García DM, Del Razo LM, Sanchez-Peña LC, Mandeville PB, Lopez-Campos C, Escudero-Lourdes C. Association of glutathione S-transferase Ω polymorphisms (A140D and E208K) with the expression of interleukin 8 (IL-8), transforming growth factor beta (TGF-β) and apoptotic protease activating factor 1 (Apaf-1) in humans chronically exposed to arsenic in drinking water. Arch Toxicol. 2012;86:857-68.28.
Evrenoglou L, Partsinevelou SA, Stamatis P, Lazaris A, Patsouris E, Kotampasi C et al. Children exposure to trace levels of heavy metals at the North zone of Kifissos river. Sci Total Environ. 2012;443C:650-61.29.
Flora SJ. Arsenic-induced oxidative stress and its reversibility. Free Radic Biol Med. 2011;51:257-81.30.
Gault AG, Rowland HA, Charnock JM, Wogelius RA, Gomez-Morilla I, Vong S et al. Arsenic in hair and nails of individuals exposed to arsenic-rich groundwaters in Kandal province, Cambodia. Sci Total Environ. 2008;393:168-76.31.
Geier DA, Kern JK, King PG, Sykes LK, Geier MR. Hair toxic metal concentrations and autism spectrum disorder severity in young children. Int J Environ Res Public Health. 2012;9:4486-97.32.
Golka K, Hengstler JG, Marchan R, Bolt HM. Severe arsenic poisoning: one of the largest man-made catastrophies. Arch Toxicol. 2010;84:583-4.33.
Guha Mazumder D, Dasgupta UB. Chronic arsenic toxicity: studies in West Bengal, India. Kaohsiung J Med Sci. 2011;27:360-70.34.
Hanh HT, Kim KW, Bang S, Hoa NM. Community exposure to arsenic in the Mekong river delta, Southern Vietnam. J Environ Monit. 2011;13:2025-32.35.
Harrison AP, Cattani I, Turfa JM. Metallurgy, environmental pollution and the decline of Etruscan civilisation. Environ Sci Pollut Res. 2010;17:165-80.36.
Hayakawa T, Kobayashi Y Cui X, Hirano S. A new metabolic pathway of arsenite: arsenic-glutathione complexes are substrates for human methyltransferase Cyt19. Arch Toxicol. 2005;79:183-91.37.
Huang RN, Lee TC. Cellular uptake of trivalent arsenite and pentavalent arsenate in KB cells cultures in phosphate-free medium. Toxicol Appl Pharmacol. 1996;136:243-9.38.
Jomova K, Jenisova Z, Feszterova M, Baros S, Liska J, Hudecova D et al. Arsenic: toxicity, oxidative stress and human disease. J Appl Toxicol. 2011;31:95-107.39.
Jomova K, Valko M. Advances in metal-induced oxidative stress and human disease. Toxicology. 2011;283:65-87.40.
Juárez-Reyes A, Jiménez-Capdeville ME, Delgado JM, Ortiz-Pérez D. Time course of arsenic species in the brain and liver of mice after oral administration of arsenate. Arch Toxicol. 2009;83:557-563.41.
Kamath SU, Pemiah B, Sekar RK, Krishnaswamy S, Sethuraman S, Krishnan UM. Mercury-based traditional herbo-metallic preparations: a toxicological perspective. Arch Toxicol. 2012;86:831-8.42.
Kazi TG, Baig JA, Shah AQ, Kandhro GA, Khan S, Afridi, HI et al. Determination of arsenic in scalp hair samples from exposed subjects using microwave–assisted digestion with and without enrichment based on cloud point extraction by electrothermal atomic absorption spectrometry. J AOAC Int. 2011;94:293-9.43.
Kim KW, Chanpiwat P, Hanh HT, Phan K, Sthiannopkao S. Arsenic geochemistry of groundwater in Southeast Asia. Front Med. 2011;5:420-33.44.
Kintz P, Ginet M, Merques N, Cirimele V. Arsenic speciation of two specimens of Napoleon’s hair. Forensic Sci Int. 2007;170:204-645.
Kobayashi Y, Negishi T, Mizumura A, Watanabe T, Hirano S. Distribution and excretion of arsenic in Cynomologus monkey following repeated administration of diphenylarsinic acid. Arch Toxicol. 2008;82:553-61.46.
Kumasaka MY, Yamanoshita O, Shimizu S, Ohnuma S, Furuta A, Yajima I et al. Enhanced carcinogenicity by coexposure to arsenic and iron and a novel remediation system for the elements in well drinking water. Arch Toxicol. 2012 [Epub ahead of print 26 Oct. 2012].47.
