Research article
Resveratrol inhibits proteinase-activated receptor-2-induced release of soluble vascular endothelial growth factor receptor-1 from human endothelial cells
Bahjat Al-Ani1[*]
1Department of Physiology, College of Medicine, King Khalid University, Abha 61421, Saudi ArabiaEXCLI J 2013;12:Doc598
Abstract
We recently reported that (i) activation of the proinflammatory receptor, proteinase-activated receptor-2 (PAR-2) caused the release of an important biomarker in preeclampsia, soluble vascular endothelial growth factor receptor-1 (sVEGFR-1, also known as sFlt-1) from human umbilical vein endothelial cells (HUVECs), and (ii) that the anti-oxidant and anti-inflammatory agent, resveratrol, is capable of inhibiting the proinflammatory cytokine-induced sVEGFR-1 release from human placenta. Based on these findings and because PAR-2 is upregulated by proinflammatory cytokines, we sought to determine whether resveratrol can inhibit PAR-2-induced sVEGFR-1 release. PAR-2 expressing cells, HUVECs and human embryonic kidney cells (HEK-293) transfected with a human VEGFR-1 promoter-luciferase reporter construct were incubated with PAR-2-activating peptide and/or resveratrol. Cell supernatants were assayed for sVEGFR-1 by enzyme-linked immunosorbent assay (ELISA), and VEGFR-1 promoter-luciferase assay was performed on the harvested cell lysates. Preincubation of HEK-293 cells with resveratrol significantly inhibited PAR-2-induced VEGFR-1 promoter activity without affecting cell viability as assessed by MTT assay. The addition of resveratrol also blocked PAR-2-mediated sVEGFR-1 release from HUVECs. The present study demonstrates that resveratrol suppressed both VEGFR-1 promoter activity and sVEGFR-1 protein release induced by PAR-2 activation, which further endorses our recent findings of a potential therapeutic role for resveratrol in preeclampsia.
Keywords: PAR-2, resveratrol, sVEGFR-1/sFlt-1, endothelial cells, preeclampsia
Introduction
Preeclampsia is a pregnancy complicated disease characterised by aggravated inflammation (Rodie et al., 2004[22]) and widespread of maternal endothelial damage with a clinical presentation of hypertension and proteinuria after 20 weeks gestation that affects about 5 % of all pregnancies and women (Sibai et al., 2005[24]). A systematic review and meta-analysis has documented that women with preeclampsia are at an increased risk of developing cardiovascular disease later in life (Bellamy et al., 2007[4]). High levels of the anti-angiogenic factor, soluble vascular endothelial growth factor (VEGF) receptor-1 (sVEGFR-1, commonly known as sFlt-1) are observed in pregnancies complicated with preeclampsia (Chaiworapongsa et al., 2005[8]; Hertig et al., 2004[13]; Koga et al., 2003[17]; Levine et al., 2004[18]). Elevated sVEGFR-1 antagonises the action of VEGF and placenta growth factor resulting in impaired human placental angiogenesis (Ahmad and Ahmed, 2004[1]) and causes glomerular endothelial cell damage, proteinuria and hypertension in rodent models (Maynard et al., 2003[20]; Sugimoto et al., 2003[27]).
PAR-2 plays an important role in inflammation and regulates vascular function (Al-Ani et al., 1995[3]; Steinhoff et al., 2005[26]; Vergnolle 1999[28]). Pro-inflammatory cytokines such as TNFα and IFNγ induce PAR-2 expression and in turn activation of PAR-2 promotes the production of TNFα, IFNγ, IL-8 and IL-18 in various cell types including the endothelium and endometrium (Cenac et al., 2004[7]; Ikawa et al., 2005[14]; Nystedt et al., 1996[21]; Zhu et al., 2006[31]). PAR-2 expression is reported to be increased in umbilical vein endothelial cells derived from preeclamptic pregnancies, and the conditioned medium from preeclamptic placental villous tissue explants up-regulate PAR-2 in cultured endothelial cells (Wang et al., 2002[29]). We recently reported the release of sVEGFR-1 from endothelial cells and an increase in the VEGFR-1 promoter activities upon PAR-2 activation (Al-Ani et al., 2010[2]). In addition, we also recently reported (Cudmore et al., 2012[10]) that in human placenta, the chemical substance obtained from the skin of grapes and other fruits, resveratrol (Bertilli et al., 1995[5]; Frankel et al., 1993[11]; Singh et al., 2011[25]) blocked the release of sVEGFR-1 from human placenta incubated with the proinflammatory cytokine, TNFα. Therefore, we speculated that resveratrol is capable of suppressing PAR-2-mediated sVEGFR-1 release and VEGFR-1 promoter activity.
