Research article
High glucose inhibits osteogenic differentiation through the BMP signaling pathway in bone mesenchymal stem cells in mice
Juncheng Wang1, Bin Wang2, Ying Li1, Dongsheng Wang1, E Lingling1, Yang Bai1, Hongchen Liu1[*]
1Department of Stomatology, Chinese PLA General Hospital and Postgraduate Military Medical School, Beijing 100853, China2Department of Stomatology, Chinese PLA The Military General Hospital of Beijing, Beijing 100700, China
EXCLI J 2013;12:Doc584
Abstract
Patients with diabetes tend to have an increased risk of osteoporosis that may be related to hyperglycemia. In vitro evidence has shown that high glucose can affect the proliferation and osteogenic differentiation of mesenchymal stem cells (MSCs). Tissue regeneration depends mainly on MSCs. However, the exact mechanisms involved in high glucose-induced bone loss remain unknown. In this study, we investigated the effects of high glucose on the proliferation and osteogenic differentiation of mice bone MSCs (BMSCs) and determined the specific mechanism of bone morphogenetic protein 2 (BMP-2) in the osteogenic differentiation of mice BMSCs in a high-glucose microenvironment. High glucose (< 25 mM) promoted cell growth but suppressed mineralization. The intracellular BMP-2 level in BMSCs cultured in a high-glucose microenvironment was significantly decreased and suppressed activation of the BMP signaling pathway. Consequently, expression of the osteogenic markers Runx2, alkaline phosphatase, and osteocalcin were decreased. Meanwhile, supplementation with ectogenic BMP-2 reversed the cell osteogenic differentiation and osteogenic marker down-regulation under high glucose. Our data indicate that BMP-2 plays an important role in regulating the osteogenic differentiation of BMSCs in a high-glucose microenvironment. Thus, it is possible that agents modifying this pathway could be used by BMSCs to promote bone regeneration in high-glucose microenvironments.
Keywords: bone mesenchymal stem cells, molecular pathways, high glucose, osteogenic differentiation, BMP-2
Introduction
Diabetes mellitus, a metabolic disease characterized by hyperglycemia, can cause disorder of bone metabolism, leading to bone loss or osteoporosis, a bone condition defined by low bone mass and decreased bone quality (Duarte et al., 2005[9]; Schwartz, 2003[31]). Moreover, diabetes is one of the risk factors that causes periodontal disease and alveolar bone loss (Al-Emadi et al., 2006[1]). Javed and Romanos (2009[15]) reported that poorly controlled diabetes negatively affected implant osseointegration, but osseointegration could successfully take place in patients with diabetes under optimal serum glycemic control, and animal studies have confirmed this premise (de Molon et al., 2013[7]). It is well known that hyperglycemia is a major cause of complications of diabetes and that maintaining blood glucose at a level as close to normal as possible is the main treatment aim, which can restore decreased bone mineral density in diabetic patients (Krakauer et al., 1995[20]). In a streptozotocin-induced mouse model of insulin-dependent diabetes mellitus, Botolin and McCabe (2006[3]) demonstrated that diabetes-associated hyperglycemia modulated osteoblast gene expression, function, and bone formation and thereby caused diabetic bone loss. However, the exact mechanism of how high glucose induces bone loss remains unclear.
Mesenchymal stem cells (MSCs), commonly isolated from adult bone marrow, have the potential to undergo multilineage differentiation into multiple connective tissue cell types, such as osteoblasts, chondrocytes, adipocytes, and myoblasts (Tuli et al., 2003[35]). Bone MSCs (BMSCs) are an excellent source of proliferative osteoprogenitor cells that can be induced to differentiate into osteoblasts (Porter et al., 2003[29]; Walsh et al., 2001[38]). During development, osteoprogenitors pass through a series of complicated stages, including proliferation, differentiation, matrix deposition, and matrix mineralization, and eventually become mature osteoblasts. This process can be affected by the microenvironment. Liu et al. (2011[25]) found that an inflammatory microenvironment inhibited osteogenic differentiation potential but promoted the proliferation of periodontal ligament stem cells through the activation of β-catenin, which suppressed the non-canonical Wnt/ Ca2+ pathway. Gopalakrishnan et al. (2006[12]) found that high concentrations of glucose inhibited BMSC proliferation, alkaline phosphatase (ALP) activity, collagen formation, and mineralization. Wang et al. (2010[39]) noted that high glucose inhibited osteogenic differentiation and increased adipogenic differentiation by activating the cAMP/ PKA/ERK pathway in MG-63 cells. In our previous study, we found that high glucose interfered with the proliferation and differentiation of rat mandible osteoblasts (Ma et al., 2011[26]). However, the mechanism by which high glucose affects BMSC osteogenic differentiation has not been clarified.
In this study, we exposed mice BMSCs to medium containing different concentrations of glucose to test the hypothesis that high glucose stimulates BMSC proliferation but inhibits their osteogenic differentiation. We found that a high glucose concentration (< 25 mM) stimulated BMSC proliferation but inhibited osteogenic differentiation in a dose-dependent manner. Furthermore, our data demonstrated that bone morphogenetic protein 2 (BMP-2) expression was decreased by high glucose, accompanied by a decreased expression of Runx2, and this inhibition effect could be reversed with additional ectogenic BMP-2. Our finding suggests that high glucose suppresses BMSC osteogenic differentiation though the BMP signaling pathway, which may be helpful to improve the osseointegration of alevolar bone implants in patients with diabetes.
Materials and Methods
Cell culture
A frozen vial of murine BMSCs (isolated from 4-5-week-old female C57/BL6 mice; Cyagen Biosciences, Guangzhou, China) was thawed and expanded according to the supplier instructions. The BMSCs used in all experiments were passage 4 cells.
