Research article
Hematological alterations in diabetic rats - Role of adipocytokines and effect of citrus flavonoids
Ayman M. Mahmoud1[*]
1Physiology Division, Zoology Department, Faculty of Science, Beni-Suef University, Beni-Suef, EgyptEXCLI J 2013;12:Doc647
Abstract
Dysfunction in adipocytes is associated with insulin resistance and type 2 diabetes. Diabetes-associated anemia has been reported due to the increased non-enzymatic glycosylation of RBC membrane proteins, which correlates with hyperglycemia. The present study was hypothesized to assess the effect of citrus flavonoids on hematological parameters and adipose tissue interleukin-6 and adiponectin in type 2 diabetic rats. Diabetes was induced by feeding rats with a high fat diet for 2 weeks followed by an intraperitoneal injection of streptozotocin. An oral dose of 50 mg/kg hesperidin or naringin was daily given for 4 weeks after diabetes induction. By the end of the experiment, blood samples were collected and were immediately used for determination of haematological parameters. Expression levels of adiponectin and interleukin-6 were assayed in adipose tissue samples. Both hesperidin and naringin significantly improved the levels of erythrocytes, leukocytes and their functional indices. Similarly, supplementation with either compound significantly ameliorated the declined adipose tissue adiponectin expression in conjunction with down-regulated interleukin-6. The findings of the present study indicated that hesperidin and naringin exert protection to diabetes-associated anemia in type 2 diabetic rats. This could be due to attenuation of proinflammatory cytokine production and stimulation of adiponectin expression.
Keywords: adiponectin, IL-6, hematological parameters, type 2 diabetes
Introduction
Diabetes mellitus (DM) is a complex metabolic disorder involving many organs and can devastate the lives of affected individuals (Khuwaja et al., 2010[20]). It is characterized with chronic high blood glucose that could lead to morbidity and mortality (Mohammed et al., 2007[36]). The number of people suffering from diabetes worldwide is increasing at an alarming rate. It is predicted that about 366 million people are likely to be diabetic by the year 2030 (Wild et al., 2004[57]).
The adipokines are signaling proteins involved in the regulation of energy and glucose metabolism (Wild et al., 2004[57]). Adipocytes secrete diverse pro-inflammatory cytokines such as interleukin (IL)-6 and tumor necrosis factor (TNF)-α, as well as anti-inflammatory cytokines such as adiponectin (Kershaw and Flier, 2004[19]). Dysfunction in adipocytes or adipose tissue is associated with insulin resistance and type 2 diabetes (Sowers, 2008[50]). A reduced level of adiponectin and increased levels of IL-6 and TNF-α can induce or exacerbate insulin resistance in adipose tissue (Blüher, 2009[7]).
Flavonoids are non-nutritive dietary components that are widely distributed in plants (Mahmoud, 2012[32]). Naringin (4´,5,7- trihydroxy flavonone 7-rhamnoglucoside) is the predominant flavonone found in grape fruit and related citrus species (Jagetia and Reddy, 2002[16]). Among the naturally occurring flavonoids, naringin has been empirically proven to have no side effects, as humans have been ingesting grapes and citrus fruits for a long time (Choe et al., 2001[9]). Hesperidin is an abundant and inexpensive byproduct of Citrus cultivation and isolated from the ordinary orange Citrus aurantium and other species of the genus Citrus (family: Rutaceae) (Kakadiya et al., 2010[17]). Recently, we have reported the anti-diabetic potentials of both hesperidin and naringin in high fat diet/streptozotocin induced diabetic rats (Ahmed et al., 2012[2]; Mahmoud et al., 2012[34], 2013[33]).
It has been reported that, ingestion of medicinal plants or drugs can alter the normal hematological values (Ajagbonna et al., 1999[3]). Therefore, hematological parameters could be an important tool in the assessment of deleterious effect of drugs, as well as medicinal plants (Yakubu et al., 2007[59]). Mansi and Lahham (2008[35]) revealed that various hematological parameters and the immune system were reported to be altered during the course of diabetes. Thus, the intention of the present study was to scrutinize the influence of oral administration of hesperidin and naringin on the levels of hematological parameters and adipose tissue IL-6 and adiponectin in type 2 diabetic rats
Materials and Methods
Chemicals
Hesperidin, naringin and streptozotocin, were purchased from Sigma Chemicals Co., St. Louis, MO, USA, stored at 2-4 °C and protected from sunlight. All other chemicals were of analytical grade and were obtained from standard commercial supplies.