Laib RJ, Moritz M. Investigation of tumor initiating and/or cocarcinogenic properties of arsenite and arsenate with the rat liver foci bioassay. Exp Pathol. 1989;37:231-3.48.
Lewin L. Die Gifte in der Weltgeschichte (pp 157-66). Berlin: Springer, 1929.49.
Li Y, Thang X, Yang L, Li H. Levels of Cd, Pb, As, Hg, and Se in hair of residents living in villages around Fenghuang polymetallic mine, Southwestern China. Bull Environ Contam Toxicol. 2012;89:125-8.50.
Lin GF, Du H, Chen JG, Lu HC, Guo WC, Golka K et al. Association of XPD/ERCC2 G23591A and A35931C polymorphisms with skin lesion prevalence in a multi-ethnic, arseniasis-hyperendemic village exposed to indoor combustion of high arsenic coal. Arch Toxicol. 2010;84:17-24.51.
Lin GF, Gong SY, Wei C, Chen JG, Golka K, Shen JH. Co-occurrence of arseniasis and fluorosis due to indoor combustion of high fluorine and arsenic content coal in a rural township on Northwest China: epidemiological and toxicological aspects. Arch Toxicol. 2012;86:839-47.52.
Liu BJ, Wu FC, Deng QJ, Mo CL, Zhu J, Zeng L et al. Pollution characteristics of antimony, arsenic and mercury in human hair at Xikuangshan antimony mining area and Guyang City, China (in Chinese). Huan Jing Ke Xue. 2009;30:907-12.53.
Liu CP, Luo CL, Gao Y, Li FB, Lin LW, Wu CA et al. Arsenic contamination and potential health risk implications at an abandoned tungsten mine, Southern China. Environ Pollut. 2010;158:820-6.54.
Liu J, Lu Y, Wu Q, Goyer RA, Waalkes MP. Mineral arsenicals in traditional medicines: orpiment, realgar, and arsenolite. J Pharmacol Exp Ther. 2008;326:363-8.55.
Ma Y, Niu R, Sun Z, Wang J, Luo G, Zhang J et al. Inflammatory responses induced by fluoride and arsenic at toxic concentration in rabbit aorta. Arch Toxicol. 2012;86:849-56.56.
Maity JP, Nath B, Kar S, Chen CY, Banerjee S, Jean JS et al. Arsenic-induced health crisis in peri-urban Moyna and Ardebok villages, West Bengal, India: an exposure assessment study. Environ Geochem Health. 2012;34:563-74.57.
Manna P, Sinha M, Sil PC. Arsenic-induced oxidative myocardial injury: protective role of arjunolic acid. Arch Toxicol. 2008;82:137-49.58.
Marchiset-Ferlay N, Savanovitch C, Sauvant-Rochat MP. What is the best biomarker to assess arsenic exposure via drinking water? Environ Int. 2012;39:150-71.59.
McClintock TR, Chen Y, Bundschuh J, Oliver JT, Navoni J, Olmos V et al. Arsenic exposures in Latin America: biomarkers, risk assessments and related health effects. Sci Total Environ. 2012;429:76-91.60.
Miklavčič A, Casetta A, Snoj Tratnik J, Mazej D, Krsnik M, Mariuz M et al. Mercury, arsenic and selenium exposure levels in relation to fish consumption in the Mediterranean area. Environ Res. 2013;120:7-17.61.
Moreno-Santini V, Mansilla-Rivera I, García-Rodríguez O, Rodríguez-Sierra CJ. A pilot study determining hair arsenic and lead levels in residents of a community on a former landfill in Puerto Rico. Bull Environ Contam Toxicol. 2012;89:572-6.62.
Naraharisetti SB, Aggarwal M, Sarkar SN, Malik JK. Concurrent subacute exposure to arsenic through drinking water and malathion via diet in male rats: effects on hepatic drug-metabolizing enzymes. Arch Toxicol. 2008;82:543-51.63.
Oeggl K. The significance of the Tyrolean Iceman for the archeobotany of Central Europe. Veget Hist Archeobot. 2009;18:1-11.64.
Orloff K, Mistry K, Metcalf S. Biomonitoring for environmental exposures to arsenic. J Toxicol Environ Health B Crit Rev. 2009;12:509-24.65.
Pandey PK, Yadav S, Pandey M. Human arsenic poisoning issues in Central-East Indian locations: biomarkers and biochemical monitoring. Int J Environ Res Public Health. 2007;4:15-22.66.
