Research article
Total phenolic, antioxidant, antimicrobial activities and toxicity study of Gynotroches axillaris blume (Rhizophoraceae)
Salam Ahmed Abed1, Hasnah Mohd Sirat1[*], Muhammad Taher2
1Department of Chemistry, Faculty of Science, Universiti Teknologi Malaysia, 81310 Skudai, Johor2Department of Pharmaceutical Technology, Faculty of Pharmacy, Universiti Islam Antarabangsa Malaysia, Kuantan, Pahang
EXCLI J 2013;12:Doc404
Abstract
The antioxidant activity and the total phenolic content, as well as the influence of petroleum ether, chloroform and methanol extracts from the leaves of Gynotroches axillaris, on microorganisms were studied. The total phenolic contents were evaluated by using Folin-Ciocalteu reagent and the obtained values ranged from 70.0 to 620 mg GAE/g. The efficiency of antioxidation, which was identified through the scavenging of free radical DPPH, exhibited that the highest IC50 was in the methanolic extract (44.7 µg/mL) as compared to the standard ascorbic acid (25.83 µg/mL) and to standard BHT (17.2 µg/mL). In vitro antimicrobial activity of extracts was tested against Gram-negative bacteria, Gram-positive bacteria and fungi. Methanol extract showed activity in the range (225-900 μg/mL) with both types, while petroleum ether and chloroform extracts were only active with Bacillus subtilis. The three extracts strongly inhibited all fungi with activity 225-450 μg/mL. The toxicity test against brine shrimps indicated that all extracts were non-toxic with LC50 value more than 1000 µg/mL. The finding of this study supports the safety of these extracts to be used in medical treatments.
Keywords: Gynotroches axillaris, Rhizophoraceae, total phenolic, antioxidant, antimicrobial, toxicity
Introduction
The Rhizophoraceae family belongs to the red mangrove, contains about 16 genera and 120 species, which can be found in tropical and subtropical parts of the world obviating the seasonal region (Ng, 1992[14]; Kochummen, 1989[10]). In Malaysia, nine genera with 28 species have been recorded, which are Anisophyllea, Bruguiera, Carallia, Ceriops, Combretocarpus, Gynotroches, Kandelia, Pellacalyx and Rhizophora. It has many usages: its timber is used in buildings, firewood and charcoal. The Brugueira species fruits are eaten as a betel substitute. Leaves of mangroves are important sources of food for marine organism such as shrimps, crabs and fish (Ashton and Macintosh, 2002[2]). On the other hand, some of the species can be used as an alternative medicine. Chemically, the constituents of Rhizophoraceae have been reported to contain hydrocarbons, terpenoids, fatty acids, sterols and alkaloids (Ashton and Macintosh, 2002[2]; Wiart, 2006[23]).
The alternative medicine used different decoctions from Rhizophoraceae in Asia and in other parts of the world. There are a few reports on phytomedicine of this family. For example, fruits and bark of Bruguiera gymnorrhiza are used to treat diarrhea. Besides that, the Chinese treated burns using the roots and bark extracts (Huang et al., 2009[8]). Methanolic extract of Kandelia candel and Rhizophora apiculata are employed as anti-hyperglycemic in India (Tiwari et al., 2008[22]). In Malaysia, Rhizophora apiculata leaves are assayed as anti-proliferative in breast cancer (Nurhanan et al., 2008[16]). Moreover, Carallia brachiate leaves are used in Vietnam to treat malaria and malaria-like symptoms (Nguyen-Pouplin et al., 2007[15]). In fact, Rhizophoraceae species introduced valued pharmacological activity.
Gynotroches genus has been reported to have only one species, i.e. G. axillaris, and in Malaysia it is known as mata keli (eyes' fish). Gynotroches is a middle tree, not more than 36 m in high, smooth bark and grayish color; leaves are decussate and changeable. Its leaves are used to decrease fever and to alleviate headache (Ashton and Macintosh, 2002[2]).