Materials and Methods
Reagents
PAR-2 peptide, 2-furoyl-LIGRLO-NH2 (2f-LIGRLO) and its reverse control peptide, 2-furoyl-OLRGIL-NH2 (2f-OLRGIL) were gift from Professor Morley D Hollenberg, Faculty of Medicine, University of Calgary, Calgary, Alberta, Canada. Human DuoSet VEGFR1 ELISA kit was purchased from R&D Systems, UK, and all other cell culture reagents and chemicals were obtained from Sigma Aldrich (Poole, UK).
Cell culture
Human umbilical vein endothelial cells (HUVEC) were isolated and cultured as described previously (Bussolati et al., 2001[6]). Informed consent was obtained from healthy pregnant women undergoing elective caesarean section at Birmingham women's hospital under ethical approval [RCS10-0546.APP/N10-014]. Cords were collected on the day of surgery and washed with PBS to remove excessive blood and proceed for HUVEC isolation and culture. Experiments were performed on second or third passage HUVEC. HEK-293 human embryonic kidney cells were maintained in DMEM containing 10 % FCS.
Enzyme-linked immunosorbent assays
Enzyme-linked immunosorbent assays to determine the concentrations of sVEGFR-1 in cell supernatants were performed as described previously (Ahmad and Ahmed, 2004[1]) using the human DuoSet VEGFR1 ELISA kit according to the manufacturer's instructions (R&D Systems, UK).
MTT assay for cell viability
HEK-293 cells were seeded at a density of 1x 104 cells/well in a 96-well plate and incubated overnight at 37 °C in growth medium. Cells were incubated in triplicate with resveratrol in medium containing 5 % FCS for 24 hours. Then, 5 mg/ml MTT (3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide was added and the plate incubated in the dark at 37 °C for 4 hours. The MTT solution was removed and DMSO (150 l/well) added and the plate agitated for 5 minutes. The optical density was measured at 570 nm and 630 nm and DMSO alone was used as a blank.
VEGFR-1 promoter reporter assays
As described previously (Al-Ani et al., 2010[2]), a 1.3 Kb fragment of the human VEGFR-1 promoter starting from -1500 bp relative to the start codon was cloned into the pGL2 luciferase reporter plasmid (pVEGFR-1luc) and was used to determine the ability of PAR-2 to activate the VEGFR-1 gene in HEK-293 cells which express high levels of PAR-2 (Kawabata et al., 1999[16]). Luciferase reporter construct was introduced into HEK-293 cells using ExGen 500 (EuroMedex, France) as described previously (Al-Ani et al., 2010[2]). Following stimulation for 24 hours, luciferase activity was determined in cell lysates using the Dual Luciferase Assay (Promega) as described previously (Al-Ani et al., 2010[2]).
Statistical analysis
All data are expressed as the mean ± SEM. Statistical analysis was performed using the two-tailed Student's t-test and p < 0.05 was considered statistically significant.
Results
To investigate whether the anti-inflammatory agent, resveratrol, can inhibit PAR-2-mediated activation of VEGFR-1 gene promoter, we transfected HEK-293 cells that express PAR-2 with a plasmid DNA construct carrying 1300 bp human VEGFR-1 promoter luciferase reporter (Al-Ani et al., 2010[2]). As expected, activation of PAR-2 with 10 νmol/L 2f-LIGRLO-NH2 peptide caused more than a 2-fold increase in VEGFR-1 promoter activity, which was almost completely blocked by resveratrol (Figure 1, A(Fig. 1)). Resveratrol also suppressed the basal level of VEGFR-1 promoter activity in the unstimulated cells (Figure 1A(Fig. 1)). The effect of resveratrol was specific since using the same inhibitory dose (25 νmol/L) was not toxic to HEK-293 cells (Figure 1B(Fig. 1)). The control reverse peptide 2f-OLRGIL-NH2 failed to activate VEGFR-1 promoter DNA and did not interfere with the inhibitory action of resveratrol (data not shown).