Cell growth assay
Cells were plated at a density of 5 × 103 cells/well in 96-well plates for 24 h, and then treated with Dulbecco's modified Eagle's medium (DMEM; Invitrogen Gibco Rockville,MD, USA)) containing normal (5.5 mM) or high glucose (16.5 mM, 25 mM, and 35 mM) concentrations. The MTT assay was carried out for nine days according to the cell proliferation kit protocol (Sigma, St. Louis, MO/USA). Absorbance was determined at 490 nm with a microplate reader (Bio-Tek Instruments, Winooski, VT, USA). All reported values are presented as the means of triplicate samples. This test was repeated three times.
Flow cytometric analysis
Cells were plated at a density of 3 × 105 cells/well in 6-well plates and cultured until 80 % confluence. The cells were starved using a serum-free medium containing 1% fetal bovine serum (FBS; Invitrogen Gibco) for 24 h, and then treated with DMEM (plus 10% FBS) containing normal (5.5 mM) or high glucose (16.5 mM, 25 mM, and 35 mM) concentrations, respectively. Flow cytometry analysis was performed at day 5. The fractions of cells in the G0/G1, G2, and S phases of the cell cycle were analyzed and the proliferation index was determined (Zheng et al., 2009[43]).
The apoptotic cells were measured using annexin V-FITC conjugated to propidium iodide (PI) by flow cytometry. On culture day seven, the harvested cells were assayed for apoptosis using an annexin V-FITC apoptosis detection kit (Becton Dickinson Bioscience, San Jose, CA, USA) according to the manufacturer instructions (Velagapudi et al., 2011[37]). Briefly, the cells were treated as described above, and then gently washed twice with annexin-binding buffer, detached from the plate using 0.05% trypsin (Sigma), suspended in cold binding buffer, and stained with annexin V-FITC and PI. Analysis was conducted for 2 × 104 cells using a flow cytometer with CellQuest software (Becton-Dickinson, Rutherford, NJ, USA) using FL1 and FL2 ranges for annexin V-FITC and PI, respectively. Annexin V-FITC-positive, PI-negative cells were scored as apoptotic. Double-stained cells were considered either necrotic or late apoptotic.
Mineralization assay
Osteogenic differentiation of the stem cells was performed as previously described (Pittenger et al., 1999[28]). Briefly, 3 × 105 BMSCs/well were plated in a 6-well plate and cultured in DMEM supplemented with 10% FBS until confluent. Then, the medium was changed to DMEM with 1% FBS for 24 h to starve the cells. Subsequently, the cells were incubated with osteogenic DMEM (100 nM dexamethasone, 50 μg/mL ascorbic acid, and 5 mM β-glycerophosphate; Sigma) for another 21 d. The medium contained 5.5, 16.5, 25, and 35 mM glucose, respectively. The medium was changed every 3-4 d. The cells were then fixed with 75% ethanol and stained with 2% Alizarin red S (Sigma). To quantify the Alizarin red S-stained nodules, the stain was solubilized with 0.5 mL 5% sodium dodecyl sulfate (SDS) in 0.5 N HCl for 30 min at room temperature. The solubilized stain (0.15 mL) was transferred to a 96-well plate, and the absorbance was measured at 405 nm.
Western blot analysis
3 × 105 BMSCs/well were plated in two 6-well dishes until confluent, divided into four groups, and incubated with osteogenic medium containing 5.5 or 25 mM glucose or DMEM containing 5.5 or 25 mM glucose (control). After seven days of culture, whole cell lysates were extracted with lysis buffer (10 mM Tris-HCl, 1 mM EDTA, 1% SDS, 1% Nonidet P-40, 1:100 proteinase inhibitor cocktail, 50 mM β-glycerophosphate, 50 mM sodium fluoride) for western blotting. The protein content of the lysate was determined using a protein assay kit (Beyotime) following the manufacturer's recommended protocol. The proteins were loaded on 8% SDS polyacrylamide gels, transferred to PVDF membranes (Millipore, Billerica, MA, USA) and blocked with 5% nonfat milk powder in PBST (phosphate-buffered saline with 0.1% Tween). The membranes were probed overnight with the following monoclonal primary antibodies: anti-BMP-2 (Abcam, Cambridge, UK) and anti-β-actin (Cell Signaling Technology, Beverly, MA, USA). The membranes were then incubated with anti-mouse horseradish peroxidase-conjugated secondary antibody (Boster, Wuhan, China). The blots were visualized using an enhanced chemiluminescence kit (Amersham Biosciences, Piscataway, NJ, USA) according to the manufacturer's recommended instructions.
ALP activity analysis
The ALP activity assay was performed with an ALP kit according to the manufacturer protocol (Jiancheng, Nanjing, China). Seven days after osteogenic induction, the cells were fixed with 70% ethanol and incubated with a solution of 0.25% naphthol AS-BI phosphate and 0.75% Fast Blue BB dissolved in 0.1 M Tris buffer (pH 9.3) for ALP staining. This test was repeated three times.
Quantitative real-time polymerase chain reaction
Cells were cultured and divided into four groups as above. Total RNA was isolated using TRIzol (Invitrogen) at day seven. Approximately 2-5 µg total RNA from each group was converted to cDNA with a SuperScript First-Strand Synthesis kit (Invitrogen). The quantitative real-time polymerase chain reaction (qRT-PCR) was performed using a QuantiTect SYBR Green PCR kit (Toyobo, Osaka, Japan) and the Applied Biosystems 7500 Real-time PCR Detection System (Applied Biosystems, Foster City, CA, USA). Two independent experiments were performed for each reaction in triplicate. The primers used are listed in Table 1(Tab. 1).