There is a growing demand to get pharmaceutical active components from the natural sources, due to the detrimental side effect of synthesized antimicrobial and antioxidants (Williams et al., 1999[24]). Therefore, the extracts and the isolated pure compounds from the medicinal plants were studied to evaluate their bioactivities. Gynotroches axillaris was proposed for this study to evaluate the bioactivities, antimicrobial, antioxidant and toxicity of extracts from leaves, since there is no report on the bioactivity of this plant.
Materials and Methods
Plant materials
Leaves of Gynotroches axillaris were collected in February 2011 and Voucher specimen has been deposited at Kulliyah of pharmacy, IIUM, Kuantan, Pahang, Malaysia.
Solvent and chemicals
Analytical grade methanol, ethyl acetate, chloroform, petroleum ether (60-80 °C), dimethyl sulfoxide (DMSO), HPLC grade chloroform, sodium carbonate and Folin-Ciocalteu's reagent were bought from Merck (Darmstadt, Germany). 1,1-Diphenyl-2-picrylhydrazyl (DPPH), gallic acid (GA) and butylated hydroxyl toluene (BHT) were ordered from Sigma-Aldrich Chemie (Steinheim, Germany).
Extraction and purification
The dried powdered leaves of the Gynotroches axillaris (300 g) were extracted sequentially with petroleum ether (60-80 °C), using shaker at room temperature for five days. Next, hot extraction in Soxhlet was performed with chloroform and methanol for 20 h. The crude extracts were filtered under vacuum and concentrated using rotatory evaporator at 50 °C to yield petroleum ether extract GP (4.6 %), chloroform extract GCL (1.6 %) and methanol extract GMOH (15 %).
Total phenolic assay
Phenolic content assay was achieved for extracts as described in the literature (Bamoniri et al., 2010[3]) with minor modifications, including gallic acid as standard reference and Folin-Ciocalteau's reagent. All samples were prepared by dissolving each sample (1 mg) in methanol (1 mL) to get concentration (1 mg/mL). A solution of each sample (0.1 mL) was pipetted into a vial containing 0.9 mL of methanol, followed by adding Folin-Ciocalteau reagent (0.05 mL), mixed well and left for two min. After that, 5 % sodium carbonate solution (0.5 mL) was added. Then methanol (2.5 mL) was added to the mixture. Incubation for one hour was allowed in the dark, at room temperature, and absorbance was recorded at 765 nm. The total phenolic content for each sample calibrated with standard curve of gallic acid at concentration 50-500 mg/mL and expressed as mg of gallic acid equivalents per gram of extract.
Antioxidant activity by free radical scavenging assay
Ability to remove free radical was determined by the capability to scavenge DPPH (2,2-diphenyl-1-picrylhydrazyl) (Bamoniri et al., 2010[3]). Stock solution of sample (1000 μg/mL of methanol) was diluted twofold by methanol to get 200 μL of a final range (15.63-1000) μg/mL, in a disposable cuvette, followed by adding 3800 μL of methanolic DPPH (1000 μg/mL) to each cuvette, then the reaction was allowed to stand in the dark for 30 min. UV spectrophotometer was used to measure the absorbance at λmax 517 nm. The absorbance of control (DPPH and methanol) was measured at 0 min. Higher absorbance of reaction solution, means lower free radical scavenging activity, and vice versa. Percentage inhibition (I%) of DPPH radical was calculated with the following formula:
I % = [(Absblank - Abssample )/ Absblank] x 100
Absblank = absorbance of control (DPPH and methanol) without sample
Abssample = absorbance of extract or standard (BHT and ascorbic acid).
By plotting graph of I% against concentration of sample or standard, 50 % percentage inhibition (IC50) value can be calculated based on the formula obtained from the graph. All data were obtained from the triplicates.
Antibacterial assay
The crude extracts (GP, GCL, GMOH) of the leaves of Gynotroches axillaris were screened to determine the antibacterial activity using disc diffusion method as qualitative assay, while minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) as quantitative assay. Gram-negative bacteria, Escherichia coli (ATCC 10536), Pseudomonas aeruginosa (ATCC 9027), and Klebsiella pneumonia (ATCC 13883) and Gram-positive bacteria, (Bacillus subtilis (ATCC 6633), Enterococcus faecalis (ATCC 19433), Staphylococcus aureus (ATCC 29737)), yeasts Candida glebreti (ATCC 2001) and Saccharomyces cerevisiae (ATCC 29737) and fungi, Aspergillus niger (ATCC16888) were selected, and obtained from the American Type Culture Collection (ATCC).
