Research article
Chemical composition, antioxidant and antigenotoxic activities of different fractions of Gentiana asclepiadea L. roots extract
Vladimir Mihailovic1[*], Sanja Matic2, Danijela Mišic3, Slavica Solujic1, Snežana Stanic2, Jelena Katanic1, Milan Mladenovic1, Nevena Stankovic1
1Department of Chemistry, Faculty of Science, University of Kragujevac, Radoja Domanovica 12, 34000 Kragujevac, Serbia2Department of Biology and Ecology, Faculty of Science, University of Kragujevac, Radoja Domanovica 12, 34000 Kragujevac, Serbia
3Institute for Biological Research "Siniša Stankovic", University of Belgrade, Bulevar despota Stefana 142, 11060 Belgrade, Serbia
EXCLI J 2013;12:Doc807
Abstract
The aim of this study was to evaluate the antioxidant and antigenotoxic activities of chloroform, ethyl acetate and n-butanol fractions obtained from Gentiana asclepiadea L. roots methanolic extract. The main secondary metabolites sweroside, swertiamarin and gentiopicrine were quantified in G. asclepiadea root extracts using HPLC-DAD analysis. Amount of total phenols, flavonoids, flavonols and gallotannins was also determined. The antigenotoxic potential of extracts from roots of G. asclepiadea was assessed using the standard in vivo procedure for the detection of sex linked recessive lethal mutations in Drosophila melanogaster males treated with ethyl methanesulfonate (EMS). The results showed that the most abundant secoiridoid in G. asclepiadea roots was gentiopicrine and its content in the n-butanol fraction (442.89 mg/g) was the highest. Among all extracts, ethyl acetate fraction showed the highest antioxidant activity, as well as total phenolics (146.64 GAE/g), flavonoids (44.62 RUE/g), flavonols (22.71 RUE/g) and gallotannins (0.99 mg GAE/g) content. All the fractions showed antioxidant activity using in vitro model systems and the results have been correlated with total phenolics, flavonoids, flavonols and gallotannins content. In addition to antioxidant activity, G. asclepiadea root extract fractions possess an antigenotoxic effect against DNA damage induced by alkylation with EMS. The antioxidant activity exhibited by G. asclepiadea depended on the phenolic compounds content of the tested extracts, while there was no significant difference in the antigenotoxic potential between fractions.
Keywords: antioxidant activity, antigenotoxicity, gentiopicrine, Gentiana asclepiadea L., phenolic contents, secoiridoids
Introduction
With recent advances in medical and nutrition sciences, natural products and health-promoting foods have received extensive attention from both health professionals and the common population (Zhao, 2007[63]). Many herbs and spices are the subject of ongoing scientific investigations related to antioxidant properties and health (Kaefer and Milner, 2008[28]). Nowadays, there is a growing interest in bioprospecting and the analysis of novel natural antioxidants for use in foods. Namely, some of synthetic antioxidants, such as butylated hydroxytoluene (BHT) and butylated hydroxyanisole (BHA), which have been widely used in foods and beverages, showed potential health hazards, because of the formation of possible toxic or carcinogenic components during their degradation (Dastmalchi et al., 2007[7]; Hsouna et al., 2011[17]). The protective effects of plant products are ascribed to several components which have distinct mechanisms of action, including low molecular weight compounds, e.g. vitamins E and C, carotenoids and so-called non-nutrient antioxidants, e.g. phenolic and polyphenolic compounds (Fürst, 2009[14]; Kim et al., 2011[32]). Most antioxidants isolated from higher plants are phenolic compounds (e.g. phenolic acids, flavonoids, flavonols, catechins, lignans and tannins), which have diverse biological activities, such as anti-inflammatory, anti-carcinogenic and anti-atherosclerotic effects, as a result of their antioxidant activity (Kaisoon et al., 2011[30]; Krishnaiah et al., 2011[35]).
In the past few years, there has been significant interest in natural products with antigenotoxic properties. Plants used in traditional medicine, as well as their secondary metabolites, have been proposed for their antigenotoxic activities (De Flora, 1998[8]; Alekperov, 2002[3]; El Hamss et al., 2003[12]). Previous studies on the genotoxicity and antigenotoxicity of plant extracts can help evaluate the safety of these herbal products (Romero-Jiménez et al., 2005[51]; Alpsoy et al., 2011[4]). Therefore, it is necessary to assess the antigenotoxic potential of plant extracts.
Gentiana asclepiadea L. belongs to the genus Gentiana that comprises about 400 species. Of the 29 Gentiana species recorded in Europe, eleven of them are distributed in Serbia (Josifović, 1973[27]). In general, the medicinal part of Gentiana spp. is the root. The underground parts of various Gentiana species have been included in many herbal formulations as remedies for poor appetite, digestive problems and as hepatoprotective agents worldwide. As natural sources of food flavoring they are utilized in alcoholic and nonalcoholic beverages (Jensen and Schripseme, 2002[25]; Szücs et al., 2002[56]; Jiang et al., 2005[26]; Aberham et al., 2011[2]). The major bioactive constituents of Gentiana plants are iridoids, secoiridoides, xanthones, and flavonoids (Krstić et al., 2004[36]; Zhao et al., 2010[64]). Swertiamarine, gentiopicrine, sweroside and amarogentin are the main secoiridoids of Gentiana plants. Because its bitter taste, secoiridoids are used in preparation different bitter tonics (Wolfender et al., 1993[59]).
Gentiana asclepiadea L. is medicinal plant which used in Serbia as traditional medicine for hepatitis infections and the local name of this plant is a "grass of jaundice". Also, herb and roots of this plant are widely used for improving digestion (Sarić, 1989[52]; Menković et al., 2011[43]). Secoiridoid-glycosides: gentiopicrine, its 6'-O-glycoside, swertiamarine and sweroside (Mpondo and Chulia, 1988[45]; Szücs et al., 2002[56]); flavone- and xanthone-C-glycosydes and phenolic acids (Kitanov and Spassov, 1992[33]) are the secondary metabolites identified in G. asclepiadea. However, no correlation has been proved between the biochemical composition of this plant and its biological activity or therapeutic uses. Potential antioxidant activity of some Gentiana species, such as G. lutea (Kušar et al., 2006[39]) and G. arisanensis (Ko et al., 1998[34]) have been confirmed. In addition, our previous studies also showed that methanol extract of aerial parts of G. asclepiadea had a good antioxidant activity in vitro (Nićiforović et al., 2010[46]) and aerial parts and roots extracts had hepatoprotective activities against carbon tetrachloride induced liver injury in rats (Mihailović et al., 2013[44]). Previous study showed that G. asclepiadea extracts are non-genotoxic to rat liver cells (Mihailović et al., 2013[44]). Therefore, the objectives of the present study were to evaluate the antioxidant, radical-scavenging and antigenotoxic activities of chloroform, ethyl acetate and n-butanol fractions obtained from the root extract of G. asclepiadea, as an alternative natural source of antioxidants which can potentially be used as functional food ingredients. In addition to antioxidant activity, the antigenotoxic potential of fractions was assessed in Drosophila melanogaster treated with the well-established mutagenic agent ethyl methanesulfonate (EMS) using the standard in vivo sex-linked recessive lethal (SLRL) test. The amounts of gentiopicrine, swertiamarin and sweroside were determined by HPLC-DAD analyses. At the same time, phenolic content of the same plant material was determined in order to evaluate their amount, as well as their contribution to the antioxidant capacity.
