Research article
Contralateral cortical role on functional recovery in a rat model of hemiplegia
Satoshi Ikeda1[*], Katsuhiro Harada1, Akihiko Ohwatashi1, Yurie Kamikawa1, Akira Yoshida1
1Department of Rehabilitation and Physical Medicine, Graduate School of Medical and Dental Sciences, Kagoshima UniversityEXCLI J 2013;12:Doc641
Abstract
The role of the contralateral cerebral cortical plasticity in functional recovery after cerebral infarction is controversial. To clarify this role, we made a second contralateral cortical infarction after recovery from the first cerebral infarction. To produce the first infarction, Wistar rats were intravenously injected with Rose Bengal to the sensorimotor area of the right hemisphere of the cerebral cortex under green-light irradiation. Two weeks after the first hemiplegia, a secondary infarction was induced in the left cerebral cortex. Functional recovery was evaluated in a beam-walking test. Hemiplegia observed 1 day after both the first and second infarctions was given a score of 1. At 14 days after the first infarction, the average recovery score (± standard error) was 6.8 ± 0.1. In contrast, functional recovery was slower after the second infarction, reaching an average score of only 3.5 ± 0.5 after 14 days. Therefore, recovery after the contralateral secondary infarction was slower than that from the first, and received a lower recovery score compared to the recovery after the first infarction. These results suggest that the undamaged contralateral cortex plays an important role in motor recovery after hemiplegia caused by cerebral infarction.
Keywords: stroke, animal model, recovery, photochemical infarction
Introduction
Plasticity on cerebral cortex has an important role in functional recovery from dysfunction on cerebral infarction. Clinically serious hemiplegia can be caused by recurrent contralateral infarctions (Jorgensen et al., 1997[10]; Kim, 1999[11]; Alexander et al., 2010[2]; Paciaroni and Bogousslavsky, 2010[14]; Sterr and Conforto, 2012[17]). Calautti and Baron (2003[4]) reported that the contralateral cortex participates in functional recovery from cerebral infarction. Functional magnetic resonance imaging (fMRI) studies of patients with hemiparetic stroke clearly demonstrate that bilateral activation occurs in contralesional premotor and motor cortical areas during movement of the affected hand (Johansen-Berg et al., 2002[9]).
Recovered forelimb function after MCA occlusion was impaired by Lidocaine injection to the contralateral undamaged cortex (Biernaskie et al., 2005[3]). Additionally, adaptive changes take place in the vicinity of the infarction (Frost et al., 2003[7]). Few studies have investigated the role of the contralateral cortex in functional recovery after bilateral cortical infarction. A report indicated that there is only relatively weak contralateral participation in an animal model of small infarction (Shanina et al., 2006[16]). However, the role of functional recovery after a large, bilateral infarction has not been studied. Therefore, to clarify the role of contralateral participation after large cerebral infarctions, we studied the functional effects of contralateral secondary infarction after functional recovery from a primary infarction.
Materials and Methods
Ten eight-week-old male Wistar rats weighing 250 - 300 g were used in this study. The study protocol was carried out in accordance with the guide for animal experimentation of the faculty in the department of medicine of Kagoshima University and the guidelines of the US national institute of health. This study was approved by the animal experiment committee of Kagoshima University. These animals were housed alone in plastic cages in an environmentally controlled room with a 12/12-hour light-dark cycle ad libitum.
Cerebral ischemia was produced via intravascular thrombosis. This thrombosis was induced by an intravenous injection of Rose Bengal (20 mg/kg) and irradiation by green light (533 nm) for 20 min under deep anesthesia. To target the sensorimotor area of the cerebral cortex, green light of 10 mm diameter was irradiated on the exposed skull at 6 mm lateral to the midline and 4 mm posterior to bregma (Horinouchi et al., 2007[8]). Two weeks after recovery from the first hemiplegia, a secondary infarction was induced in the left cerebral cortex. The postoperative locomotion of rats was evaluated using a beam-walking task on an elevated narrow beam (1,220 mm length x 25 mm width). Performance was rated daily on a 7-point scale. The lowest score, 1, was given if the animal was unable to traverse the beam and could not place the affected hind limb onto the horizontal surface. A score of 2 was given if the animal was unable to traverse the beam but placed the affected hind limb on the horizontal surface of the beam and maintained balance. An animal received a score of 3 if it traversed the beam while dragging the affected hind limb. If an animal traversed the beam and placed the affected hind paw on the horizontal surface of the beam at least once, it was given a score of 4. A score of 5 was given if the animal used the affected limb in less than half of the steps along the beam. When an animal traversed the beam and used the affected limb to aid more than 50 % of its steps along the beam, it received a score of 6. The highest score, 7, was given if the animal traversed the beam normally with fewer than 2 independent foot slips (Feeney et al., 1982[6]). Rats that were given a score of 1 the day after the operation were used in subsequent analysis. After perfusion fixation, animal brains were removed and the infarction area was measured. Statistical analyses were carried out using the Wilcoxon signed-rank test (P < 0.01).