Experimental animals
White male albino rats (Rattus norvegicus) weighing about 130-150 g were used. They were obtained from the animal house of the National Research Center, El- Giza, Egypt. They were kept under observation for about 15 days before the onset of the experiment to exclude any intercurrent infection. The chosen animals were housed in plastic well aerated cages at normal atmospheric temperature (25 ± 5 °C) and normal 12 hour light/dark cycle. Moreover, they had free access to water and were supplied daily with standard diet of known composition ad libitum. All animal procedures were in accordance with the recommendations of the Canadian Committee for Care and Use of Animals (Canadian Council on Animal Care, 1993[8]).
Induction of diabetes mellitus
Type 2 DM was experimentally induced by feeding a high fat diet (HFD) for an initial period of 2 weeks followed by an intraperitoneal injection of 35 mg/kg b.wt streptozotocin dissolved in citrate buffer pH 4.5 (Srinivasan et al., 2005[51]). Seven days after the injection, rats were screened for serum glucose levels. Rats having serum glucose ≥ 200 mg/dl, after 2 hours of glucose intake, were considered diabetic and selected for further pharmacological studies. The rats were allowed to continue to feed on their respective diets until the end of the study.
Experimental design
The experimental animals were divided into four groups, each group comprising six rats as detailed follows.
- Group 1: Normal control rats (Fed normal fat diet)
- Group 2: Diabetic control rats
- Group 3: Diabetic rats administered with hesperidin (50 mg/kg b.wt.) orally for 4 weeks
- Group 4: Diabetic rats administered with naringin (50 mg/kg b.wt.) orally for 4 weeks.
The dosage was adjusted every week according to any change in body weight to maintain similar dose per kg body weight of rat over the entire period of study for each group. By the end of the experiment, animals were sacrificed and blood samples and adipose tissue were obtained.
Haematological estimations
The blood samples were collected into tubes containing EDTA and were immediately used for determination of haematological parameters. Total red blood cell (RBC) and white blood cell (WBC) counts were estimated according to the visual method of Dacie and Lewis (1991[10]). The percentage packed cell volume (PCV) was determined according to the hematocrit method (Alexander and Grifiths, 1993[4]) while the blood haemoglobin (Hb) concentration in all samples was estimated according to the cyanomethaemoglobin method using Drabkin's reagent (Alexander and Grifiths, 1993[4]). Mean cell volume (MCV), mean corpuscular hemoglobin (MCH) and mean corpuscular hemoglobin concentration (MCHC) were calculated as outlined in Dacie and Lewis (1991[10]). Differential white blood cell counts were estimated using the method of Osim et al. (2004[43]).
RNA isolation and reverse transcriptase ploymerase chain reaction (RT-PCR)
Total RNA was isolated from visceral adipose tissue using total RNA isolation kit (Fermentas, USA). First strand of cDNA was synthesized from 5 µg of total RNA by using a high-capacity cDNA reverse transcription kit with RNase inhibitor (Applied Biosystems, USA). The produced cDNA was amplified using Green master mix (Fermentas, USA) using the following sets of primers:
Adiponectin
Up 5′-AATCCTGCCCAGTCATGAAG-3′
Down 5′-TCTCCAGGAGTGCCATCTCT-3′
IL-6
Up
5′-CTTCCAGCCAGTTGCCTTCT-3′
Down
5′-GACAGCATTGGAAGTTGGGG-3′
β-actin
Up
5′-AAGTCCCTCACCCTCCCAAAAG-3′
Down
5′-AAGCAATGCTGTCACCTTCCC-3′
The reaction tubes were placed on a double heated led thermal cycler and the reaction series was performed as follows: initial denaturation at 95 °C for 2 min, then 35 cycles each was 95 °C (30 s), 59.5 °C (30 s), 72 °C (45 s).
PCR products were loaded in 1.5 % agarose gel (Sigma) and electrical power was applied then the samples were left to migrate for suitable time. After migration, the cDNA bands were observed in the gel using UV transilluminator. Gel images were analyzed by scanning densitometry (ImageJ, NIH) and values were normalized to the quantity of β-actin, and presented as % mRNA relative to control.
