Research article
Sorption of DNA by diatomite-Zn(II) embedded supermacroporous monolithic p(HEMA) cryogels
Kabil Özcan Tozak1, Mahmut Erzengin1[*], Idris Sargin1, Nuri Ünlü1
1Aksaray University, Faculty of Art and Science, Chemistry Department, 68100 Aksaray, TurkeyEXCLI J 2013;12:Doc670
Abstract
In this study, the DNA sorption performance of diatomite-Zn(II) embedded supermacroporous monolithic p(HEMA) cryogels were investigated for the purpose of designing a novel adsorbent that can be utilized for DNA purification, separation and immunoadsorption studies such as removal of anti-dsDNA antibodies from systemic lupus erythematosus (SLE) patient plasma. Poly(2-hydroxyethyl methacrylate) [p(HEMA)]-based monolithic cryogel column embedded with Zn2+-diatomite particles was prepared by free radical cryo-copolymerization of 2-hydroxyethyl methacrylate (HEMA) with N,N'-methylene-bis-acrylamide (MBAAm). The polymerization reaction was initiated by N,N,N',N'-tetramethylene diamine (TEMED) and ammonium persulfate (APS) pair in an ice bath. After thawing, the monolithic composite cryogels were used for affinity sorption and then subsequent desorption of DNA molecules from aqueous solutions. Diatomite (DA) particles were characterized by XRF and BET method. The characterization of composite cryogel was done through SEM imaging. The effects of pH of the solution, initial DNA concentration, ionic strength, temperature and flow rates on adsorption were investigated to determine the optimum conditions for adsorption/desorption experiments. The particle embedding procedure was shown to yield significantly enhanced adsorption of DNA on the adsorbent. Furthermore, considering its excellent bio-compatibility, p(HEMA) cryogels are promising a candidate for further DNA sorption studies.
Keywords: diatomite, particle embedding, composite cryogel, immobilized metal affinity chromatography
Introduction
Diatoms, which are aquatic microscopic plants related to algae, extract silica from natural surface water to form an outer skeletal structure (Subramanyam and Roesli, 2000[19]). Diatomite, consisting primarily of amorphous SiO2, is formed from accumulation of siliceous crusts of these unicellular plants. Due to its high porosity, good hydrophilicity, high chemical inertness, low cost, and environmental friendly nature, diatomite is widely used as filtering material, adsorbent and catalyst carrier (Wu et al., 2011[26]). Active adsorption sites on diatomite surface are tought to be as following: (i) insulated free silanol groups (-SiOH), (ii) free dual silanol group (-Si(OH)2), (iii) -Si-O-Si bridges with oxygen atoms on the surface. Silanol groups have a tendency to react with many polar organic compounds with various functional groups (Zhuravlev, 2000[30]; Caliskan et al., 2011[1]).
In recent years, there have been studies to remove anti-dsDNA antibodies in the body fluids of patients suffering from autoimmune diseases, characterized by an abnormal immune response directed against the host's own tissues. Extracorporeal immunoadsorption has been widely utilized as a therapeutic modality to remove pathogenic antibodies containing from plasma of patients. Systemic lupus erythematosus (SLE), commonly known as lupus, is an autoimmune disease system characterized by dysregulation in multiple arms of the immune system and the development of anti-nuclear antibodies, inducing complement activation, inflammation and tissue damage (Tan, 1989[20]; Jeoung et al., 2004[5]).
Immunoadsorption has become available and also is appeared to be a relatively safe effective therapeutic option in recent years (Stummvoll et al., 2009[18]). There have been various attempts for development of DNA-attached adsorbents for immunoadsorption application. Some workers developed and utilized various immunoadsorbents for antibody removal; DNA-immobilized activated carbon hemoperfusion column (Nicolaev et al., 1992[11]), poly(ethylene vinyl alcohol) hollow fiber carrying histidine (Ventura et al., 2001[25]), DNA-immobilized non-woven poly (ethylene terephthalate) fabric fibers (Zhu et al., 1999[29]), DNA-immobilized hydroxy-ethyl cellulose crosslinked chitosan beads (Yu and He, 1999[28]), DNA-carrying poly (ethylene terephthalate) microfibers (Kato and Ikada, 1996[6]), Cu2+-attached magnetite nanoparticles embedded supermacroporous monolithic composite cryogels (Ceylan et al., 2011[2]), poly(hydroxyethyl methacrylate) based magnetic DNA-affinity beads (Odabaşı and Denizli, 2001[12]), poly(hydroxyethyl methacrylate) based affinity membranes (Uzun et al., 2010[24]).