Results
Hemiparesis was observed after the first infarction since animals could not traverse the beam or position their affected hind limbs on the horizontal surface. The average scores for left hemiplegia were 1.0, 3.5, 6.8, and 6.8 at 1, 3, 7, and 14 d after the first infarction, respectively. All animals had almost fully recovered from left hemiplegia by day 14 after the first infarction. In contrast, after the second infarction, the average scores for right hemiplegia were 1.0, 1.2, 2.7, and 3.5 after 1, 3, 7, and 14 d, respectively. A significant difference in the rate of recovery was observed only 2 days after the second infarction (Figure 1(Fig. 1)).
On the second day after infarction, the average beam-walking score for the contralateral secondary infarction group remained at 1, compared to 3 for the unilateral primary infarction group. After 3 days, animals from the secondary infarction group began to recover. Seven days after the procedure, the average score for rats in the secondary infarction group had increased to 2.7, while the score for the unilateral primary infarction group had improved to 6.2.
After functional analysis, experimental animal brains were removed to measure the area of infarction. Scar of first infarction is seen on right hemisphere (Figure 2A, B(Fig. 2)) and subacute infarcted lesion is located on left hemisphere (Figure 2A, C(Fig. 2)). There was no significant difference between the average area of the affected region in the right hemisphere after the first infarction and corresponding value in the left hemisphere after the second infarction (Table 1(Tab. 1)).
Discussion
Previous clinical reports suggest that patients with a contralateral recurrence after their first stroke showed more severe functional disability after rehabilitation than did patients with an ipsilateral recurrence (Jorgensen et al., 1997[10]; Kim, 1999[11]; Alexander et al., 2010[2]; Paciaroni and Bogousslavsky, 2010[14]; Sterr and Conforto, 2012[17]). The photothrombotic ischemic model has been used in recent years (Abo et al., 2001[1]; Horinouchi et al., 2007[8]; Zheng et al., 2010[19]; Diederich et al., 2012[5]) to induce different sized strokes, while decreasing surgical stress in experimental rats. In this study, serious paralysis was shown to result from a series of bilateral secondary infarctions, and this finding mirrors previous clinical reports. Similarly, a study of motor cortical excitability in the non-lesioned hemispheres of patients with large cortical infarctions showed that intracortical inhibition was reduced (Liepert et al., 2000[13]). In a recent clinical study using transcranial magnetic stimulation, some patients were able to recover upper limb function, suggesting an alteration in the cortical representation to the ipsilateral target muscle in the non-affected hemisphere (Summers et al., 2007[18]). Similarly, in patients with an infarction of the posterior limb of the right internal capsule, the bilateral primary sensorimotor cortices are activated by movements of the affected (left) hand, and ipsilateral motor-evoked potentials are found in the affected muscle after stimulation of the unaffected (left) motor cortex (Kim et al., 2004[12]). Furthermore, an fMRI study measuring recovery from infarction in rats showed that electrical stimulation of the previously paretic hindlimb activated both the non-damaged contralateral sensorimotor cortex and the region lateral to the damaged area (Abo et al., 2001[1]). Immunohistochemical analysis of brain sections of animal models of infarction showed that gamma-aminobutyric acid (A)-receptor subunits were observed in the contralateral cortex homotopic to the infarct (Redecker et al., 2002[15]). These findings suggest that the plasticity of contralateral undamaged cortex plays an important role in cerebral damage.
However, in a study of contralateral secondary infarction, Shanina et al. (2006[16]) induced small ischemic cortical infarcts in the sensorimotor cortex and demonstrated that the contralateral cortex homotopic to the lesion plays only a minor role in functional recovery. In contrast, this study demonstrates that recovery from hemiplegia caused by a secondary infarction was relatively slow (the average beam-walking score remained at only 3.5 after 2 weeks), suggesting participation of the contralateral cortex in functional recovery.
These results suggest that both the contralateral cortex and the region surrounding the infarction participate in the functional recovery process. In the present study, the infarct size after the first procedure was relatively large leaving only a comparatively small area of intact cortex remaining on the ipsilateral side. Therefore, we consider participation of the contralateral cortex to be necessary for functional recovery from large cortical lesions.