Statistical analyses
Statistical analysis was performed using SPSS v.16. Results were articulated as mean ± SE and all statistical comparisons were made by means of one-way ANOVA test followed by Duncan's multiple range test post hoc analysis. A P value < 0.05 was considered significant.
Results
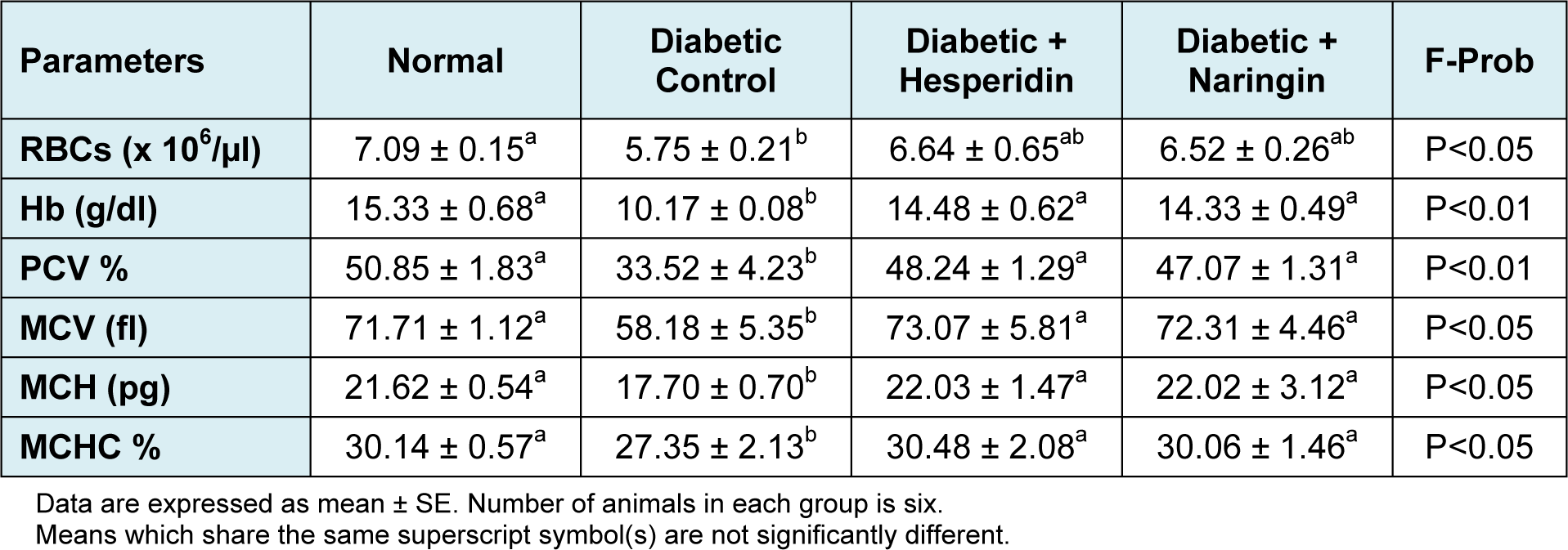
RBC count of diabetic control rats (5.75 ± 0.21 x 106/µl) showed a significant decrease (P < 0.05) as compared to normal control rats (7.09 ± 0.15 x 106/µl). Both treatment agents showed a detectable amelioration of RBC count of diabetic rats; the recorded values were 6.64 ± 0.65 and 6.52 ± 0.26 x 106/µl for hesperidin and naringin respectively (Table 1(Tab. 1)).
The recorded values of diabetic rats showed a notable (P < 0.01) decrease in blood hemoglobin content (10.17 ± 0.08 g/dl) as compared with the normal control rats (15.33 ± 0.68 g/dl). Treatment with hesperidin and naringin produced a highly significant increase of the lowered blood hemoglobin content of diabetic rats; the values being 14.48 ± 0.62 and 14.33 ± 0.49 g/dl, respectively (Table 1(Tab. 1)).
The significant decrease in the levels of PCV, MCH, MCV and MCHC observed in the diabetic animals was drastically increased to near normal level following hesperidin and naringin administration.