Materials and Methods
Chemicals and apparatus
All chemicals used in experiments were of analytical grade and were purchased from Sigma Chemical Co. (St. Louis, MO, USA), Aldrich Chemical Co. (Steinheim, Germany) and Alfa Aesar (Karlsruhe, Germany). Standard compounds gentiopicrine (≥ 98 % purity) was obtained from Roth, Karlsruhe, Germany. Swertiamarin and sweroside (both 98 % purity) were obtained from Oskar Tropitzsch, Marktredwitz, Germany and mangiferin (≥ 98 % purity) was purchased from Sigma-Aldrich, Steinheim, Germany. The spectrophotometric measurements were performed on UV-VIS spectrophotometer MA9523-SPEKOL 211 (ISKRA, Horjul, Slovenia).
Plant material
G. asclepiadea was collected from Jadovnik Mountain (Gostun-Kumanica, at the Serbia-Montenegro border) in the month of October during its flowering time. The collected plant was authenticated and voucher specimen (No. 16337) was deposited in the Herbarium of the Department of Botany, Faculty of Biology, University of Belgrade, Belgrade, Serbia.
Preparation of the extracts
The roots (90 g) of G. asclepiadea were dried and fine powdered. The obtained powder was extracted with methanol (3 × 500 ml, 24 h each) at room temperature. After filtration through Whatman No. 1 filter paper, the extract was concentrated under vacuum at 40 °C by using a rotary evaporator, yielding brown residues (32.49 g). The resulted residue was suspended in 500 ml of distilled water and sequentially partitioned into chloroform (3 × 100 ml), ethyl acetate (3 × 100 ml) and then n-butanol (3 × 100 ml). The resulting three solutions were concentrated in vacuo to dryness, to give chloroform (0.621 g), ethyl acetate (0.217 g) and n-butanol (2.846 g) soluble fractions. The extracts were stored in darkness at 4 °C until used.
Determination of total phenolics content
The total phenolics content was determined by the method as described by Singleton et al. (1999[53]). A reaction mixture consisting of 0.5 ml of extracts (1 mg/ml), 2.5 ml of Folin-Ciocalteu reagent (diluted 10-fold) and 2 ml of NaHCO3 (7.5 %) was prepared. After incubation for 15 min at 45 °C, the absorbance was measured at 765 nm. The mean of three independent analyses were used and the total phenol content was expressed in milligrams of gallic acid equivalents/g extract (mg GAE/g extract).
Determination of flavonoids content
The total flavonoid content determination was based on the method reported Brighente et al. (2007[6]). 0.5 ml of 2 % AlCl3 and the same volume of methanol solution of plant extracts were incubated for 1 h at room temperature, and the absorbance was measured at 415 nm. The mean of three independent analyses were used and the total flavonoids content was expressed in milligrams of rutin equivalents/g extract (mg RUE/g extract).
Determination of flavonols content
The content of flavonols was determined by Yermakov et al. (1987[62]) method. The rutin calibration curve was prepared by mixing 2 ml of various concentrations of ethanolic solutions of rutin with 2 ml (20 mg/ml) aluminium trichloride and 6 ml (50 mg/ml) sodium acetate. The absorbance at 440 nm was read after 2.5 h. The same procedure was used for 2 ml of plant extract (10 mg/ml) instead of rutin solution. All determinations were carried out in three independent experiments. The content of flavonols was calculated as rutin equivalents (mg RUE/g dry extract).
Determination of gallotannins content
The content of gallotannins was determined by Haslam method (1965[16]). The assay is based on the reaction of potassium iodate (KIO3) with galloyl esters, which will form a red intermediate and ultimately a yellow compound. The concentration of the red intermediate can be measured spectrophotometrically at 550 nm. The reaction was initially performed by adding 1.5 ml of a saturated potassium iodate solution to 3.5 ml of methanol solution of plant extracts, followed by incubation at 15 °C until a maximum absorbance was reached (regardless of the time) (Bate-Smith, 1977[5]). Gallotannins content was determined as gallic acid equivalents (mg GAE/g extract) and the values are presented as means of three independent experiments.
HPLC analysis of secoiridoids and xanthones
The amount of compounds swertiamarin, gentiopicrine, sweroside, and mangiferin in fractions obtained from the root extract of G. asclepiadea was determined by the HPLC-DAD method as described previously (Mihailović et al., 2013[44]).
Determination of total antioxidant capacity
Determination of total antioxidant capacity is based on the reduction of Mo (VI) - Mo (V) by the antioxidant compounds. In order to determine total antioxidant capacity of extracts the method of Prieto et al. (1999[49]) was used. The total antioxidant activity of extracts was monitored by the formation of a green phosphate/Mo (V) complex at acid pH. Briefly, 0.3 ml of extracts in methanol (1 mg/ml) was added to 3 ml of reagent solution (0.6 M sulfuric acid, 28 mM sodium phosphate and 4 mM ammonium molybdate). Obtained mixtures were incubated at 95 °C for 90 min. The absorbance was recorded at 695 nm after cooling to room temperature. The results were evaluated through the standard curve of ascorbic acid (AA) obtained by the same procedure. The total antioxidant capacity is expressed as milligrams of AA per gram of the dry extract.
Determination of DPPH free-radical scavenging activity
In order to determine scavenging DPPH radical activity of the extracts, the method proposed by Takao et al. (1994[57]) was used with slight modification (Kumarasamy et al., 2007[37]). Different concentracions of extracts in methanol (2 ml each) were mixed with DPPH (2 ml, 80 νg/ml). After 30 min of incubation at room temperature, the absorbance was measured at 517 nm. Each sample was analyzed in three independent experiments. Ascorbic acid (AA), gallic acid (GA) and butylated hydroxytoluene (BHT) were used as reference standards. The precentage of DPPH free radical scavenging activity was calculated by following equation:
% inhibition = [(AC - AS)/AC] x 100
where AC is the absorbance of the control solution and AS is the absorbance for the fraction in the presence the DPPH solution. The concentration of extracts providing 50 % scaviening (IC50) was calculated from the sigmoidal dose-response curve plotted scavening percentage against extract concentration (νg/ml).
Determination of the inhibitory activity toward lipid peroxidation
The lipid peroxidation inhibition activity of extract fractions was determined using the ferric thiocyanate method (Hsu et al., 2008[18]). The linoleic acid emulsion was prepared by homogenizing 0.2804 g of linoleic acid, 0.2804 g of Tween-20 as emulsifier and 50 ml 40 mM phosphate buffer (pH 7.0). Reaction mixture was composed of 2.5 ml linoleic acid emulsion and 0.5 ml extract solution prepared in different concentrations. The final volume was adjusted to 5 ml with 40 mM phosphate buffer (pH 7.0) and samples were incubated at 37 oC in the dark. After 72 h of incubation, 4.7 ml of ethanol (75 %) was mixed with 0.1 ml of the reaction mixture and 0.1 ml FeCl2 (20 mM). Finally, 0.1 ml ammonium thiocyanate (30 %) was added to this solution and the absorbance was measured at 500 nm, after it was stirred for 3 min. Inhibition percent of linoleic acid peroxidation was calculated using following formula:
% inhibition = [(AC - AS)/AC] x 100
Measurement of reducing power
In order to determine reducing power of extracts, the method of Oyaizu (1986[47]) was used. Different concentrations of extracts (2.5 ml) prepared in distilled water were mixed with same volume of 0.2 M sodium phosphate buffer (pH 6.6) and 1 % potassium ferricyanide. The mixture was incubated at 50 °C and after 20 min of incubation, reaction mixture acidified with 2.5 ml of 10 % trichloroacetic acid (w/v). Reaction mixture then centrifuged at 1000 rpm for 8 min and the upper layer (5 ml) was mixed with 1 ml of FeCl3 (0.1 %). The absorbance was measured at 700 nm. Increased absorbance indicates a higher reducing activity.
Antigenotoxic potential of chloroform, ethyl acetate and n-butanol fractions
The SLRL test was applied on D. melanogaster available from Bloomington Stock Centre (Indiana, USA). Canton S males and marked balancer Basc females, with inverted X-chromosomes, were used in the experiment. The fruit flies were cultivated on corn flour medium with yeast, agar, sugar and nipagin (Cas No. H5501, Sigma-Aldrich, St. Louis, MO, USA) at 25 °C and 60 % relative humidity in a 12:12 h light/dark regime.