Pastoret A, Marcos R, Sampayo-Reyes A, Saucedo-Cardenas O, Lozano-Garza GH, Hernandez A. Inhibition of hepatocyte nuclear 1 and 4 alpha (HNF1alpha and HNF4alpha as a mechanism of arsenic carcinogenesis. Arch Toxicol. 2012 [Epub ahead of print 5 Oct. 2012].67.
Pimparker BD, Bhave A. Arsenicosis: review of recent advances. J Assoc Physicians India. 2010;58:617-24.68.
Roychowdhury T. Groundwater arsenic contamination in one of the 107 arsenic-affected blocks in West Bengal, India: status, distribution, health effects and factors responsible for arsenic poisoning. Int J Environ Health. 2010;213:414-27.69.
Sanz E, Muñoz-Olivas R, Cámara C, Sengupta MK, Ahamed S. Arsenic speciation in rice, straw, hair and nails samples from the arsenic-affected areas of Middle and Lower Ganga plain. J Environ Sci Health A Tox Hazard Subst Environ Eng. 2007;42:1695-705.70.
Savabieasfahani M, Hoseiny M, Goodarzi S. Toxic and essential trace metals in first baby haircuts and mother hair from Imam Hossein Hospital Tehran, Iran (2012). Bull Environ Contam Toxicol. 2012;88:140-4.71.
Shah AQ, Kazi TG, Baig JA, Afridi HI. Correlation between arsenic concentration in fish and human scalp hair of people living in arsenic-contaminated and non-contaminated areas of Pakistan. Biol Trace Elem Res. 2011;144:197-204.72.
Shi H, Shi X, Liu KJ. Oxidative mechanism of arsenic toxicity and carcinogenesis. Mol Cell Biochem. 2004;255:67-78.73.
Singh AP, Goel RK, Kaur T. Mechanisms pertaining to arsenic toxicity. Toxicol Int. 2011;18:87-93.74.
Sinha D, Biswas J, Bishayee A. Nrf2-mediated redox signalling in arsenic carcinogenesis: a review. Arch Toxicol. 2012 [Epub ahead of print 23 Aug. 2012].75.
Tokar EJ, Diwan BA, Thomas DJ, Waalkes MP. Tumors and proliferative lesions in adult offspring after maternal exposure to methylarsenous acid during gestation in CD1 mice. Arch Toxicol. 2012;86:575-82.76.
Tseng CH. An overview on peripheral vascular disease in blackfood disease-hyperendemic villages in Taiwan. Angiology. 2002;53:529-37.77.
Tseng HY, Liu ZM, Hunag HS. NADPH oxidase-produced superoxide mediates EGFR transactivation by c-Src in arsenic trioxide-stimulated human keratinocytes. Arch Toxicol. 2012;86:935-45.78.
Uchino T, Roychaudhury T, Ando M, Tokunaga H. Intake of arsenic from water, food composites and excretion through urine, hair from a studied population in West Bengal, India. Food Chem Toxicol. 2006;44:455-61.79.
Wang L, Kou MC, Weng CY, Hu LW, Wang Yj, Wu MJ. Arsenic modulates heme oxygenase-1, interleukin-6, and vascular endothelial growth factor expression in endothelial cells: roles of ROS, NFκ-B, and MAPK pathways. Arch Toxicol. 2012;86:879-96.80.
Wang Z, Guo X, Bai G, Lei Y. Analysis of risk factors of skin lesions from burning high-arsenic-contaminated coal in Southern Shanxi Province (in Chinese). Wei Sheng Yan Jiu. 2007;36:343-6.81.
Watanabe T, Hirano S. Metabolism of arsenic and its toxicological relevance. Arch Toxicol. 2012 [Epub ahead of print. 19 Jul. 2012].82.
WHO. Arsenic and arsenic compounds. 2nd ed. 2001;Geneva: World Health Organization, 2001. (Environmental Health Criteria Monographs, No. 224).83.
WHO. Guidelines for drinking-water quality. 2nd ed. Vol 2. Health criteria and other supporting information. Geneva: World Health Organization, 1996.84.
Wu F, Liu B, Mo C, Chen B, Corns W, Liao H. Health risk associated with dietary co-exposure toi high levels of antimony and arsenic in the world’s largest antimony mine area. Sci Total Environ. 2011;409:3344-51.85.
Xu Y, Li Y, Pang Y, Ling M, Shen L, Jiang R et al. Blockade of p53 by HIF-2α, but not HIF-1α, is involved in arsenite-induced malignant transformation of human bronchial epithelial cells. Arch Toxicol. 2012;86:947-59.86.