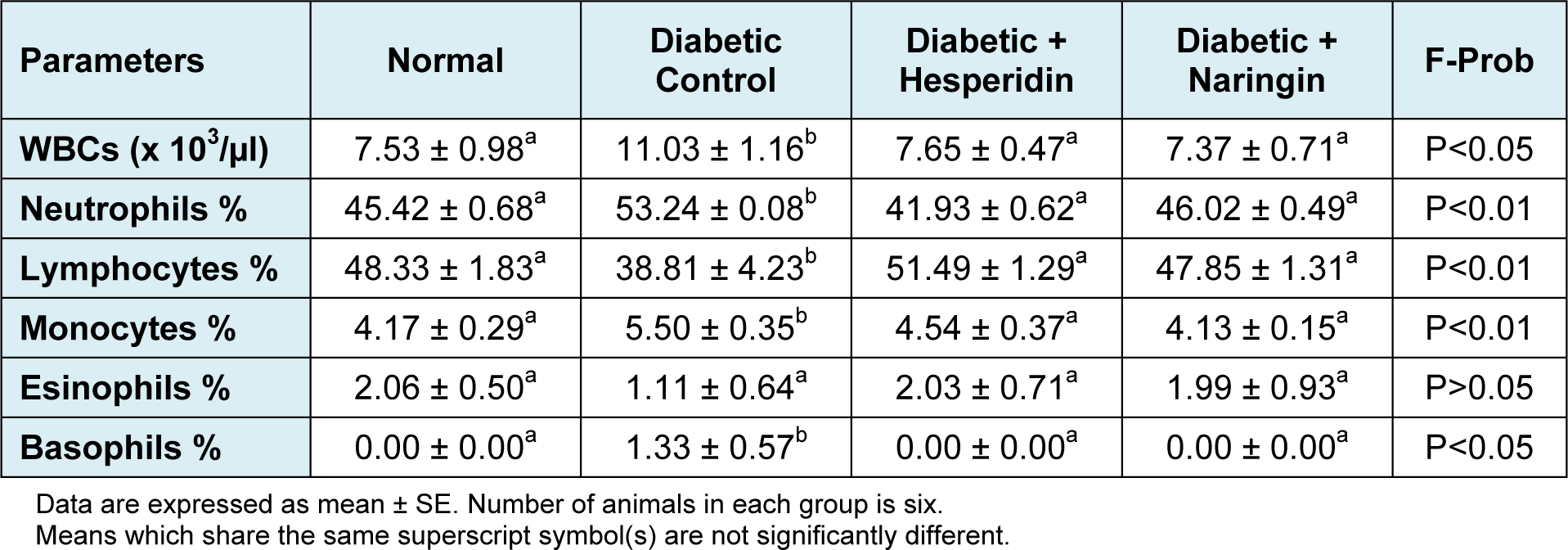
Concerning white blood cell count the present data showed a significant increase (P < 0.05) in diabetic group when compared with control group (Table 2(Tab. 2)). Diabetic group showed non-significant change (P > 0.05) in the percentage of eosinophils, however, there was a significant increase (P < 0.01) in neutrophils, monocytes and basophils. On the other hand, lymphocytes showed a significant decrease (P < 0.01) when compared with normal control group throughout the experimental period. Both hesperidin and naringin supplementation significantly reversed these parameters.
Densitometric analysis revealed a significant (P < 0.01) increase of adipose tissue IL-6 mRNA expression in HFD/STZ diabetic rats as compared with the normal control group. The treatment of the diabetic animals with either hesperidin or naringin significantly (P < 0.01) lowered the elevated IL-6 mRNA expression (Figure 1(Fig. 1)). Hesperidin seemed to be more potent than naringin in decreasing the adipose IL-6 expression.
Adipose tissue adiponectin expression exhibited a different behavioral pattern where it was significantly (P < 0.01) depressed as a result of STZ administration following the HFD. Both hesperidin and naringin supplementations produced a highly significant (P < 0.01) amelioration of the depressed adipose tissue adiponectin mRNA content (Figure 2(Fig. 2)).