Our research group has designed particle-embedded composite cryogel columns for separation and purification of biomolecules. Previously, we produced Cu2+-attached sporopollenin embeded cryogels for human serum albumin (HSA) adsorption (Erzengin et al., 2011[3]), Cu2+-attached magnetite nanoparticles embedded supermacroporous monolithic composite cryogels for DNA adsorption (Ceylan et al., 2011[2]), Ni2+-attached diatomite particles embedded in composite monolithic cryogels for HSA (Ünlü et al., 2011[23]).
In this study, we prepared Zn2+-embedded p(HEMA) based composite cryogels for removal of anti-DNA antibodies. Considering the high sorption capacity and biocompatibility of p(HEMA) based adsorption columns, the composite cryogels we obtained are emerging as good candidate for achieving higher removal of anti-DNA antibodies for immunoadsorption studies.
Materials and Methods
Materials
Hydroxyethyl methacrylate (HEMA) was purchased from Fluka A.G. (Buchs, Switzerland), distilled under reduced pressure in the presence of hydroquinone inhibitor, and kept sealed at 4 °C until use. N,N'-methylene-bis-acrylamide (MBAAm), ammonium persulfate (APS) DNA (from herring testes) were obtained from Sigma (St. Louis, MO, USA). All other chemicals were of reagent grade and were purchased from Merck AG (Darmstadt, Germany). N,N,N',N'-tetramethylethylenediamine (TEMED) were obtained from FlukaA.G. (Buchs, Switzerland). The natural DA of Ihlara-Selime (Aksaray, Turkey) origin was used. Water used in the experiments was purified using a Barnstead (Dubuque, IA, USA) ROpure LP® reverse osmosis unit with a high flow cellulose acetate membrane (Barnstead D2731), followed by a Barnstead D3804 NANOpure® organic/ colloid removal and ion exchange packed bed system.
Preparation of diatomite particles (DAPs)
The natural DA particles were rinsed thoroughly with double-distilled water to get rid of dirt and any other substances. The washed DA particles were sedimented to obtain deposits of about 2 µm. Subsequently, the particles obtained through sedimentation treatment were heated at 130 °C for 5 h to remove any substance of bio-organic origin to attain larger pore size and surface area on the particles.
Attachment of Zn2+ ions to diatomite particles (DAPs- Zn2+)
Zn2+ solution of 100 mg/L concentration was prepared by dissolving 454.95 g of Zn(NO3)2.6H2O salt in deionized water. The pH of the Zn2+ solution was adjusted to 5.0 with 0.1 M HCl and NaOH solutions. Then, 10 mg of DA was treated with 10 mL of Zn2+ solution at room temperature for 3 h. The Zn2+-DA particles were filtered and rinsed several times with deionized water. Zn2+ ion concentration in initial and final solutions was determined with an atomic absorption spectrometer (GFAAS, Analyst 800/PerkinElmer, USA). Any possible occurrence of metal leakage from Zn2+-DAPs was investigated in 1.0 M NaCl solution at pH values, ranging from 5.0 to 8.0. Zn2+-DAPs were agitated for 24 h at room temperature. Then, Zn2+ ion concentration in the supernatant was detected using an atomic absorption spectrometer (GFAAS, Analyst 800/PerkinElmer, USA). Zn2+-DAPs were dried and kept in an air-tight container.
Preparation of p(HEMA)- DAPs -Zn2+ cryogels
The monolithic composite cryogels were produced by free radical polymerization as follows: The gels were produced in plastic syringe (5 mL, id 0.8 cm) in an ice bath. Firstly, 70 mg N,N'-methylene-bis-acrylamide and 370 µL 2-hydroxyethyl methacrylate were dissolved in deionized water (5 mL) and the mixture was degassed under vacuum for about 5 min to eliminate soluble oxygen. Then, APS (100 µL) (10 % w/v) was added to the polymerization medium. The mixture was kept in an ice bath for 5 min. Following this step, TEMED (20 µL) was added and the reaction mixture was stirred for 1 min. Finally, 10 mg of Zn2+-DAPs was added to the polymerization mixture. The polymerization solution in the syringes was frozen at - 12 °C for 24 h and then thawed at room temperature. To eliminate any remaining unreacted monomers, activator and initiator, a washing solution (i.e. a dilute HCl solution and a water-ethanol mixture) was run through the composite cryogels until purity of the gels was ensured. Purity of the composite cryogel was agreed by observing the change of optical densities of the samples taken from the liquid phase in the circulation system. After washing, the composite cryogel was stored in buffer containing 0.02 % sodium azide at 4 °C until use.