Chloroform, ethyl acetate and n-butanol fractions obtained from G. asclepiadea roots methanolic extract were dissolved in 1 % sucrose and 5 % solutions were used immediately in treatments. EMS (Cas No. 62-50-0, Sigma-Aldrich, St. Louis, MO, USA) in concentration of 0.75 ppm was used as a positive control and as the DNA damage-inducing agent.
In order to evaluate the antigenotoxicity of chloroform, ethyl acetate and n-butanol fractions obtained from G. asclepiadea roots methanolic extract, Canton S males were divided into five groups, with fifteen males in each and starved for 5 h prior to treatments. The first group of males, referred as the negative control, was treated by acute feeding exposure to 1 % sucrose for 24 h (Lewis and Bacher, 1968[40]). The second group, referred as the positive control group, was treated for 24 h with EMS. The third, fourth and fifth groups were treated with EMS for 24 hours before to treatment with single dose of 5 % chloroform, ethyl acetate and n-butanol fractions, respectively.
The SLRL test on D. melanogaster was carried out according to the standard procedure originally described by Würgler and Graf (1985[60]). The frequencies of sex linked recessive lethal mutations are detected by examining males' eye color and shape in the second generation (F2). By taking into account the fact that recessive lethal mutation is expressed before the adult stage, the absence of wild type males in the F2 indicates the occurrence of recessive lethal mutation.
According to test procedure, cells were exposed in successive spermatogenetic stages (three broods), so the effect of chloroform, ethyl acetate and n-butanol fractions on frequency of mutations is assayed at each of the germ-line stages: the first and the second broods (I and II) gives the effect of treated postmeiotic stages, spermatozoids and spermatids, respectively, while the third brood (III) reveals the effect on the premeiotic stage (spermatocytes).
Statistical evaluations
The data are expressed as mean ± standard deviation (SD). The IC50 for in vitro antioxidant potential was calculated using nonlinear regression analysis from the sigmoidal dose-response inhibition curve. Statistical analyses on the data were analyzed using analysis of variance (ANOVA) and the group means were compared by the least significant difference test (LSD). The results were considered statistically significant if the p < 0.05, p < 0.01 and p < 0.001. For SLRL test, by testing for large independent samples, the statistically significance of percentage differences in lethal cultures was calculated, and results are analyzed for difference in proportions (Petz, 1985[48]).
Results and Discussion
Chemical composition of extract fractions
The crude methanolic extract of G. asclepiadea roots was fractionated by sequential extraction with solvents of increasing polarity: chloroform, ethyl acetate and n-butanol. The total phenolics, flavonoids, flavonols and gallotannins contents of various solvents fractions are presented in Table 1(Tab. 1). The total phenolics content of each fraction varied between 55.57 and 146.64 mg/g of extract and ethyl acetate fraction showed the highest total phenolics content. As for total phenolics, flavonoids and flavonols contents also varied depending on the solvent extraction with maximum values of 44.62 and 22.71 mg RUE/g of extract, respectively, in ethyl acetate fraction (Table 1(Tab. 1)). Results showed that all fractions contain low amount of gallotannins (0.34 to 0.99 mg GAE/g) compared with other groups of examined phenolic compounds. The effect of solvent on gallotannins solubility showed approximately the same classification as phenolics. The total phenolics, flavonoids, flavonols and gallotannins contents were found to be significantly higher (p < 0.05) in the ethyl acetate than in the chloroform and n-butanol fractions. The total flavonoids and flavonols contents were significantly higher (p < 0.05) in the n-butanol than in the chloroform fraction, while the amounts of total phenolics and gallotannins did not show significant variations (p > 0.05) among n-butanol and chloroform fractions. The variability in the phenolic contents in different extracts could be the result of the varying solubility of the phenolic compounds; this variation in solubility may be driven by the solvent polarity (Marinova and Yanishlieva, 1997[42]). In the present study, chloroform was less effective for extracting phenolic compounds than the n-butanol, while ethyl acetate extraction is able to extract phenolics, flavonoids, flavonols and gallotannins in relative good amounts.
HPLC-DAD analyses of extracts showed that secoiridoid compounds; sweroside, swertiamarin and gentiopicrine, exist in G. asclepiadea root extracts at different amounts, depending on the solvent used for the extraction (Table 2(Tab. 2)). The results showed that the most abundant secoiridoid compound in the extracts was gentiopicrine, and its amount varied within the range of 14.39 to 442.89 mg/g, depending on the extraction procedure. The sweroside contents in the extracts were 1.52-27.85 mg/g, while the swertiamarin contents ranged from 7.12 to 16.62 mg/g. The n-butanol fraction possessed the highest amount of secoiridoid compounds and its swertiamarin, sweroside and gentiopicrine contents were found to be 16.62, 27.85, and 442.89 mg/g of extract, respectively. The lowest amounts of sweroside and gentiopicrine were found in the chloroform fraction, while no significant difference (p > 0.05) in concentrations of swertiamarin between extracts. In root fractions of G. asclepiadea, mangiferin was not present in detectable amounts. Quantitative data obtained for G. asclepiadea root in the present study are in good agreement with previously published data suggesting that gentiopicrine is the main secoiridoid compound in this plant (Dević et al., 2006[10]; Szücs et al., 2002[56]).
Amounts of the total secoiridoid glycosides and total phenolics, flavonoids, flavonols, and gallotannins investigated in the underground parts of G. asclepiadea, expressed in percents of fractions are shown in Figure 1(Fig. 1). It is obvious that n-butanol and ethyl acetate fractions contained significant higher (p < 0.001) concentration of secoiridoids than phenolic compounds while chloroform fraction contained significant higher (p < 0.001) concentration of total phenolics than secoiridoid compounds. The highest amount of secoiridoids was present in n-butanol fraction (48.74 %) while the chloroform fraction contained all examined compounds in significantly lower concentration compared with n-butanol and ethyl acetate fractions. That was also confirmed in other studies where the highest amount of secoiridoids was extracted with n-butanol (Krstić et al, 2004[36]; Janković et al., 2005[24]).
Total antioxidant capacity
The results for total antioxidant capacity are given in Table 3(Tab. 3). Ethyl acetate fraction had a significantly higher (p < 0.05) capacity (401.33 mg AA/g) than the other fractions. The total antioxidant activity of the plant extracts are in the order: ethyl acetate fraction > n-buthanol fraction > chloroform fraction. The dependence of total antioxidant capacity in relation to the total phenolics, flavonoids, flavonols and gallotannins content, was also evaluated. The strong linear relationship of the total antioxidant capacity results and total phenolics and gallotannins contents were observed (R2 = 0.9322 and R2 = 0.9996), suggesting that total phenolics and gallotannins were likely to be contributing to the total antioxidant capacity of these extracts. The total flavonoids and flavonols contents of the extracts had a less linear correlation with their total antioxidant capacity (R2 = 0.5573 and R2 = 0.5750, respectively), while no correlation or insignificant relationship between the secoiridoids and total antioxidant capacity of the extracts (R2 < 0.5).
DPPH radical scavenging activity
DPPH assay was employed in our study to determine the proton-radical-scavenging activity based on the reaction that the purple color of DPPH solution fades quickly when it reacts with proton-radical scavengers. BHT, ascorbic acid and gallic acid are the reagents used as standards. The results of DPPH radical scavenging activities of the chloroform, ethyl acetate and n-butanol fractions obtained from the G. asclepiadea root extract, along with the reference standards, are shown in Table 3(Tab. 3). All examined extracts exhibited considerable levels of free radical scavenging activity in the DPPH assay, indicated by IC50, i.e., the concentration at which the compound reduces 50 % of the DPPH.