Discussion
The link between chronic diseases and anemia is well characterized (Weiss and Goodnough, 2005[55]). The occurrence of anaemia in diabetes mellitus has been reported due to the increased non-enzymatic glycosylation of RBC membrane proteins, which correlates with hyperglycemia (Oyedemi et al., 2011[44]). Oxidation of these proteins and hyperglycaemia in diabetes mellitus causes an increase in the production of lipid peroxides that lead to haemolysis of RBC (Arun and Ramesh, 2002[5]). The major pathological consequences of free radical induced membrane lipid peroxidation include increased membrane rigidity, decreased cellular deformability, reduced erythrocyte survival, and lipid fluidity (Kolanjiappan et al., 2002[22]). In this study, the RBC membrane lipid peroxide levels in diabetic rats were not measured. However, RBC parameters such as Hb, MCHC, MCH, PCV and MCV were studied to investigate the effect of hesperidin and naringin on the anemic status. The decrease in MCH and MCHC values, observed after administration of STZ, is an indication of abnormal hemoglobin synthesis, failure of blood osmoregulation, and plasma osmolarity (Stookey et al., 2007[52]).
Following hesperidin and naringin administration, the level of RBCs and its related indices were appreciably improved. This gives an indication that both flavonoids can stimulate the formation or secretion of erythropoietin, which stimulates stem cells in the bone marrow to produce red blood cells (Ohlsson and Aher, 2012[41]). The stimulation of this hormone enhances rapid synthesis of RBC which is supported by the improved level of MCH and MCHC (Abu-Zaiton, 2010[1]). These parameters are used mathematically to define the concentration of haemoglobin and to suggest the restoration of oxygen carrying capacity of the blood. We have previously reported that the action mechanism of both flavonoids may be attributed to their ability to lower lipid peroxidation level (Mahmoud et al., 2012[34]) that causes haemolysis of erythrocytes.
The expression of hepcidin, a 25 amino acid peptide hormone, in the liver is increased dramatically by inflammation and because of chronic disease (Nicolas et al., 2002[40]). Proinflammatory cytokines, such as IL-6 and IL-1 (Nemeth et al., 2004[38]; Lee et al., 2004[30]), are thought to be central to this mechanism. IL-6 in particular has been shown to exert its effects on hepcidin gene transcription via Janus kinase (JAK)/signal transducer and activator of transcription (STAT)3 interactions (Wrighting and Andrews, 2006[58]; Verga et al., 2007[54]), which are essential to cytokine receptor signaling (Murray, 2007[37]). Once released, hepcidin is thought to bind to the iron efflux protein ferroportin (Nemeth et al., 2004[39]) and thereby act as a negative regulator of body iron homeostasis, inhibiting the release of iron recycled from senescent red blood cells by reticuloendothelial macrophages (Knutson et al., 2005[21]) and the absorption of dietary iron by intestinal enterocytes (Laftah et al., 2004[27]). Moreover, it has been reported that in obese subjects the presence of chronic low-grade inflammation, that resulted from the enhanced production and release of a cocktail of proinflammatory cytokines and adipokines from the adipose tissue (Lago et al., 2007[28]) lead to an increase in the expression of hepcidin (Knutson et al., 2005[21]). Thus, by attenuating pro-inflammatory cytokines production (Mahmoud et al., 2012[34]), hesperidin and naringin may participate directly to the amelioration or RBC indices and correction of the anemic status in type 2 diabetic rats.
Peripheral WBC count has been shown to be associated with insulin resistance, type 2 diabetes (Ohshita et al., 2004[42]), coronary artery disease (Lee et al., 2001[29]), stroke (Lee et al., 2001[29]), and diabetes micro- and macrovascular complications (Tong et al., 2004[53]). Peripheral blood leukocytes are composed of polymorphonuclear cells, including monocytes as well as lymphocytes. Polymorpho- and mononuclear leukocytes can be activated by advanced glycation end products (Pertynska-Marczewska et al., 2004[46]), oxidative stress (Shurtz-Swirski et al., 2004[49]), angiotensin II (Lee et al., 2004[31]), and cytokines (Scherberich, 2003[47]) in a state of hyperglycemia.
Leukocytes may be activated through the release of cytokines, such as TNF-α (Shanmugam et al., 2003[48]), transforming growth factor-1 (Korpinen et al., 2001[23]), superoxide (Kedziora-Kornatowska, 1999[18]), nuclear factor κB (NF-κB) (Hofmann et al., 1998[14]), monocyte chemoattractant protein 1, interleukin-1β, and others (Shanmugam et al., 2003[48]) to participate in the pathogenesis of diabetic micro- and macrovascular complications. The profile of the WBC count reflects the balance between the rate of granulocyte production and that of WBC. Kozlov et al. (1995[24]) reported that diabetes in mice was accompanied by moderate neutrophilic leukocytosis and prolonged circulation times of neutrophils and monocytes, and a shortened circulation time of lymphocytes, which increases the susceptibility to infection. The raised leukocyte count may also reflect low-grade inflammation.