Yajima I, Uemura N, Nizam S, Khalequzzaman Md, Thang ND, Kumasaka MY et al. Barium inhibits arsenic-mediated apoptotic cell death in human squameous cell carcinoma cells. Arch Toxicol. 2012;86:961-73.87.
Yanez J, Fierro V, Mansilla H, Figueroa L, Cornejo L, Barnes RM. Arsenic speciation in human hair: a new perspective for epidemiological assessment in chronic arsenicism. J Environ Monit. 2005;7:1335-41.
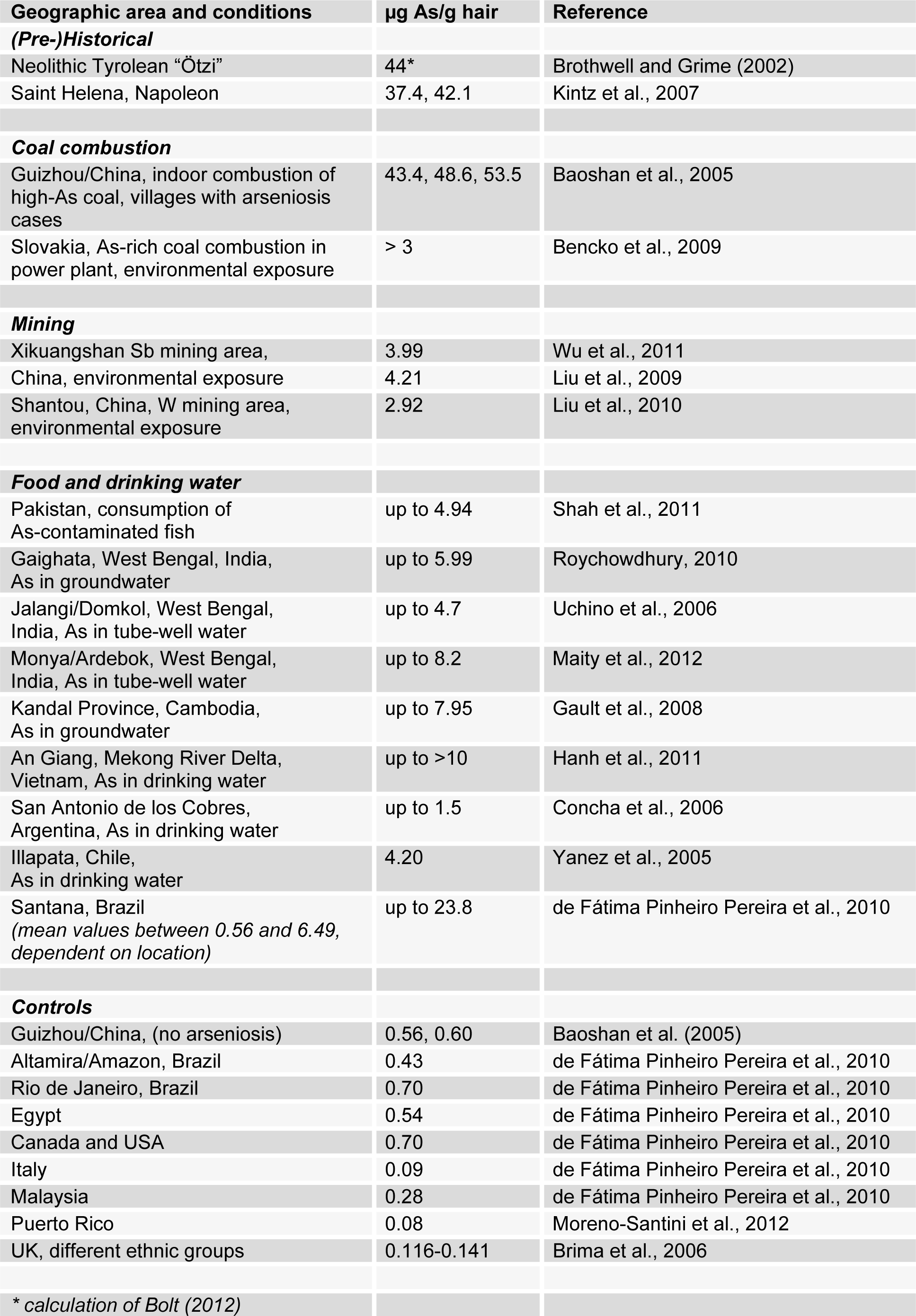
Table 1: Recently reported elevated As levels in human hair specimens (mean values; single data, when indicated)