The most active radical scavengers were ethyl acetate fraction, with IC50 of 107.28 µg/ml, while chloroform fraction was considerably less effective radical scavengers. Gallic acid showed the greatest radical scavenging activity (IC50 = 3.79 µg/ ml) among examined referent antioxidants, its IC50 value was approximately 30 times lower than the concentration of ethyl acetate fraction to reduce 50 % of DPPH starting concentration. Based on the results of DPPH assay, the extract performing the highest antioxidant activity had the highest concentration of total phenolics, flavonoids, flavonols and gallotannins. Excellent correlation of IC50 values of DPPH scavenging activity to the total flavonols (R2 = 0.9830) and flavonoids (R2 = 0.9781) content was noticed, while IC50 values correlated to the total phenolics and gallotannins content with R2 = 0.9035 and R2 = 0.7189, respectively.
IC50 values were determined by nonlinear regression analysis. Results are mean values ± SD from three independent experiments; AA, ascorbic acid; -, Not tested. Means in the same column with superscript with different letters are significantly different at p < 0.05
Inhibitory activity toward lipid peroxidation
The inhibitory activity of the extracts against lipid peroxidation was determined by the thiocyanate method, and the results are given in Table 3(Tab. 3). The ferric thiocyanate method measures the amount of peroxide produced during the initial stages of oxidation, which is the primary product of lipid oxidation. In this assay, indirect measurement was performed for the amount of hydroperoxides produced from linoleic acid emulsion by auto-oxidation during the experiment period (Inatani et al., 1983[23]). All tested extracts added to linoleic acid emulsion were able to reduce the formation of hydroperoxide. The IC50 values of the G. asclepiadea extracts ranged from 40.96 to 183.16 μg/ml. Ethyl acetate fraction has been found as the highest effective (p < 0.05) among the G. asclepiadea extracts, with IC50 value of 40.96 μg/ml. In the linoleic acid system (Table 3(Tab. 3)), referent compounds α-tocopherol (IC50 = 0.48 µg/ml) and BHT (IC50 = 1 µg/ml) exhibited much lower IC50 values than examined plant extracts. In comparison, ethyl acetate, n-butanol and chloroform fractions showed higher activity than referent compounds gallic acid and ascorbic acid.These results together confirm that the polar antioxidants are less effective in protecting the emulsified linoleic acid, whereas lipophilic antioxidants due to higher partition into the lipid phase reveal greater activity in the emulsion (Terpinc et al., 2012[58]). The results show that the amounts of total phenols, flavonoids and flavonols correlated strongly IC50 values obtained in this method, with linear correlation coefficients R2 = 0.9126, 0.9732, and 0.9787, respectively, which somewhat better than correlation with gallotannins content (R2 = 0.7330).
Reducing power
Reducing power reflects the electron donating capacity of bioactive compounds and is associated with antioxidant activity. Antioxidants can be reductants and inactivate oxidants (Gülçin, 2009[15]). The reducing capacity of extracts and referent compounds was measured by the direct reduction of [Fe3+(CN)6]3- to [Fe2+(CN)6]4-. Reduced product reacts with FeCl3 and forms the intense Perl's Prussian blue complex, Fe4[Fe(CN)6]3, which has a strong absorbance at 700 nm.
An increase in absorbance of the reaction mixture would indicate an increase in the reducing capacity due to an increase in the formation of the complex. Figure 2(Fig. 2) shows the reductive capability of fractions obtained from methanol root extract of G. asclepiadea, compared to ascorbic acid, gallic acid and BHT. The results indicate that ethyl acetate extract exhibited a greater reduction power than the chloroform and n-butanol extract. As in previous described antioxidant studies, in this work, strong reducing power of the ethyl acetate extract was expected, since it had a high content of phenolic compound.
In the present study, a nonsignificant correlation was found between antioxidant activities and sweroside, swertiamarin and gentiopicrine contents in G. asclepiadea root extracts, R2 values were less than 0.5. Results showed that the polyphenol-enriched fraction (ethyl acetate fraction) possessed higher antioxidant activities than any other fractions. Therefore, total phenolics, flavonoids, flavonols and gallotannins, appear to be the major contributors to the high antioxidant activities.
Antigenotoxic potential of chloroform, ethyl acetate and n-butanol fractions
The use of plant extracts to cure various diseases is very common in traditional medicine. Although a large number of plant species is a potential source of biologically active compounds, the effect on genetic material is often unknown. In this study, the efficacy of three different fractions of G. asclepiadea root extract against DNA damage induced by EMS was tested. The aim was to investigate new potential antigenotoxic agents from natural sources, for possible use to protect the integrity of DNA from various genotoxic agents.
As can be seen from Table 4(Tab. 4), EMS caused statistically significant increase in frequency of lethal mutations in comparison to the sucrose, observed as an increase in the percentage of lethal (33.63 %). Although EMS increased the frequency of mutation, chloroform fraction from G. asclepiadea roots decreased the genotoxicity of EMS (Table 5(Tab. 5)). Ethyl acetate fraction at concentration of 5 % statistically significantly reduced the frequency of mutations induced by EMS in all three broods. Post-treatment with n-butanol fraction from G. asclepiadea drastically reduced the frequency of mutations induced by EMS with high significance in all broods (p < 0.001***).
Compared to the negative control all fractions decreased the genotoxicity of EMS in postmeiotic germinative cell lines - at spermatozoids and spermatids and in premeiotic line - spermatocytes (Table 6(Tab. 6)). The average values in frequencies of mutations for I, II and III broods indicate that all fractions in concentration of 5 % exhibit antigenotoxic effect, as they significantly reduces the rate of mutation compared to EMS. Results analyzed by simultaneous comparison between three fractions from G. asclepiadea revealed no statistically significant difference in the protective potential of the extract fractions (Table 7(Tab. 7)).
According to Hung et al. (2009[22]) and Kuroda et al. (2001[38]) there are two main groups of protective mechanisms for DNA: desmutagenesis, in which tested substances act on mutagens or inactivate them, and the other is bio-antimutagenesis in which tested substances act on the processes of mutagenesis or repair DNA damages that result in a decrease in the frequency of mutation. Desmutagens could be detected with pre-treatment, while bio-antimutagenicity could be better detected with post-treatment (De Flora, 1998[8]). Considering the genotoxin administered with tested fractions, there is a possibility that the antimutagens in fractions exert protective effect in bio-antimutagenic manner, which suppresses mutation fixation after DNA is damaged by EMS.
Mutagenic action of alkylating agents may be related to their ability to form O6-methylguanine (O6MeG) (Stephanou et al., 1996[55]). O6MeG is repaired by protein termed O6-methylguanine DNA methyltransferase (MGMT) (Margison and Santibáñez-Koref, 2002[41]), by transferring ethyl group from a base lesion to a cysteine residue, thus restoring the integrity of the DNA without creating additional DNA damage (Kaina and Christmann, 2002[29]). This means that fractions from G. asclepiadea have the ability of anti-alkylation damage and improves the activity of MGMT. This conclusion is supported by De Flora et al. (2005[9]) who reported that the bioantimutagenic agents reverted the mutagenic effects and preventing the fixation of mutations acting on the physiological mechanisms of DNA protection and repair.
Potential genotoxicity and/or antigenotoxicity of the methanolic and aqueous extracts from the haulm and flower of G. asclepiadea were investigated previously in human lymphocytes, human embryonic kidney (HEK 293), and monkey kidney (COS 1) cell lines using the comet assay (Hudecová et al., 2010[21]; Hudecová et al., 2012[20], 2012[19]). In those studies, extracts of G. asclepiadea had neither genotoxic nor cytotoxic effect in a wide range of concentrations. Likewise, our previous study showed that in vivo exposure to the methanol extracts of aerial parts and roots of G. asclepiadea did not induce DNA damage in liver cells of Wistar rats (Mihailović et al., 2013[44]).