The mechanism responsible for leukocytosis in obesity, diabetes, or atherosclerosis is largely unknown. Recent evidence suggests that leptin and the leptin receptor are parts of a pathway that stimulates hemopoiesis (Peelman et al., 2004[45]). Leptin receptors exhibit structural similarity to class I cytokine receptors (Frühbeck, 2006[13]). Interestingly, this class of receptors also includes the gp130 subunit of the IL-6 receptor family, suggesting that IL-6 and leptin may operate via a similar mode of action (Baumann et al., 1996[6]). This may explains the raised leukocyte count in conjunction with reported elevation in serum IL-6 and adipose tissue IL-6 mRNA expression. The decrease in the WBC count, in diabetic treated rats shows the anti-inflammatory property of both hesperidin and naringin.
The commonly used marker for total body iron stores, serum ferritin, is responsive to inflammatory stress (De Domenico et al., 2008[11]), so increased ferritin in diabetes could simply reflect the inflammatory component of that disease (Hotamisligil, 2006[15]). Recent studies have found a negative correlation between serum ferritin and the insulin-sensitizing adipokine, adiponectin (Fargnoli et al., 2008[12]; Ku et al., 2009[25]). The hypothesis that adiponectin links iron and insulin resistance is appealing, as decreased adiponectin levels are associated with obesity and type 2 diabetes (Weyer et al., 2001[56]) and are causally linked with insulin resistance (Kubota et al., 2002[26]). Our results showed a decreased expression of adipose tissue adiponectin mRNA expression which coincides with anemia. Administration of hesperidin as well as naringin potentially ameliorated the depressed adiponectin which may partially explain the alleviation in the hematological parameters in diabetic rats.
Thus, the present study revealed that both hesperidin and naringin could protect form diabetes associated anemia via their effects on the adipose tissue through attenuating the pro-inflammatory cytokine production and enhancement of adiponectin expression.
Conclusion
The findings of the present study indicate that hesperidin and naringin exert protection to diabetes-associated anemia in HFD/STZ diabetic rats. This could be due to the prevention or inhibition of lipid peroxidative system by their antioxidant and maintenance of cellular integrity, attenuation of proinflammatory cytokine production and stimulation of adiponectin expression. Moreover, the results suggest that both hesperidin and naringin are safe and capable of normalizing hematological abnormalities associated with diabetes mellitus thus could be prescribed as adjunct to dietary therapy and main therapy for diabetes mellitus.
References
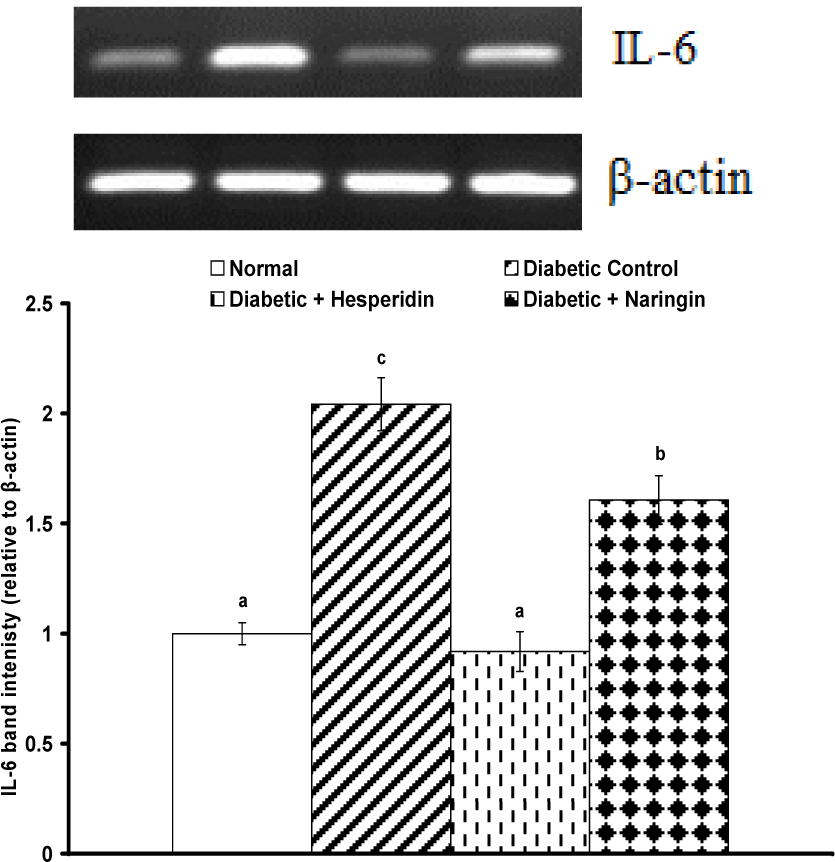
Figure 1: RT-PCR analysis of adipose tissue IL-6 and β-actin expression of normal, diabetic control and diabetic rats treated with hesperidin and naringin.