Endothelial cells have been shown to secrete sVEGFR-1 in response to PAR-2 activation (Al-Ani et al., 2010[2]). To determine whether the observed inhibitory effect of resveratrol on PAR-2 stimulating VEGFR-1 gene can further be extended to include inhibition of the release of sVEGFR-1 protein, HUVECs were incubated for 24 hours with 10 νmol/L 2f-LIGRLO-NH2 in the presence or absence of 50 νmol/L resveratrol, and sVEGFR-1 was assayed in cell supernatants. Resveratrol significantly inhibited sVEGFR-1 release under basal condition and after stimulation (Figure 2, A(Fig. 2)). The control reverse peptide 2f-OLRGIL-NH2 caused no release of sVEGFR-1 and did not interfere with the action of resveratrol (Figure 2, B(Fig. 2)). We have shown before that resveratrol up to 100 νmol/L were not toxic to HUVECs (Cudmore et al., 2012[10]).
Discussion
The present report demonstrates that resveratrol, found in high concentrations in the skin of red grapes, inhibits VEGFR-1 promoter activation by PAR-2. Furthermore, it shows that resveratrol suppresses PAR-2-induced sVEGFR-1 release from endothelial cells and supports our recent study showing that resveratrol inhibits cytokine-induced sVEGFR-1 release from human placenta (Cudmore et al., 2012[10]).
The WHO (2005[30]) reports over 60,000 maternal deaths worldwide per year as a consequence of preeclampsia. Preeclampsia is responsible for 30 % of all premature deliveries, and delivery of the baby and the placenta is still the only available effective therapy (Haram et al., 2003[12]). We suggested before (Al-Ani et al., 2010[2]) that activation of PAR-2 may be a contributing factor to the increases in the circulating levels of sVEGFR-1 in preeclampsia. This was based on the increased procoagulation activity in this syndrome and the ability of the coagulation proteinases to activate PAR-2 (Kaibara et al., 1999[15]; Manithody et al., 2012[19]; Rowland et al., 2000[23]; Wang et al., 2002[29]). Furthermore, our reports on the augmentation of PAR-2-mediated sVEGFR-1 levels upon inhibition of heme oxygenase 1 (HO-1) enzyme (Al-Ani et al., 2010[2]) and that higher levels of sVEGFR-1 were secreted from lung biopsy explants of HO-1 knockout mice compared with wild-type litter mates (Cudmore et al., 2007[9]) suggesting that HO-1 is a negative regulator of PAR-2-mediated sVEGFR-1 release in endothelial cells and loss of HO-1 activity may be central to the pathogenesis of preeclampsia. Therefore, our recent publication in this field that resveratrol upregulates HO-1 (Cudmore et al., 2012[10]) would strengthen the finding in this study showed resveratrol inhibited PAR-2-induced sVEGFR-1 release and would further promote resveratrol as a potential drug to ameliorate the symptoms of preeclampsia.
Acknowledgements
The support and input of Dr Peter Hewett from the School of Experimental Medicine, College of Medicine and Dentistry, University of Birmingham, United Kingdom, is highly appreciated by the author of this work. Also, the author acknowledges the help of Dr Luke Nwoye from the Department of Physiology, College of Medicine, King Khalid University, Abha, KSA. This work was supported by King Khalid University Research grant number 245. We report no conflict of interest.