Culture media and material
Culture media to generate bacteria was achieved in sterilized nutrient broth (NB) at 37 °C for 16-18 h while, fungi and yeast were in sterilized Sabouraud dextrose broth (SDB) for 48 h. Nutrient broth (NB, 8 g/L), nutrient agar (NA, 20 g/L), Sabouraud dextrose broth (SDB) and Sabouraud dextrose agar (SDA) were dissolved in distilled water. The glasses (pipettes, tubes, Z-rode and beakers), filter paper discs (6 mm in diameter) and solution (NB, NA, SDB and SDA) were sterilized in autoclave for 2.5 h at 121 °C. The concentration of bacteria, yeast and fungi cultures were prepared by comparing with McFarland solution (9.95 mL of H2SO4 solution 1 % in broth, and 0.05 mL of BaCl2 solution 1 % in broth) equivalent to 150x106 colony-forming unit (CFU)/mL. Crude extracts (1800 μg/mL) were prepared by dissolving 3.6 mg in 0.5 mL DMSO.
Disc diffusion method
Three extracts (GP, GCL, and GMOH) were investigated for antibacterial activity by disc diffusion method according to published report (Magina et al., 2009[12]) with some modifications. First, the Petri dishes (90×15 mm) were spread with sterilized NA (17 mL) and SDA (17 mL) solutions, followed by 200 μL of bacteria stock (150×106 CFU/mL) and fungi (150×106 CFU/mL); each was spreaded on the nutrient agar (NA) and Sabouraud dextrose agar (SDA) medium using Z-glass rod, after that, 2 paper discs were individually impregnated with 20 μL of extract (180 μg/mL), 2 blank discs (with DMSO only), standard disc of streptomycin sulphate (10 μg/disc) for bacteria while nystatine standard for fungi was placed and arranged on NA and SDA Petri dish. Finally all plates were sealed and incubated at 37 °C for 24 h.
Determination of minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC)
Minimum inhibitory concentration (MIC) for samples was achieved in the 96-wells plate. First samples (14.4 mg) were dissolved in DMSO 2.0 mL. The concentration of stock solution 1800 μg/mL was gradually diluted twofold to get concentration for each sample in the range of 14.07-1800 μg/mL, then 96-wells were impregnated with 100 μL of organism and were covered for incubation overnight at 37 °C. All samples were assessed in duplicates. The last clear well is assumed as MIC value. The last clear well (showing no macroscopically visible growth) was used to determine MBC. Fresh sterilized nutrient agar NA (5 mL) and SDA (5 mL) were poured in Petri-dish (50x15 mm), then sample (20 μL) of last clear well was spread over NA and SDA Petri dishes. After that all Petri-dishes were incubated at 37 °C for 16-20 h. The last concentration that does not show any visible growth of microorganism was recorded as MBC value (Salleh et al., 2011[20]).
Toxicity Analysis
Materials and preparation of samples
Brine shrimp eggs (Artemia salina) were purchased from a local aquarium shop. Artificial seawater was prepared by dissolving sea salt (19 g) in distilled water (500 mL). The samples were prepared by dissolving extract 4 mg in 4 mL of a proper solvent as stock solution. Petroleum ether extract and chloroform extract were dissolved in chloroform. Methanol extract was dissolved in methanol. From stock solutions, concentration 10, 100 and 1000 µg/mL were prepared in three vials, and each concentration was done in triplicates. Then, these vials were evaporated overnight at room temperature.
Biological screening
Three extracts were screened with brine shrimps test (Lachumy et al., 2010[11]). Eggs of brine shrimps, Artemia salina were hatched in a tank containing artificial seawater (200 mL). The tank was separated into two parts; dark and lighted sides. Eggs were incubated in the dark at room temperature, for 48 h. The hatched shrimps swam were arranged in 10 larva/vial in triplicate. For each vial, the sea water was added until 5 mL. Also, 10 larvae were added for negative control, which had only sea water. The number of dead shrimps was recorded after 24 h. The data were analyzed using Finney program, to obtain the LC50 (lethal concentration that kills 50 % of brine shrimps) values with confidence intervals (95 %) (Onocha et al., 2011[17]).