Top: Gel photograph depicting representative IL-6 and β-actin PCR products.
Bottom: Corresponding densitometric analysis of adipose IL-6 PCR products from 3 different experiments, represented as the IL-6 band intensity relative to β-actin.
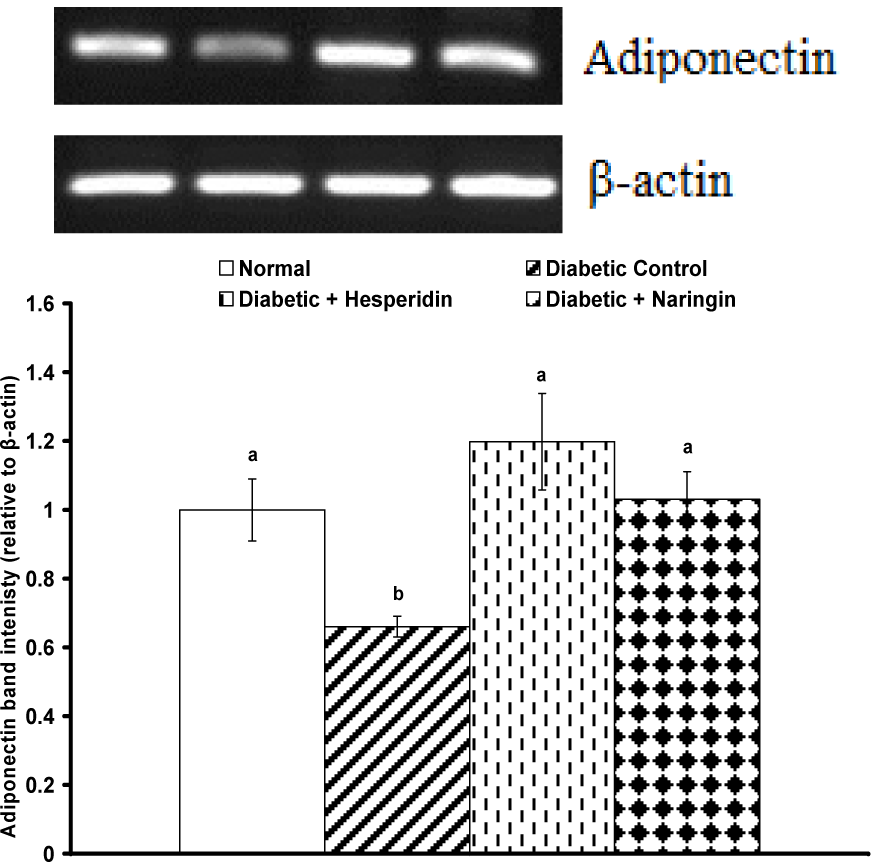
Figure 2: RT-PCR analysis of adipose tissue adiponectin and β-actin expression of normal, diabetic control and diabetic rats treated with hesperidin and naringin.
Top: Gel photograph depicting representative adiponectin and β-actin PCR products.
Bottom: Corresponding densitometric analysis of adipose adiponectin PCR products from 3 different experiments, represented as the adiponectin band intensity relative to β-actin.
[*] Corresponding Author:
Ayman M. Mahmoud, Physiology Division, Zoology Department, Faculty of Science, Beni-Suef University, Beni-Suef, Egypt; Tel.: +201144168280, eMail: ayman.mahmoud@science.bsu.edu.eg - aymano911@yahoo.com