References
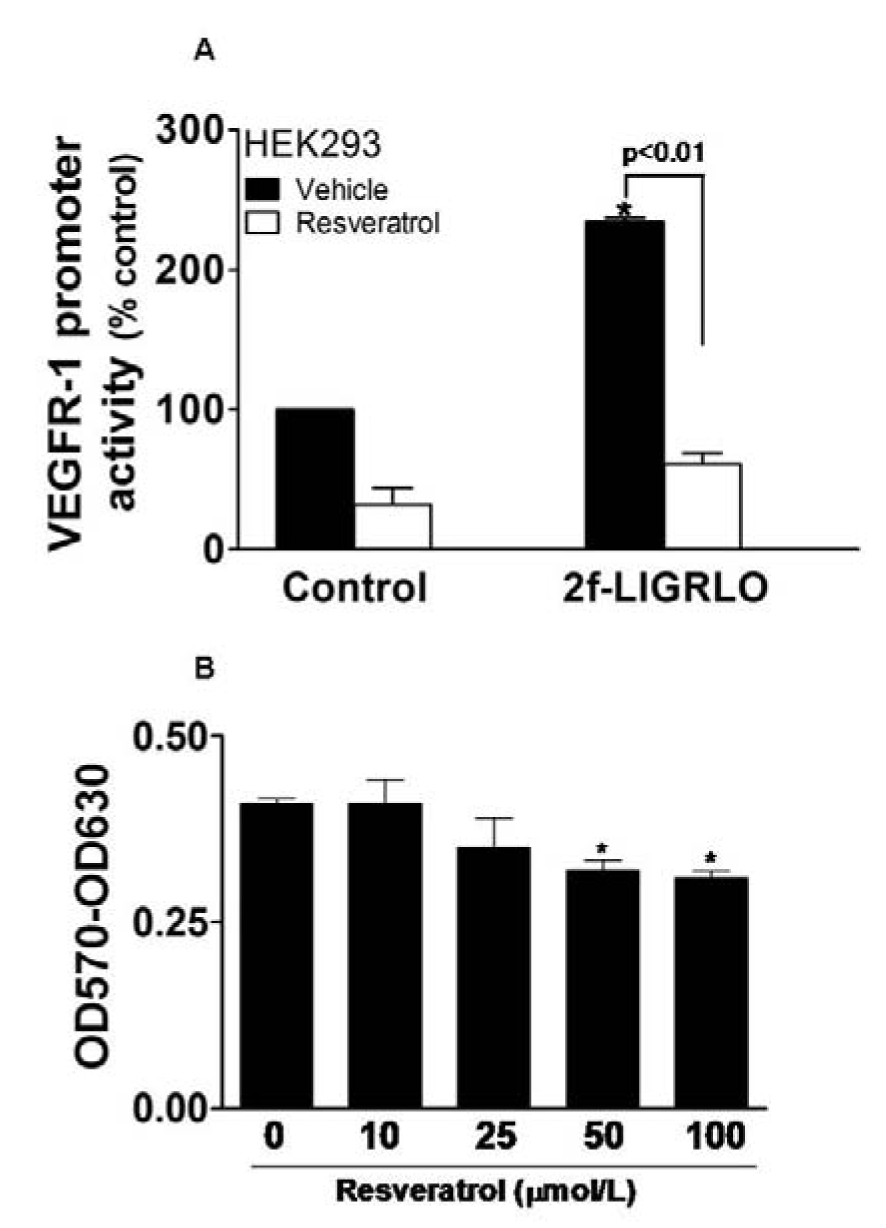
Figure 1: Resveratrol inhibits PAR-2-induced activation of the VEGFR-1 gene promoter.
A: HEK-293 cells transfected with a ~1.3 kb fragment of the VEGFR-1 promoter with a luciferase reporter were incubated with the specific PAR-2 activator, 2f-LIGRLO-NH2 (10 νmol/L) for 24 hours in the presence and absence of 25 νmol/L resveratrol. VEGFR-1 promoter activities in cell lysates were determined by luciferase assay.