Results and Discussion
In this study, sequential extraction was used in the preparation of extracts, to show the effect of polarity of solvents on the antibacterial and antioxidant activities. The highest yield was obtained in methanolic extract, while the lowest percentage was in chloroform. The percentages of different extracts were in the order of methanol (15 %) > petroleum ether (4.6 %) > chloroform (1.6 %).
Antioxidant activity
The bioactivities of extracts of G. axillaris were assessed for their total phenolic content and ability to scavenge DPPH free radical using UV spectrometer. The antioxidant results are listed in Table 1(Tab. 1). Phenolic compounds are the most important products of secondary metabolites, and are suggested to protect plant leaves from being damaged, due to environmental factors such as UV radiation and hot weather, in which they act as antioxidant (Banerjee et al., 2008[5]). In this study, the total phenolic content in the extracts of the leaves was evaluated according to Folin-Ciocalteu's method and expressed as equivalent to gallic acid mg per g of the extract. Among all the extracts, methanol extract (620 mg/g) had the highest content, while petroleum ether extract had the lowest content (70 mg/g). The effect of increasing the polarity of solvent revealed that the total phenolic content in the extracts was enhanced with the increasing polarity of solvent, as shown in Table 1(Tab. 1). The total phenolic yields were in the order of methanol (620 mg/g) > chloroform (145 mg/g) > petroleum ether (70 mg/g). Thus, methanol solvent yielded the highest phenolic content, which may associate with antioxidant activity.
The antioxidant property has an important role in human health and conserving food. It acts by different mechanisms, control and scavenging of free-radical, inhibits chain propagation and repairing mechanisms, in addition to the chelating of transition metal (Murkovic, 2003[13]; Kathiresan and Bingham, 2001[9]). The antioxidants were stated to decrease oxidation of low density lipoproteins, which is the cause of arteriosclerosis and then heart attack. The antioxidant activity of extracts was assessed by reacting with stable free radical DPPH, which has a purple colour. Whenever, the DPPH was exposed to electron donator, the colour changed into yellow, so that the extract is considered as an antioxidant and also radical hunter. The results of scavenging DPPH free radical are listed in Table 1(Tab. 1).
Table 1(Tab. 1) exhibited that the IC50 of methanol extract (44.7 µg/mL) was the highest antioxidant capacity among the extracts as compared to the standard ascorbic acid (25.83 µg/mL) and standard BHT (17.2 µg/mL). The weakest capacity of the antioxidant was recorded in petroleum ether, followed by the chloroform extracts.
In this study, the total phenolic contents (TPC) and the percentage inhibition (I%) of different extracts were correlated by SPSS ver. 16, as shown in Figure 1(Fig. 1). The positive correlation (R2=0.999) was observed, and the significant (p < 0.05) was in one tailed. In another words, most of the total phenolics in these different extracts of the leaves from G. axillaris are phenolic compounds such as flavonoids and tannins. Other studies reported that Rhizophoraceae species are rich in tannins (Banerjee et al., 2008[5]; Bandaranayake, 1998[4]). Besides that, the HPLC investigation of Rhizophora apiculata (Rhizophoraceae) showed, that catechin monomer was the antioxidant flavonoid (Rahim et al., 2008[18]). These studies conformed to our suggestions, so more search is needed in the phytochemistry of G. axillaris to investigate the nature of the phenolic compounds.