BMP-2 treatment
The cells were plated in 6-well dishes, and cultured and grouped as above. The cells were then treated with or without (as control) 100 ng/mL BMP-2 (Wyeth Research Genetics Institute) (Pountos et al., 2010[30]). On culture day seven, the cells were harvested and assayed for osteogenic differentiation. Whole-cell lysates were extracted with lysis buffer for western blot analysis to verify the BMP-2 content. Total RNA was isolated from eight groups and qRT-PCR was utilized to test the expression of RUNX2, ALP, and osteocalcin (OCN) mRNA.
Statistical analysis
The data were subjected to an independent samples t-test or one-way ANOVA using SPSS. The results are expressed as mean ± SD. For all analyses, P < 0.05 was used to indicate statistical significance.
Results
High-glucose microenvironment interfered with BMSC proliferation and osteogenic differentiation
As the osteogenic differentiation of MSCs would be affected by their microenvironment, we systematically explored the biological function of mouse BMSCs in a high-glucose microenvironment. We used four different glucose concentrations (5.5, 16.5, 25, and 35 mM) in our study.
It was demonstrated that, based on the glucose content, high glucose had a contrary effect on BMSC proliferation. From day five, the growth of BMSCs cultured in DMEM containing 25 mM glucose was better than that of BMSCs cultured in DMEM containing 5.5 mM glucose (P < 0.05). From day seven, DMEM containing 16.5 and 25 mM glucose both promoted BMSC proliferation. Furthermore, the medium containing 25 mM glucose exerted the best effects on BMSC proliferation in the four groups (P < 0.05). The medium containing 35 mM glucose had a notable effect on BMSC proliferation. Initially, it promoted BMSC proliferation; at day three, it was evident that BMSCs cultured in the DMEM containing 35 mM glucose had the best growth compared to the other three groups (P < 0.05). However, the BMSC proliferation deteriorated and had worse growth than the other three groups after three days (P < 0.05; Figure 1A(Fig. 1)). The flow cytometric analysis showed that the BMSC proliferation indices were as follows: 25 mM group > 16.5 mM group > 5.5 mM group > 35 mM group (P < 0.05; Table 2(Tab. 2); Figure 1B, 1D(Fig. 1)). The annexin V-FITC/PI quantification assay demonstrated that the number of apoptotic cells cultured in DMEM containing 35 mM glucose was much greater (approximately 31.28 %) than that of the other three groups (< 16 %, P < 0.05; Figure 1C, 1E(Fig. 1)).
The Alizarin red S staining showed that the mineralization of BMSCs was inhibited by high glucose (16.5 - 35 mM) on day 21 (Figure 2A(Fig. 2)). Inverted microscopy revealed that the number and size of mineral nodules decreased as the glucose concentration increased. It was extremely difficult to detect typical mineral nodules in the BMSCs grown in medium containing 35 mM glucose. The optical density values from the Alizarin red S staining quantification were 0.440, 0.303, 0.262, and 0.113 in 5.5 mM, 16.5 mM, 25 mM, and 35 mM glucose, respectively, indicating a negative relationship between osteogenic ability and high glucose concentration (P < 0.01; Figure 2B(Fig. 2)). Based on the effects of high glucose on the proliferation and osteogenic differentiation of the BMSCs, we selected 5.5 mM glucose as the normal group (control) and 25 mM glucose as the high-glucose group for further experiments.
High glucose inhibited BMP-2 activity
Recent studies have demonstrated that the BMP pathway appears to play an important role in MSC osteogenic differentiation (Lin and Hankenson, 2011[24]) and BMP-2, which is a key protein of the BMP signaling pathway, was altered in high-glucose conditions (Chen et al., 2006[4]). Thus, we postulated that the high-glucose microenvironment would inhibit the BMP signaling pathway during the osteogenic differentiation process. In the present study, the expression of BMP-2 was investigated following culture in osteogenic medium (containing 5.5 or 25 mM glucose) for seven days. Our data revealed that BMP-2 levels were increased in BMSCs cultured in osteogenic medium (containing 5.5 mM or 25 mM glucose) relative to the control. Additionally, the BMP-2 concentration was lower in the BMSCs cultured under high-glucose osteogenic conditions compared to those ltured in normal glucose osteogenic conditions (Figure 3A(Fig. 3)).
High glucose inhibited ALP activity and expression of RUNX2, ALP, and OCN
To confirm the inhibition effects of high glucose on the osteogenic differentiation of BMSCs, we carried out ALP staining and ALP activity analysis. ALP staining revealed the suppression effect of high glucose on the osteogenic differentiation of BMSCs, and ALP activity analysis revealed that the ALP activity was 1.443, which was higher than that in the undifferentiated high-glucose group, but was decreased by 1.39-fold compared to that in the differentiated normal-glucose group (P < 0.01; Figure 3B(Fig. 3)). Furthermore, we extracted mRNA from the BMSCs cultured in both types of osteogenic medium for seven days, and performed qRT-PCR to determine the expression levels of three osteoblast marker genes: RUNX2, a critical transcription factor for osteoblastic differentiation; ALP, an early osteoblast marker; and OCN, a mature osteoblast marker. In the differentiated high-glucose group, there was a 1.56, 1.48, and 1.55-fold decrease in the expression level of RUNX2, ALP, and OCN, respectively (P < 0.01; Figure 3C(Fig. 3)).
In vitro regulation of osteogenic differentiation by the BMP pathway
BMP-2 is known to positively regulate osteogenic differentiation. To verify whether treatment of BMSCs with BMP-2 can promote osteogenic differentiation and rescue the osteogenic suppression of high glucose, we tested the intracellular BMP-2 levels and RUNX2, ALP, and OCN expression in the BMSCs after treatment with ectogenic BMP-2. The intracellular BMP-2 in both osteogenic groups (5.5 mM and 25 mM glucose) was significantly elevated (Figure 4A(Fig. 4)). The expression of RUNX2, ALP, and OCN was also increased by 1.72, 1.47, and 1.47-fold in BMP-2-treated BMSCs cultured in osteogenic medium containing 5.5 mM glucose. And in BMSCs cultured in osteogenic medium containing 25 mM glucose, the expression of these three osteoblastic genes was increased by 1.70, 1.53, and 1.48-fold, respectively (P < 0.05; Figure 4B(Fig. 4)), which was consistent with increase of BMP-2. Taken together, our results suggest that the BMP pathway plays an important role in regulating the osteogenic differentiation of BMSCs in a high-glucose environment.