B: HEK-293 cells were incubated for 24 hours with increasing concentrations of resveratrol in medium containing 5 % FCS and cell viability assessed by MTT assay. Results represent the mean (±SEM). *, p < 0.01 vs control for (A) and *, p < 0.05 vs control for (B)
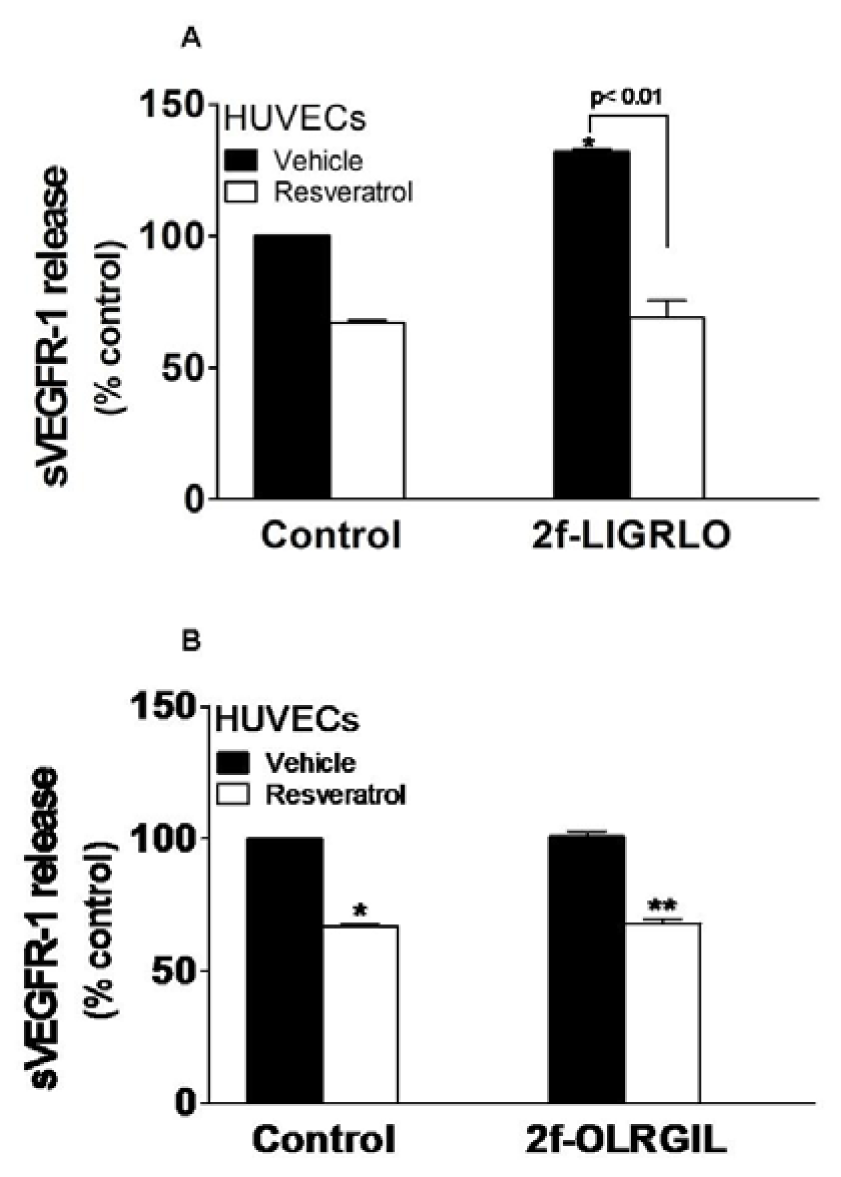
Figure 2: Resveratrol inhibits PAR-2-induced sVEGFR-1 release. HUVECs were incubated with (A) 10 µmol/L 2f-LIGRLO-NH2 or (B) 10 νmol/L 2f-OLRGIL-NH2 control peptide for 24 hours in the presence and absence of 50 µmol/L resveratrol. Cell supernatants were assayed for sVEGFR-1 by ELISA. Results represent the mean (±SEM). *, p < 0.01 vs control vehicle; **, p < 0.01 vs control peptide
[*] Corresponding Author:
Bahjat Al-Ani, Department of Physiology, College of Medicine, King Khalid University, Abha 61421, Saudi Arabia; Tel: + 966-7-2418818, eMail: bahjat_alani@yahoo.com