Antimicrobial activity
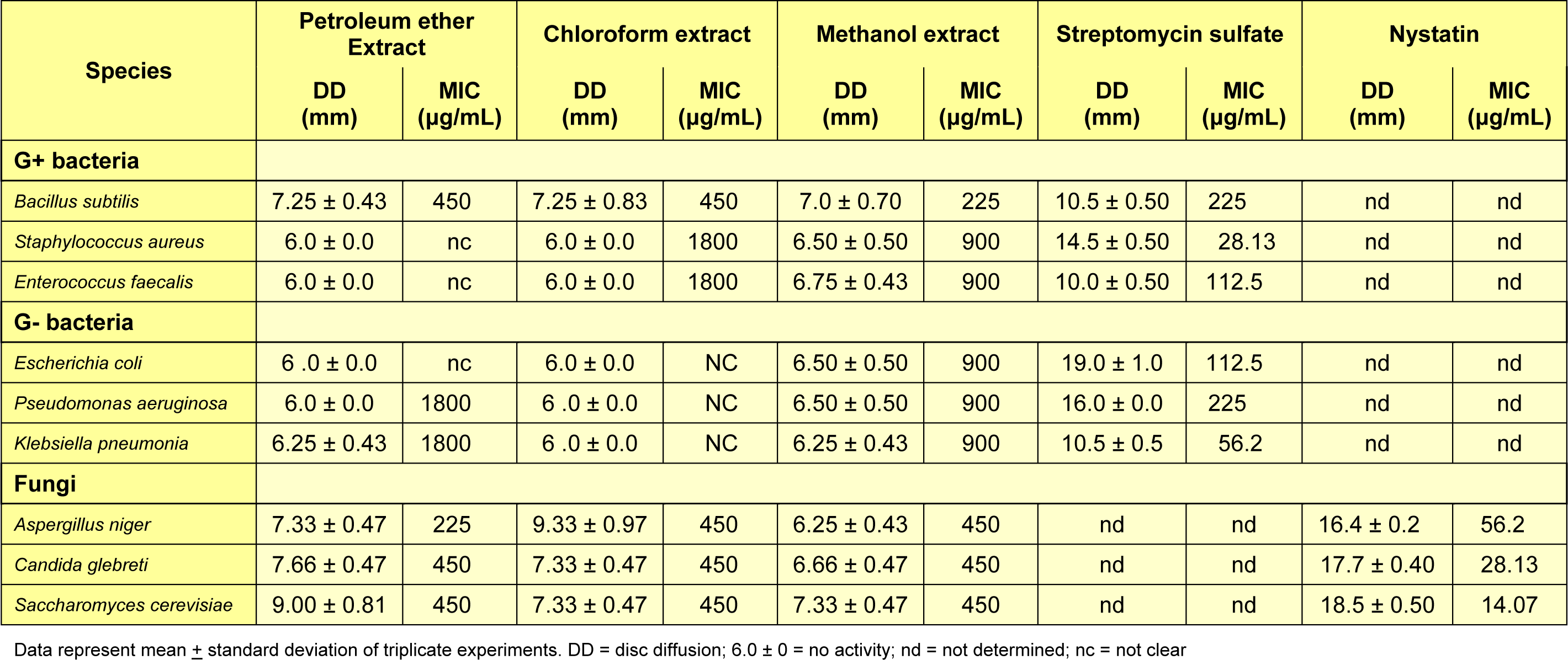
Activity of petroleum ether, chloroform and methanol extracts of the leaves of G. axillaris were tested against selected Gram-negative bacteria (Escherichia coli, Pseudomonas aeruginosa, and Klebsiella pneumonia), Gram-positive bacteria (Bacillus subtilis, Enterococcus faecalis, and Staphylococcus aureus), yeast (Candida glebreti, Saccharomyces cerevisiae) and fungi (Aspergillus niger), obtained from the American Type Culture Collection (ATCC). The antibacterial test was carried out using disc diffusion method as qualitative assay, while minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) as quantitative assay.
The in vitro antibacterial activity was based on the MIC results (Table 2(Tab. 2)).
- Strong inhibitors, if MIC is less than 500 μg/mL;
- Moderate inhibitors, if MIC is in between 600 and 1500 μg/mL;
- Weak inhibitors, if MIC is more than 1600 μg/mL (Magina et al., 2009[12]).
The results showed that petroleum ether and chloroform extracts were strong inhibitors towards B. subtilis and inactive with the other bacteria. Methanol extract was the strongest as inhibitor (225 μg/mL) towards B. subtilis, but moderate (900 μg/mL) towards the other bacteria. In the same activity, yeasts (C. glebreti. and S. cerevisiae) and fungi (A. niger.) were strongly inhibited by all three extracts.