Discussion
Diabetes is a common metabolic disorder characterized by hyperglycemia due to impaired insulin secretion, insufficient insulin action, or both (King, 2008[17]). A complex syndrome with more than one cause, diabetes is responsible for numerous complications affecting the whole body. Compared to individuals without diabetes, patients with diabetes are more susceptible to periodontal disease, which is recognized as the sixth most common complication of diabetes (Dakovic and Pavlovic, 2008[6]; Javed et al., 2007[14]). Moreover, moderate to severe alveolar bone loss is more prevalent in diabetes mellitus patients (Al-Emadi et al., 2006[1]). Implant treatment is an attractive alternative to traditional fixed/removable prostheses (Levin et al., 2007[21], 2006[22]). However, individuals with poorly controlled diabetes are more susceptible to developing complications after implant therapy than individuals with well-controlled diabetes (Fiorellini et al., 2000[11]). The treatment of diabetes focuses on the attainment of optimal glycemic control to prevent complications.
The prevailing literature accounts for both the detrimental and beneficial effects of high glucose on MSCs, which is perplexing. Previously, we found that high glucose affected the biological function of rat osteoblasts (Ma et al., 2011[26]). The potential of MSCs in tissue regeneration is gaining increasing attention. Therefore, we focused on the biological function of stem cells in high-glucose conditions. Our initial assays determined that different concentrations of high glucose affected the biological function of BMSCs differently. In good agreement with an earlier observation in MC3T3 cells (Balint et al., 2001[2]) and in telomerase-immortalized MSCs (Li et al., 2007[23]), we found that high glucose (< 25 mM) greatly promoted proliferation. However, if the glucose concentration was too high (35 mM), it would significantly suppress proliferation and induce cell apoptosis, which would be harmful to tissue regeneration. Our findings were the opposite of the data obtained in rats. In rat MSCs, the reduction of glucose stimulated cell proliferation, and high glucose enhanced apoptosis (Stolzing et al., 2006[32]). The discrepancy may be due to differences in the experimental protocols and cell lines used, and further studies are necessary to investigate the difference.
The osteogenic differentiation of mesenchymal stromal/stem cells is precisely regulated by signaling molecules and transcription factors. Other physical factors such as inflammation and biomechanics also influence bone formation (Liu et al., 2011[25]). As a physical factor, the effects of high glucose on osteogenesis have also been extensively studied (Li et al., 2007[23]; Wang et al., 2010[39]). Our mineral test demonstrated that high glucose (16.5, 25, 35 mM) inhibited the osteogenic differentiation of BMSCs dose-dependently. Our data are in accord with that of some previous studies. For example, Hyun Kim et al. (2006[16]) found that the mineralized nodule formation of periodontal ligament (PDL) cells in high glucose was lower than that in normal glucose medium. Gopalakrishnan et al. (2006[12]) found that ALP activity, the number of nodules formed, and the area stained for collagen in BMSCs were greatly reduced in a high-glucose environment.
Hyperglycemia has been thought to be a potential contributor to diabetic osteoporosis (Schwartz, 2003[31]), but the mechanism of bone loss is still unclear. To differentiate into mature osteoblasts, BMSCs undergo a series of differentiation stages, such as proliferation, matrix development, and matrix mineralization. The transition from proliferation to the matrix differentiation stage is characterized by the up-regulation of genes associated with matrix development and maturation, with that of the genes for collagen and ALP occurring the earliest. However, proliferation slows down with the expression of osteoblast-specific markers. There is a reciprocal correlation between BMSC proliferation and differentiation (Pittenger et al., 1999[28]). Our findings were in accordance with these conclusions. In our study, high glucose (16.5 and 25 mM) promoted proliferation but inhibited osteogenic differentiation. High glucose probably provides sufficient nutrients to BMSCs, thus promoting the energy metabolism of BMSCs, but the mechanism still requires exploration.
We focused on the mechanism of high glucose on the osteogenic differentiation of BMSCs. We used 25 mM glucose as our test group to compare with the control group (5.5 mM). A member of the transforming growth factor-b (TGF-b) super family, BMP-2 is a key signaling factor for inducing the differentiation of stem cells into osteoblasts (Yamaguchi et al., 2000[42]). Therefore, we investigated the level of BMP-2 by western blot analysis. The expression of RUNX2, an important osteogenic transcription factor, ALP, and OCN, two key osteogenic markers, was quantified by real-time PCR.