The antigenotoxic effect can be attributed to a wide range of phytochemicals including simple phenols, flavonoids and flavonols (Aaronson, 2000[1]; Siriwardhana et al., 2003[54]; Karawita et al., 2005[31]), detected in extracts of G. asclepiadea. Previous studies prove the ability of phenolics and flavonoids in scavenging mutagens or free radicals (Rice-Evans et al., 1996[50]; Yao et al., 2004[61]). Also, the antigenotoxic activity of some flavonoids was induced by radical scavenging activity (Edenharder and Grunhage, 2003[11]). Fedeli et al. (2004[13]) reported that tannins are able to protect DNA against damage in low concentrations, while at high concentrations they could be genotoxic. To the best of our knowledge, there are no available literature data about the antimutagenic effect of the secoiridoids. Regarding the fact that chloroform fraction contains lower amounts of phenolic compounds and secoiridoids in relation to the other two fractions (Figure 1(Fig. 1)), and that they exhibit the same level of antigenotoxicity (Table 7(Tab. 7)), it cannot be conclude with certainty which of the above mentioned compounds are responsible for the protective effect. It can be assumed that the presence of all these components in the extracts cumulatively may have been responsible for reduction of DNA binding and ethylation with EMS.
Conclusions
The present study demonstrates that different extract fractions obtained from the root of G. asclepiadea possessed antioxidant, radical-scavenging and antigenotoxic activities. The obtained results indicate that the ethyl acetate fraction shows a stronger antioxidant activity than other solvent fractions. It seems the reason for this difference just is that the phenolic content in ethyl acetate fraction is higher than in other fractions. Regarding the fact that n-butanol fraction contains the highest amount of secoiridoids compounds in relation to the other two fractions and that the ethyl acetate fraction exhibit stronger antioxidant activity, it can be concluded that phenolics are main compounds responsible for the antioxidant effects. Experiments performed in D. melanogaster showed an antigenotoxic effect of three different extract fractions of G. asclepiadea by post-treatment. These results suggested that extracts act intracellulary by affect the DNA repair damage induced by EMS in a bio-antimutagenic manner.
Acknowledgements
This work was supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia (projects No. III 43004 and 41010).
References
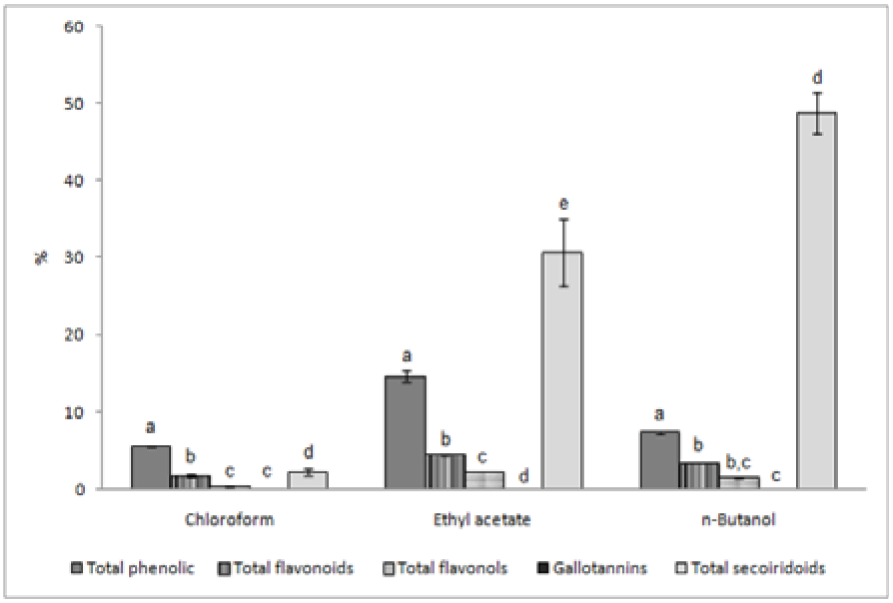
Figure 1: The content of total secoiridoid glycosides, phenolics, flavonoids, flavonols and gallotannins in roots of G. asclepiadea expressed in % of dry extracts (mean values of three independent measurements with error bars representing SD); Means for same solvent fraction with different letters are significantly different at p < 0.001
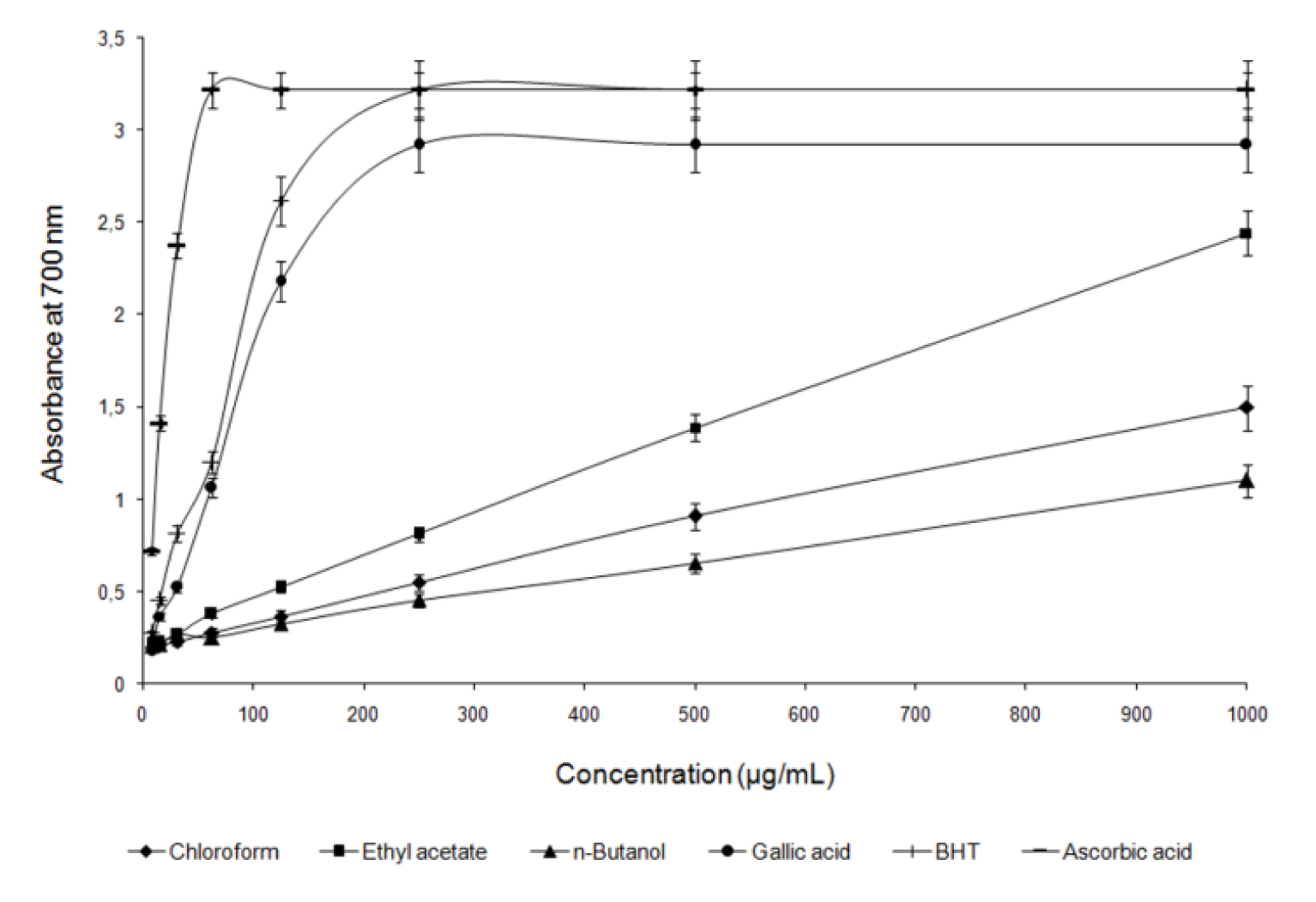
Figure 2: Reducing power of extract fractions obtained from G. asclepiadea roots compared to reducing power of standards at different concentrations. Each value is the average of three independent measurements with error bars representing SD.