Cell wall of the Gram-negative bacteria is thicker and more complicated than the Gram-positive bacteria. It has an outer membrane cell consisting of a high content of lipid-polysaccharide sheet, which may defend against the passage of hydrophobic groups (Sulaiman et al., 2011[21]; Ratledge and Wilkinson, 1988[19]). Petroleum ether and chloroform extracts are very rich with hydrophobic compounds (Abed and Sirat, 2011[1]); therefore, they were inefficient to inhibit Gram-negative bacteria while, methanol extract was active because of the higher content of hydrophilic compound such as, phenolic compound. In fact, there was no previous study concerning the antimicrobial activity of the leaf extracts of Gynotroches axillaris. Similar studies were established about another plant from Rhizophoraceae, and exhibited methanol and ethanol extracts of leaves and bark from Bruguiera gymnorrhiza had antibacterial activities with both Gram-positive (B. cereus and S. aureus) and Gram-negative (E. coli and P. aeruginosa). Whereas, the chloroform extract did not show any inhibition with Gram-negative (Haq et al., 2011[6]). It completely agrees with our findings.
Toxicity against brine shrimps
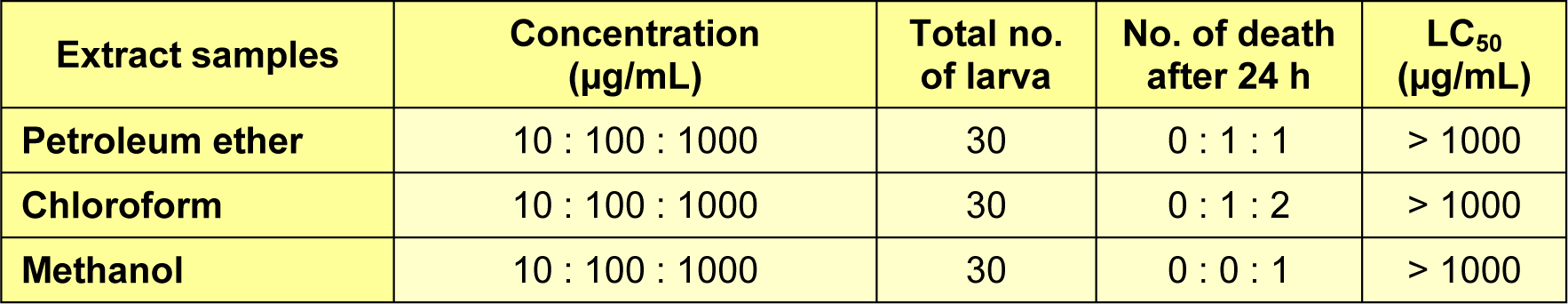
Artemia salina nauplii and mammalian cells have similar sensitivity to the toxicity in many cases (Hong et al., 2011[7]). Toxicity to brine shrimps is used to evaluate the validity of extracts in Medication Safety, and the value of LC50 which is more than 1000 µg/mL is considered non-toxic (Lachumy et al., 2010[11]). The results of toxicity test against brine shrimps indicated that the leaves extracts were non-toxic. As shown in Table 3(Tab. 3), the petroleum ether, chloroform and methanol extracts showed LC50 value more than 1000 µg/mL, which are not toxic. The finding of the toxicity test supports the safety of these extracts to be used in medical treatments.
Acknowledgements
The authors thank Research University Grant (GUP) from the Ministry of Higher Education (MOHE) for financial support under vote QJ130000.7126.01H01 and the Faculty of Science, Universiti Teknologi Malaysia for the NMR and bioactivity facilities. The authors would also like to thank Kerbala University in Iraq for partial financial support.