Osteogenic differentiation of BMSCs is precisely regulated by signaling pathways and transcription factors. In particular, the modulation of the molecular differentiation into bone in MSCs by the BMP signaling pathway is well known. The importance of this precise regulation of BMP signaling and receptor binding is reflected in the appearance of developmental disorders and dysfunctions in humans, such as bone and cartilage diseases (Thomas et al., 1997[33], 1996[34]) or cancer, in which specific components of the BMP pathway are defective (Hartung et al., 2006[13]). BMPs are multifunctional growth factors that play very important roles in the BMP signaling pathway, and have been identified based on their ability to initiate ectopic bone formation in adult animals (Wozney et al., 1988[41]). BMPs also play critical roles during embryonic development (Wills et al., 2006[40]). Among them, BMP-2 is one of the most effective inducers of osteogenic differentiation (Cheng et al., 2003[5]). Runx2 (also called Cbfa1), is a transcription factor required for osteoblastogenesis. Ducy et al. (1997[10]) reported that Runx2 binds the OCN promoter and is expressed in osteochondral progenitors as well as in the early stages of osteoblastic differentiation. The forced expression of Runx2 in non-osteoblastic fibroblasts was sufficient to induce the expression of osteoblastic markers such as type I collagen, bone sialoprotein, OCN, and osteopontin. Moreover, Runx2-knockout mice exhibited no intramembranous or endochondral ossification (Komori et al., 1997[19]; Otto et al., 1997[27]). Runx2 thus serves as an essential regulator of bone formation by osteoblasts and is at the intersection of many signaling pathways regulating osteoblast differentiation. While both BMP signaling and Runx2 expression have been implicated in the expression of an osteoblast phenotype, the precise relationship between BMPs, Runx2 expression, and the transcription of osteoblast-related genes is still being studied. It has been reported BMP-2 could regulate the activity of Runx2 and one of the BMP-Smad target genes is RUNX2 (Derynck and Zhang, 2003[8]). In the present study, our data revealed that BMP-2 levels and Runx2 expression were increased in BMSCs cultured in osteogenic medium containing 5.5 mM and 25 mM glucose relative to the control. This implies that the BMP signaling pathway plays an important role in the osteogenic differentiation of mouse BMSCs and that Runx2 was actually activated in this process. Additionally, we found that under high glucose (25 mM) osteogenic conditions, the BMP-2 concentration in the BMSCs was decreased, and the Runx2 expression was decreased simultaneously, which indicated another perspective to the relationship between BMP-2 and Runx2. Recent work from several groups has clarified that ectogenic BMP-2 could be used to promote the osteogenic differentiation of MSCs (Koch et al., 2005[18]; Pountos et al., 2010[30]; Turgeman et al., 2002[36]). Therefore, we hypothesized that treatment with BMP-2 enhances the osteogenic activity of BMSCs under high-glucose conditions. Our data showed that the osteogenic differentiation of BMSCs was improved through supplementation with BMP-2 in a high-glucose environment. It could be inferred that the inhibition effects of high glucose could be reversed by addition ectogenic BMP-2.
In summary, we have demonstrated that glucose in the microenvironment markedly affects gene regulation, proliferation, and differentiation, as well as the apoptosis of stem cells. According to our current analyses, the high-glucose environment actually affected BMP-2 activation in the BMSCs during the osteogenic differentiation process. BMP signaling pathway could enhance the bone regeneration of BMSCs through the activation of Runx2. This work provides a new insight into the effects of high glucose on the osteogenic differentiation of mouse BMSCs. However, the complex mechanism of the interaction between high glucose and osteogenic differentiation of BMSCs still requires further investigation.
Disclosure
The authors indicate no potential conflict of interest.
Acknowledgements
We thank the staff and faculty of the Dental Institute of General Hospital of People Liberation Army of China for their assistance. We also thank Dr. Se Fei and Dr. Chen Yizhi for their unselfish technical assistance. This work was supported by grants from the Nature Science Foundation of China (81271180) and the Innovative Foundation for Doctor (11BCZ04).
References
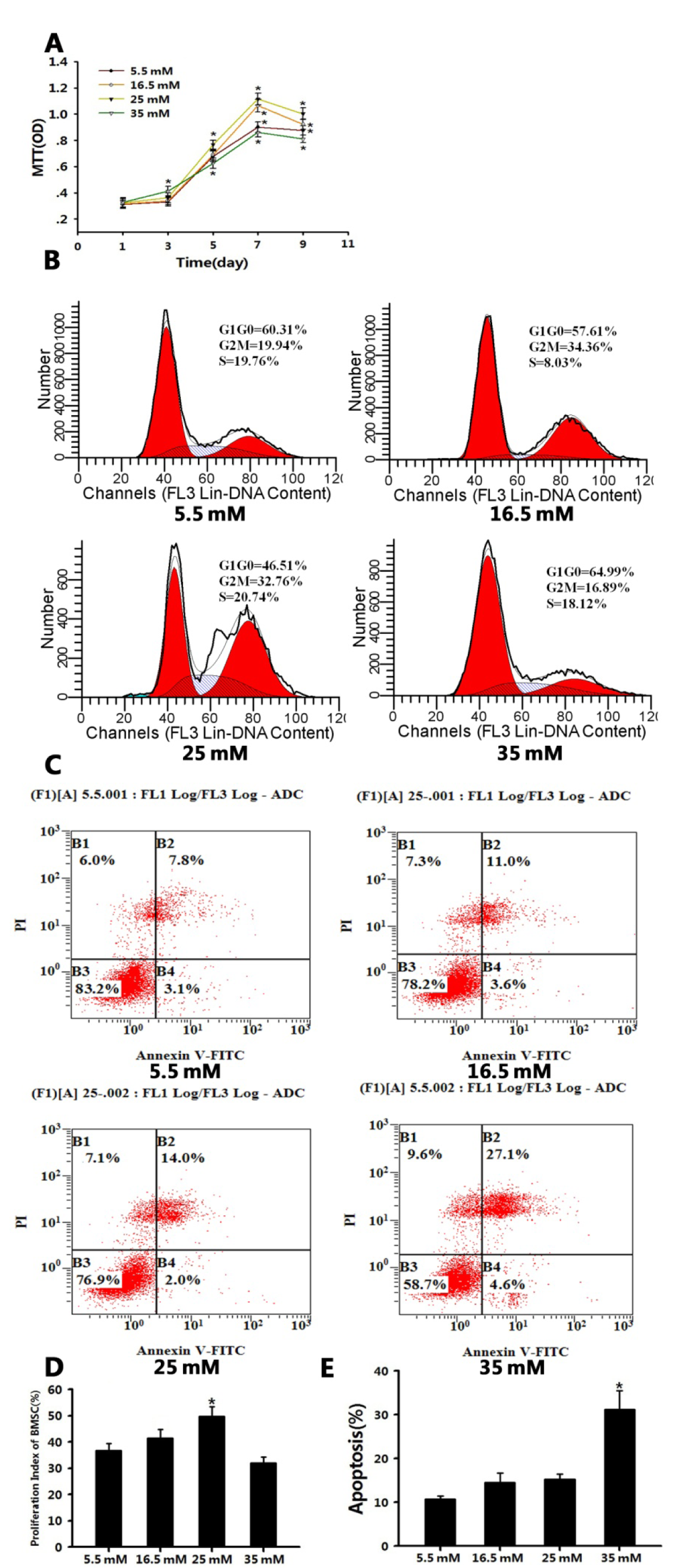
Figure 1: Different concentrations of high glucose had different effects on BMSC proliferation.