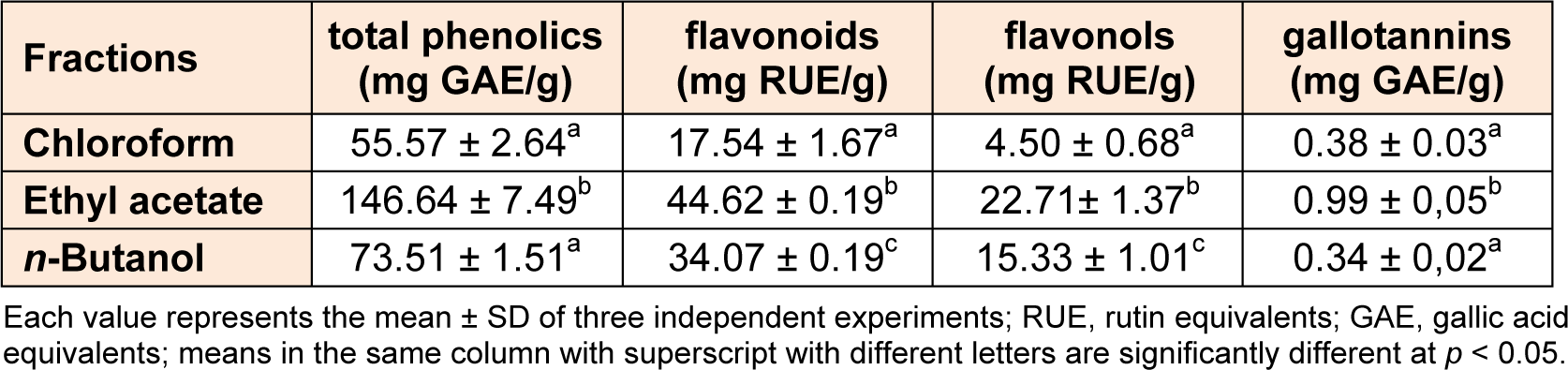
Table 1: Total phenolics, flavonoids, flavonols and gallotannins content of chloroform, ethyl acetate and n-butanol fractions from Gentiana asclepiadea roots
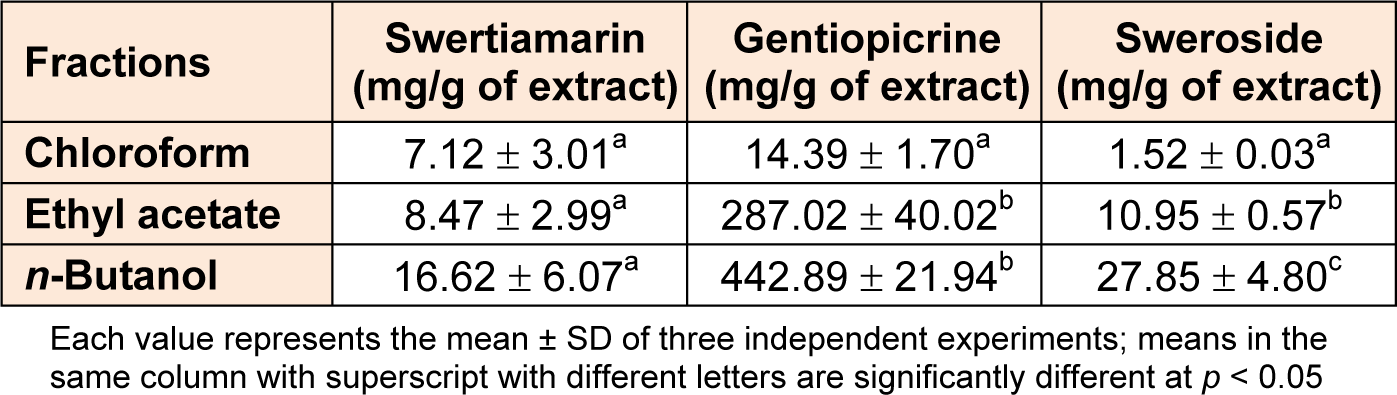
Table 2: HPLC analysis of secoiridoid compounds contents in chloroform, ethyl acetate and n-butanol fractions from Gentiana asclepiadea root extract
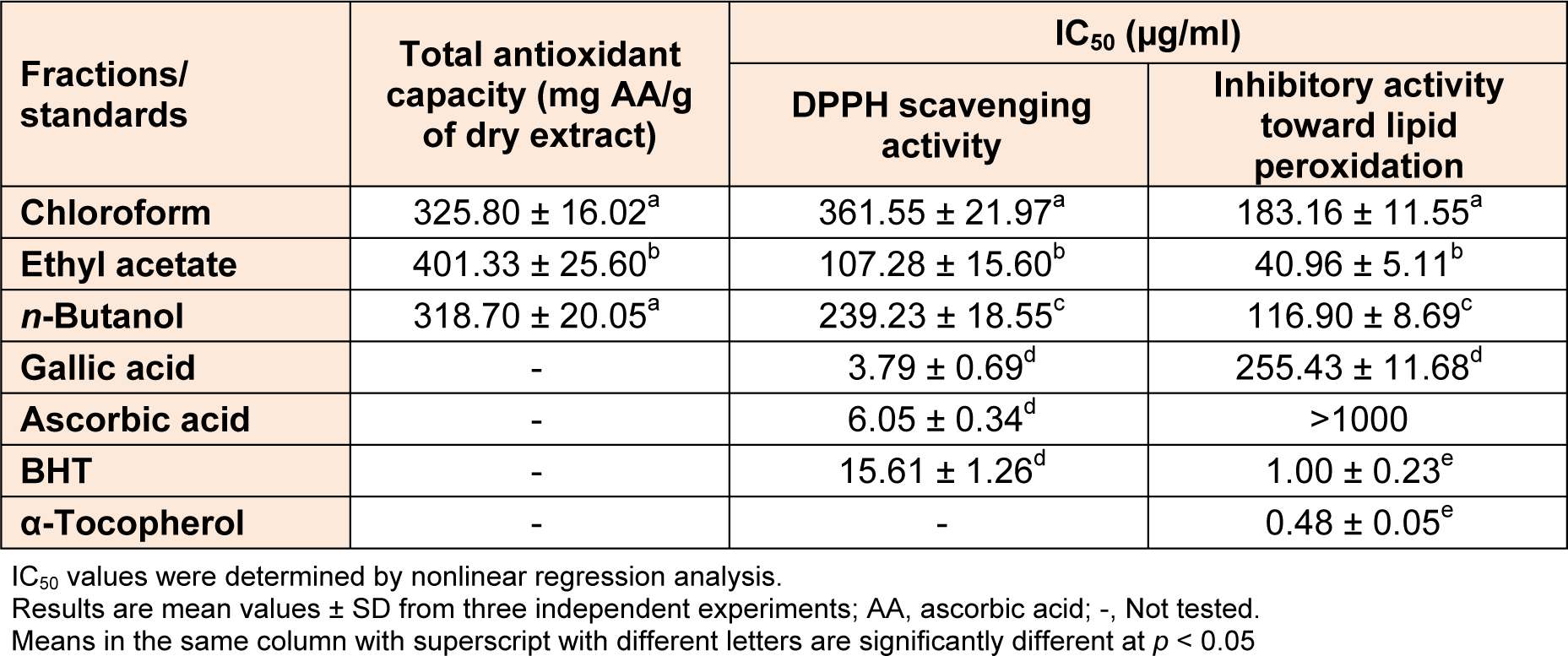
Table 3: Antioxidative activity of chloroform, ethyl acetate and n-butanol fractions from Gentiana asclepiadea root extract
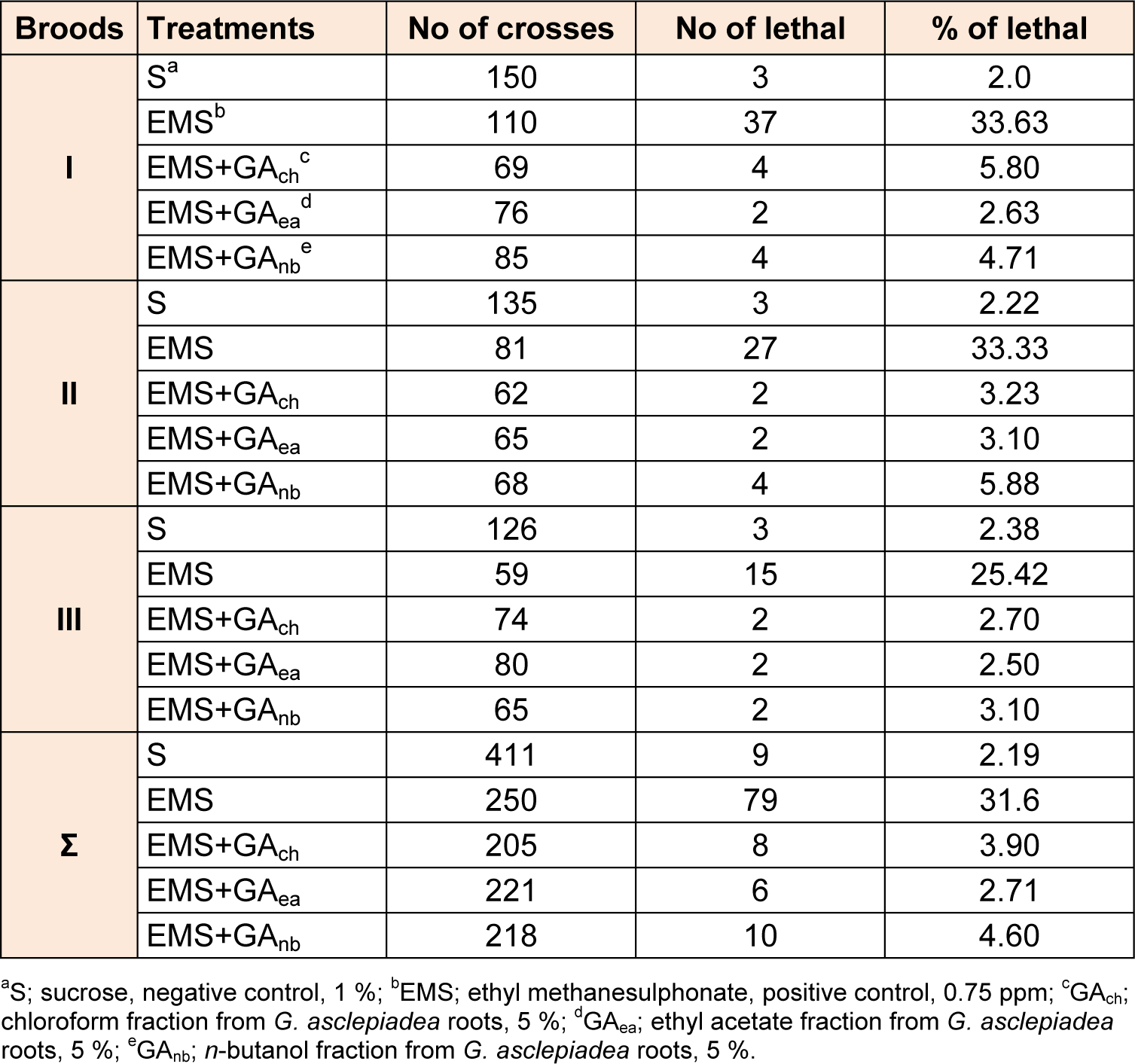