References
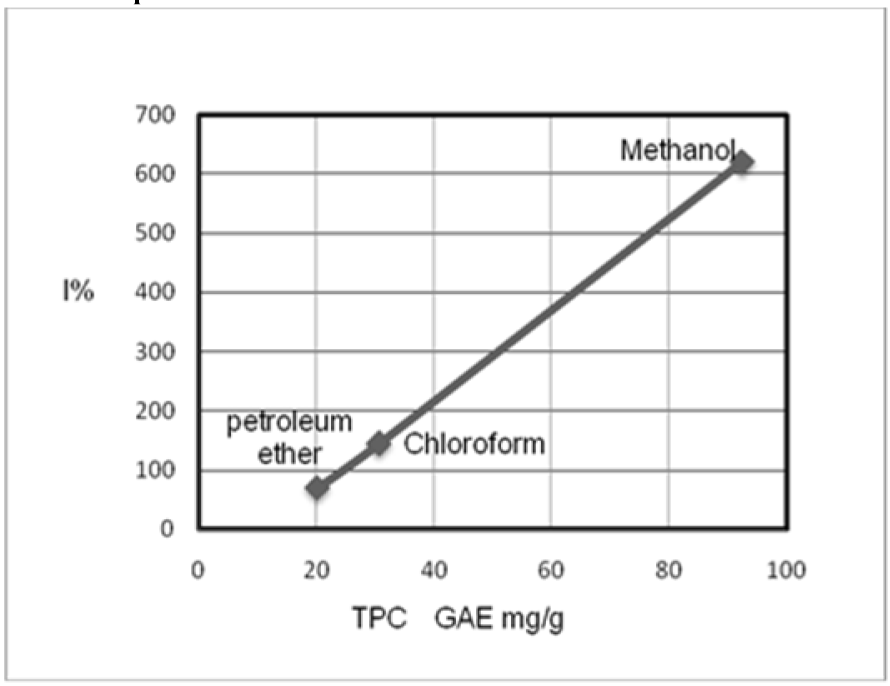
Figure 1: Linear correlation between the total phenolic contents (TPC) and percentage inhibition (I%) of the leaf extracts of G. axillaris (Rhizophoraceae)
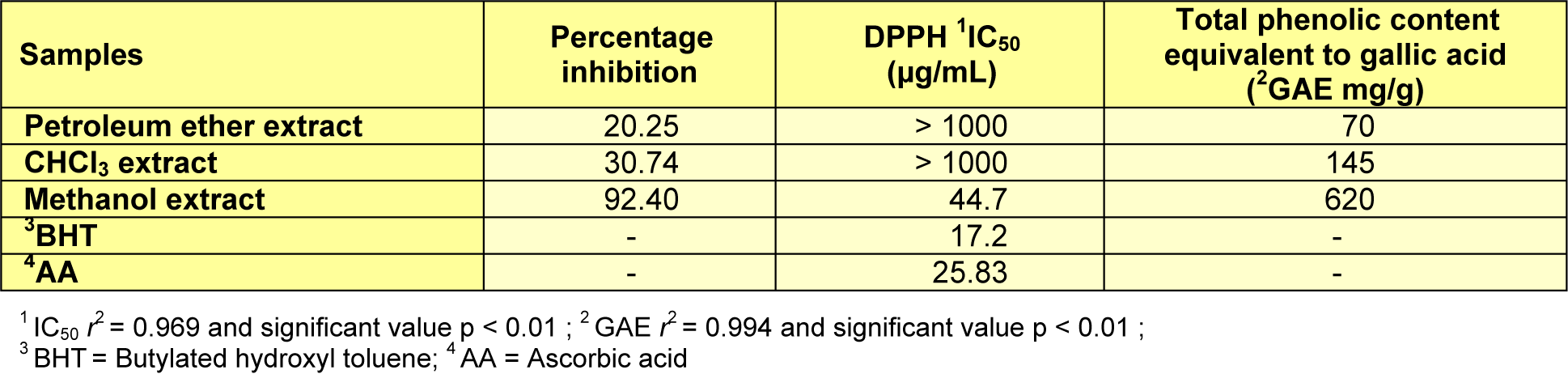
Table 1: Antioxidant activity and total phenolic content of the extracts from leaves of Gynotroches axillaris
[*] Corresponding Author:
Hasnah Mohd Sirat, Department of Chemistry, Faculty of Science, Universiti Teknologi Malaysia, 81310 Skudai, Johor, eMail: hasnah@kimia.fs.utm.my