A: High-glucose microenvironments (16.5 and 25 mM) promoted BMSC proliferation. Another high-glucose environment (35 mM) inhibited BMSC proliferation. Values are the mean ± SD for triplicate samples from a representative experiment. Student's t-test was performed to determine statistical significance. *P < 0.05
B, D: Flow cytometry analysis of the cell cycle showing that 25 mM glucose had the best effects on BMSC proliferation than the other three glucose concentrations. One-way ANOVA was performed to determine statistical significance. *P < 0.05
C, E: Annexin V-FITC conjugated to PI by flow cytometry showed that 35 mM glucose caused more significant cell apoptosis than the other three glucose concentrations. One-way ANOVA was performed to determine statistical significance. *P < 0.05
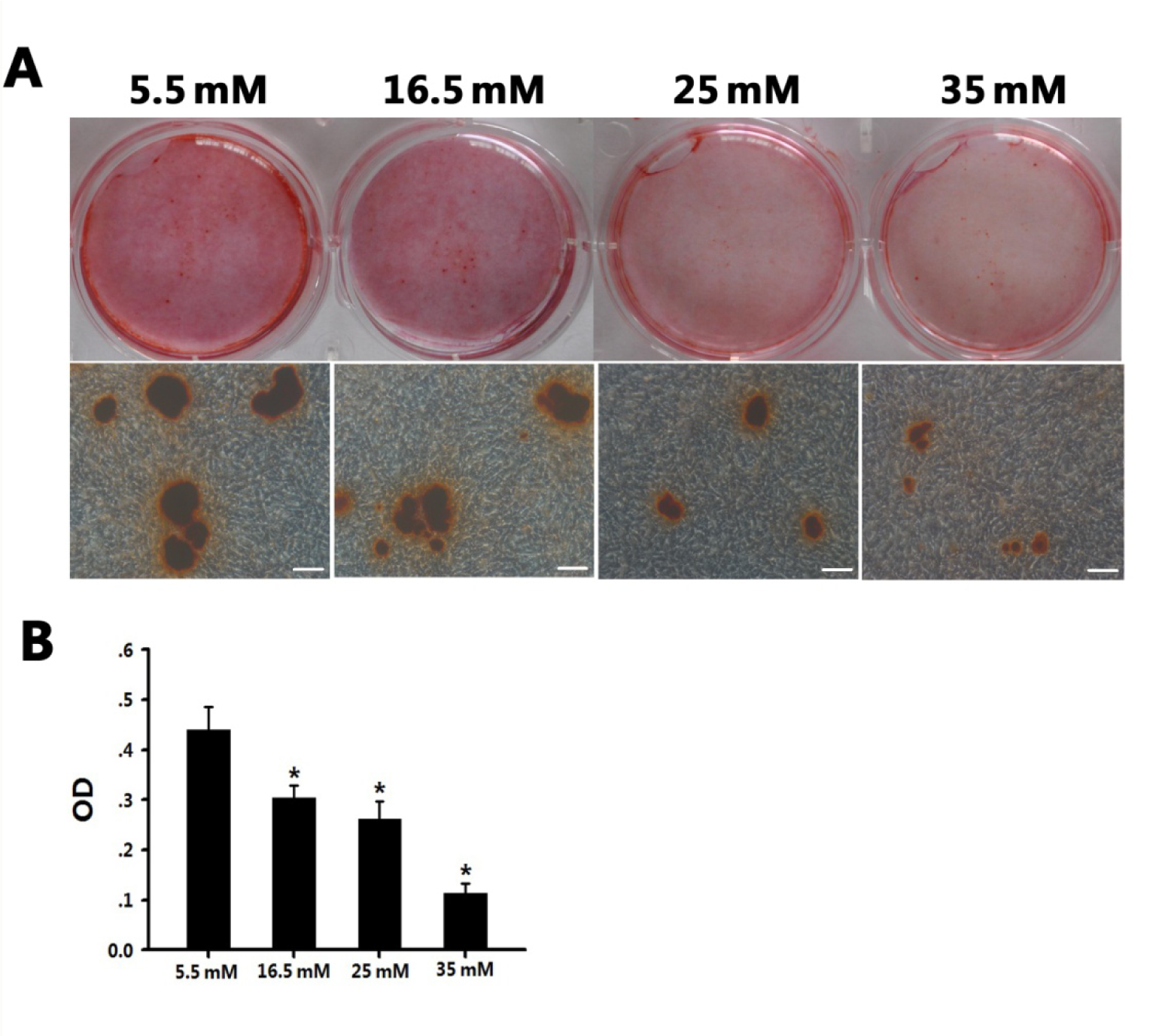
Figure 2: High glucose had an inhibitory effect on BMSC osteogenic differentiation.
A: BMSCs were cultured in four osteogenic media (glucose concentrations: 5.5 mM, 16.5 mM, 25 mM, and 35 mM, respectively) for 21 days; mineralized nodules were detected following Alizarin red S staining. Scale bars represent 100 µm.