Table 4: Frequencies of sex linked recessive lethal mutations in the Drosophila melanogaster test after post-treatments with EMS and G. asclepiadea roots extract fractions
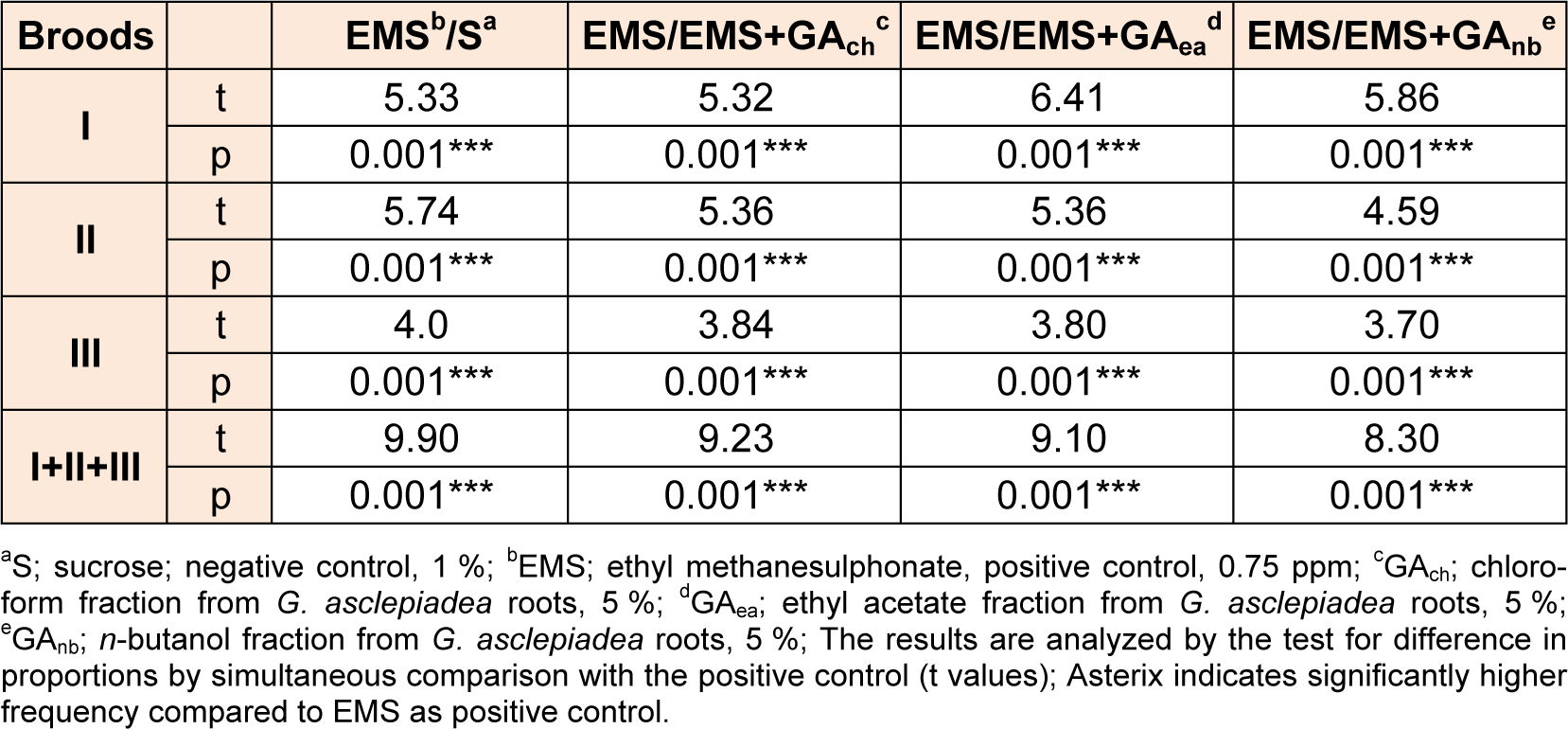
Table 5: The significance of differences in the frequency of recessive lethal X-linked mutations in Drosophila melanogaster males after post-treatments with G. asclepiadea roots extract fractionscompared to the positive control
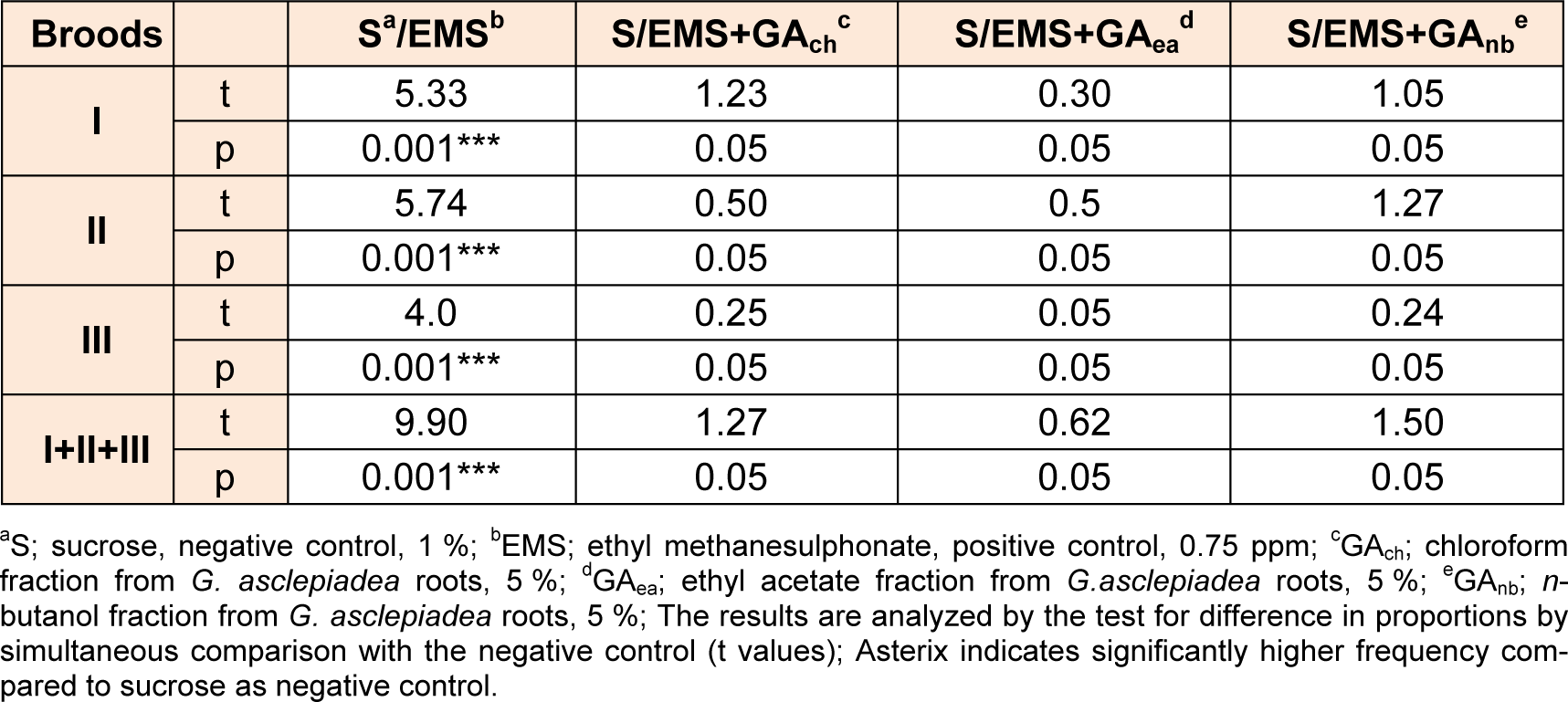
Table 6: The significance of differences in the frequency of sex-linked recessive lethal mutations of Drosophila melanogaster