In previous studies on animal models of cerebral infarction, almost all rats recovered from functional disability within 2 weeks (Feeney et al., 1982[6]; Horinouchi et al., 2007[8]). Alternatively, in our bilateral infarction model, functional disability lasted for more than 2 weeks. These findings suggest that our infarction model is more useful for understanding lasting disability.
Conclusion
Here, we found that functional recovery after a large contralateral secondary infarction was slow and unsatisfactory compared to recovery after a large primary infarction. Our results suggest that the undamaged contralateral cortex plays an important role in motor recovery after hemiplegia caused by large cerebral infarction. Furthermore, we believe that this experimental model accurately mirrors lasting disability in patients.
Conflict of interest
The authors declare no competing financial interests.
Acknowledgements
The authors thank the staff of The Frontier Science Research Center Kagoshima University to manage animals.
References
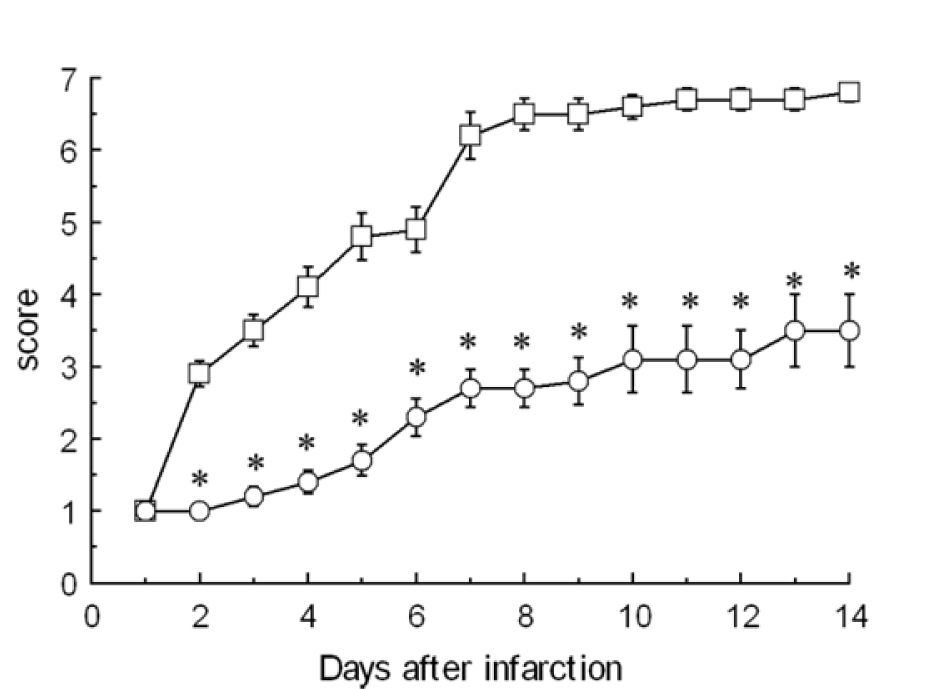
Figure 1: Changes in functional recovery between first and second contralateral infarctions. The average score for the beam-walking test is shown (n = 10). Circles denote the average score after the first infarction. Squares denote the average scores after the second infarction. Asterisk (*) indicates significant difference (P < 0.01) between first and second infarctions according to the Wilcoxon signed-rank test.

Figure 2: Representative brain removed 2 weeks after the second infarction (A) and photomicrograph of sliced section (B, C). The first infarction is located in the right hemisphere (A, B), while the second infarction is located in the left hemisphere (A, C).
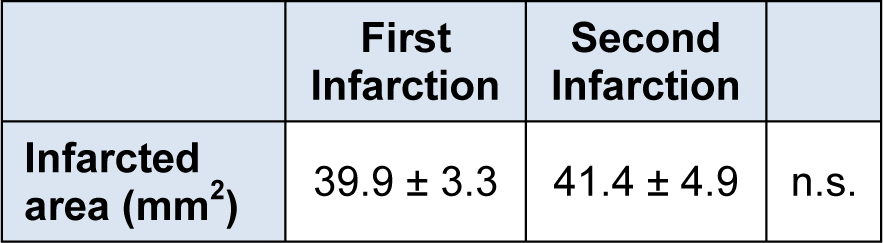
Table 1: Infarction sizes across all experimental animals. The infarction area (mean ± standard error) for all 10 animals of first and second infarctions
[*] Corresponding Author:
Satoshi Ikeda, Department of Rehabilitation and Physical Medicine, Graduate School of Medical and Dental Sciences, Kagoshima University, 8-35-1 Sakuragaoka, Kagoshima-shi, Kagoshima 890-8520, Japan; Tel.: +81-99-275-5339; Fax: +81-275-1273, eMail: ikedas@m2.kufm.kagoshima-u.ac.jp