B: Graph illustrating the quantitative evaluation of Alizarin red S staining of the four groups. *P < 0.05 vs. the normal/Diff group
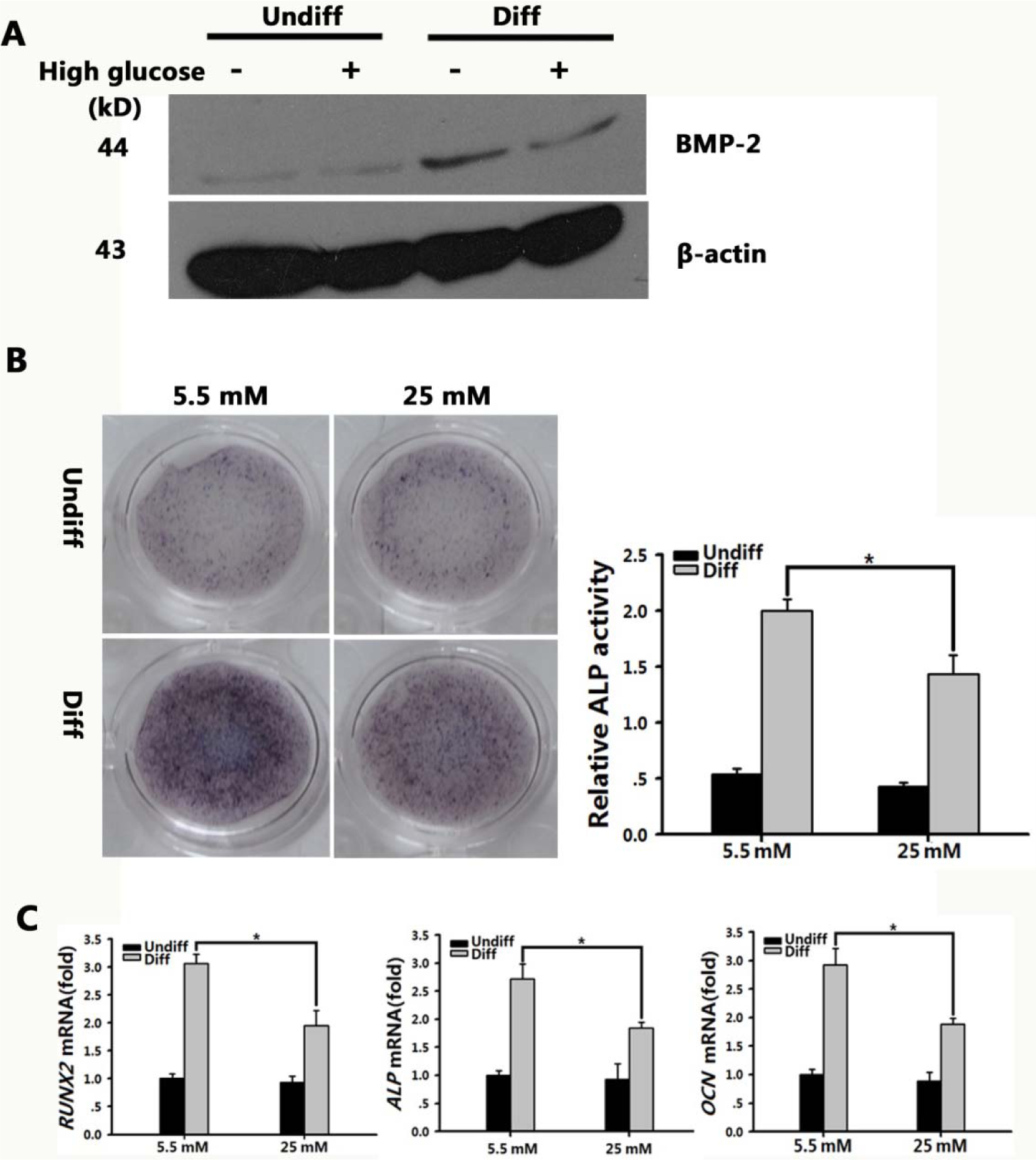
Figure 3: BMP signaling pathway in BMSCs was inhibited in high-glucose microenvironment.
A: BMP-2 levels were tested after BMSCs were grown in normal or high-glucose osteogenic medium for seven days. β-actin was used as an internal control.