males after post-treatments with G. asclepiadea roots extract fractions compared to the negative control
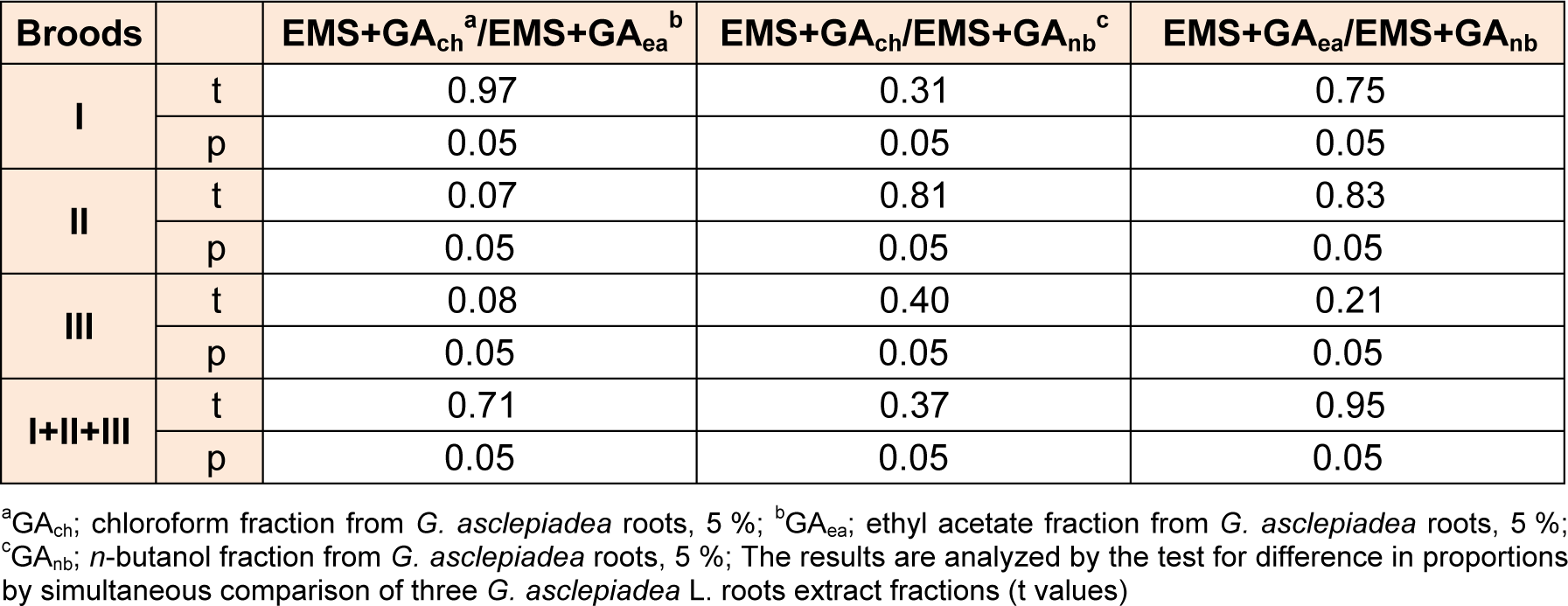
Table 7: A comparison in the significance of differences in the frequency of sex-linked recessive lethal mutations of Drosophila melanogaster males after post-treatments with three G. asclepiadea roots extract fractions
[*] Corresponding Author:
Vladimir Mihailovic, Department of Chemistry, Faculty of Science, University of Kragujevac, Radoja Domanovica 12, 34000 Kragujevac, Serbia; Tel.: +381 34 336 223; Fax: +381 34 335 040, eMail: vladam@kg.ac.rs