B: Scanned images of ALP staining of BMSC after 7-d culture in osteogenic medium containing normal (5.5 mM) or high glucose (25 mM). Graph depicts the quantitative evaluation of ALP activity. The results represent the mean ± SD. *P < 0.05 vs. the normal/Diff group, n = 3
C: Real-time PCR analysis of RUNX2, ALP, and OCN expression in normal and high-glucose conditions grown in basal (Undiff) or osteogenic medium (Diff) for 7 d. The expression levels were normalized to that of β-actin. The results represent the mean ± SD from three independent experiments performed in triplicate. *P < 0.05 vs. the normal/Diff group
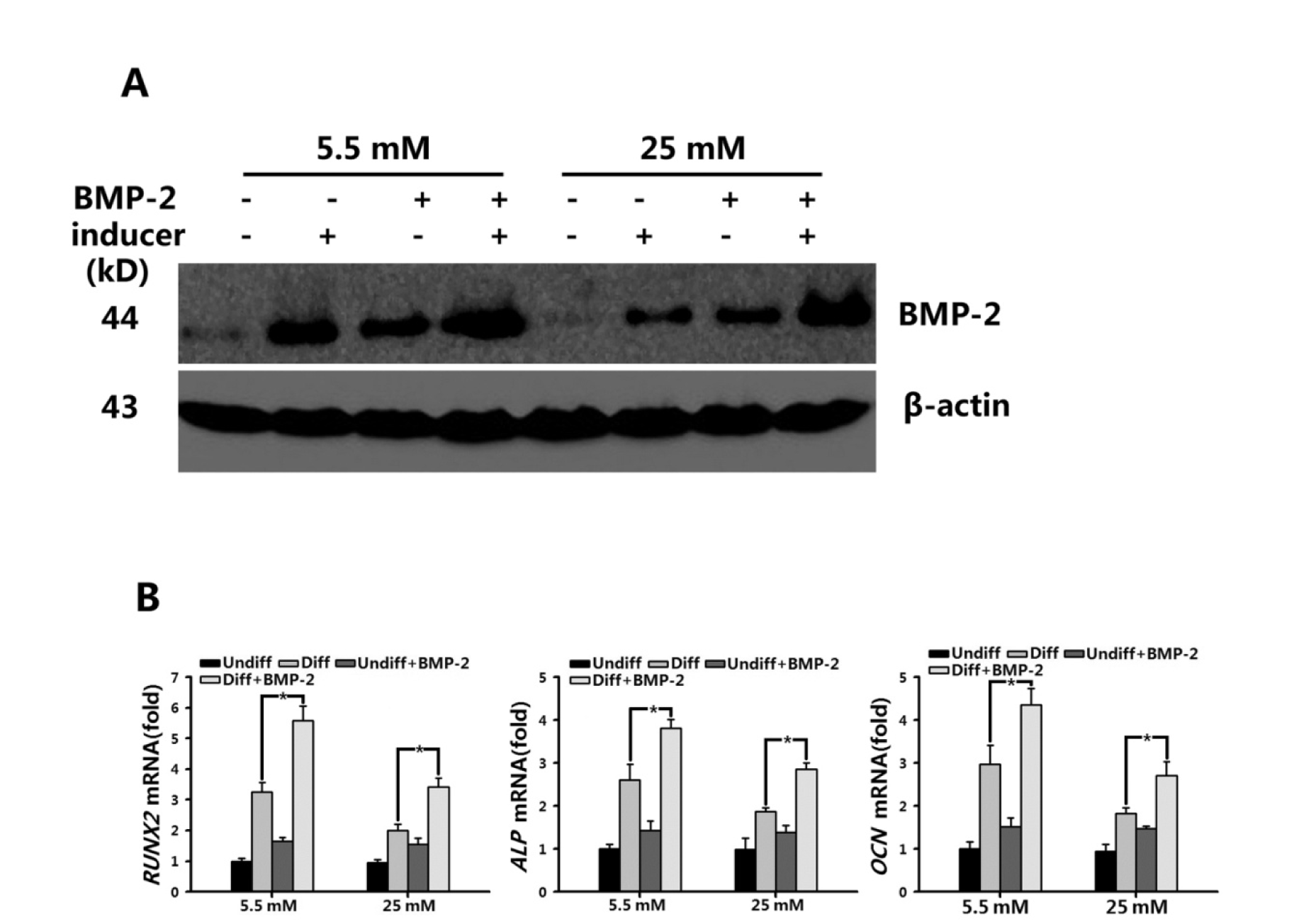
Figure 4: Up-regulated expression levels of RUNX2 by BMP-2 promoted BMSC osteogenic differentiation in normal (5.5 mM) and high-glucose (25 mM) conditions.
A: The level of endogenic BMP-2 in BMSCs grown in basal osteogenic medium containing normal (5.5 mM) or high glucose (25 mM), or in the absence or presence of BMP-2 (100 ng/mL) for 7 d were measured by Western blot.
B: Real-time PCR analysis of RUNX2, ALP, and OCN expression in BMSCs after 7-d culture in basal medium (Undiff) and osteogenic medium (Diff), containing normal (5.5 mM) or high glucose (25 mM) in the absence or presence of 100 ng/mL BMP-2. β-Actin was used as a control for equal loading. Results represent the mean ± SD from three independent experiments performed in triplicate. *P < 0.05 vs. the normal /Diff groups
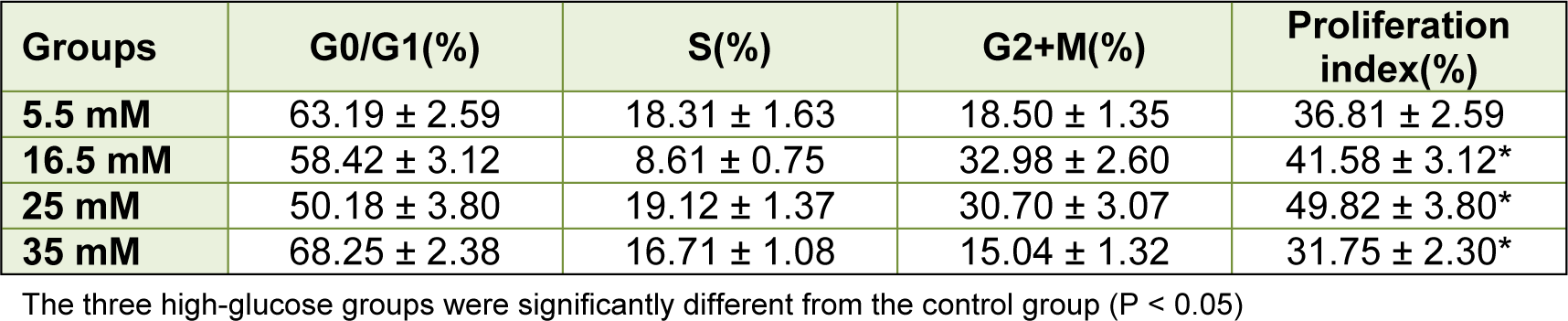
Table 2: Flow cytometric analysis of cell cycles and proliferation indices between four groups (mean ± SE), proliferation index = (S + G2/M), (G0/G1 + S + G2/M) × 100 %
[*] Corresponding Author:
Prof. Hongchen Liu, Stomatology Department of the General Hospital of Chinese PLA, 28 FuXing Road, Beijing 100853, China; Tel.: +86-10-66936254; Fax: +86-10-66936254, eMail: liuhcpla@163.com