Characterization of cryogel: The porosity of p(HEMA)-DAPs-Zn2+ composite cryogel column
Measurement of volume and free water content of the cryogel samples were done as follows: To estimate porosity, φ value, a piece of cryogel sample was saturated with deionized water, and it was subsequently immersed in water in a vial having volume V1, then, the total volume of the vial was marked as volume V2. Water-saturated cryogel volume, V0, was estimated by the volume difference, i.e.,

The mass of water-saturated cryogel, mw, was weighted out. The cryogel sample was squeezed to rid of the free water within the macropores of the gel. The mass of the cryogel sample without free water, ms, was weighted as described previously (Yao et al., 2006[27]). The porosity was calculated by the following formula:
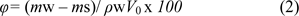
where ρw is the density of deionized water. Then the cryogel sample was dried in the oven at 60 ◦C for 12-24 h to a constant and the dried cryogel mass, md, was determined, and the total water content (TWC) was calculated by the following formula:
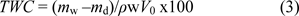
Characterization of cryogel: Surface morphology
The morphology of a cross section of the dried cryogel was coated with gold-palladium (40:60) and investigated by JEOL JSM 5600 SEM (Tokyo, Japan).
DNA adsorption from aqueous solutions
A continuous system equipped with a water jacket for temperature control was employed to carry out adsorption and desorption studies of DNA. Prior to each adsorption study, the cryogel column was first washed with deionized water (30 mL) and then equilibrated with 0.02 M Tris-HCI buffer containing 0.02 M NaCl at pH 6.0. Then DNA solution (i.e., 20 mL) was pumped through the column (volume of 5 mL). The fractions were assayed by UV spectrophotometry at 260 nm. The effects of DNA concentration (in the range of 0.1- 4.0 mg/mL), medium pH (i.e., 5 - 8), flow rate (i.e., 1 - 4 mL/min), ionic strength (i.e., 0.025 - 0.2 M NaCl) and temperature (i.e., 5, 15, 25 and 35 °C) on adsorption capacity were studied. Following each DNA adsorption study, desorption of the adsorbed DNA molecules was performed as described below: 30 mL of desorption agent (i.e., 0.1 M phosphate buffer containing 0.75 M NaCl at pH 8.) was run through the monolithic column at a flow rate of 1.0 mL/min. The final DNA concentrationin the desorption medium was spectroscopically determined. Upon achieving desorption, the cryogel was cleaned with 1M NaOH and then re-equilibrated with 0.02 M Tris-HCI buffer containing 0.02 M NaCl (pH 6.0). The ratio of desorption was calculated from the amount of DNA adsorbed on the cryogel and the final DNA concentration in the desorption medium. To test the reusability of the gels, DNA adsorption/desorption cycle was repeated for 25 times using the same cryogel column. To regenerate and sterilize, following desorption procedure, the cryogel was washed with 1 M NaOH solution.
Results and Discussion
Characterization studies
The natural DA samples were used without any chemical treatment for the Zn2+ attachment. Quantitative chemical analysis of diatomite obtained by X-ray fluorescence technique (Thermo Scientific, ARL 9900 XRF) revealed that it consists primarily of amorphous silica SiO2 (84.07 %) and alumina Al2O3 (5.45 %). The specific surface area of DA measured by BET-method is 65.3 m2/g.
No leakage of immobilized Zn2+ ions was detected from p(HEMA)-DAPs-Zn2+ monolithic cryogels. Apparently, immobilized metal ions are stable toward exposure to Tris-HCI and phosphate buffer between pH 5.0 and 8.0 during the both adsorption and elution procedure. Adsorbed DNA molecules could thus be eluted conveniently by using Tris-HCI containing buffer to give reproducible results. Moreover, this made it possible to use the p(HEMA)-DAPs-Zn2+ monolithic cryogel columns for repeated runs without the need for regeneration and recharging of the columns with fresh Zn2+ ions after each use.
The SEM image of a cross section of a p(HEMA)-DAPs-Zn2+- monolithic cryogel is shown in (Figure 1(Fig. 1)). This porous support contained interconnected macropores. Voids, i.e., porous structures in polymer networks are known to be formed as a result of a phase separation during the course of the copolymerization process. The pores have the major and minor diameters of approximately 50 and 20 nm, respectively and thin polymer walls, thus providing channels for the mobile phase to flow through. As seen in the (Figure 1(Fig. 1)), Zn2+-DAPs were uniformly distributed into the p(HEMA) cryogel network. Pore size of the matrix is large enough to allow DNA molecules to pass easily. Therefore, mass transfer resistance is practically negligible.
The porosity, ρ, and the total water content, TWC, of the cryogel column embedded with Zn2+-DAPs were measured with deionized water as 68.4 and 91.3 % v/v, respectively. These results indicate that about 22.9 % of the total water was bound by the small pores of polymer matrix where almost no flowing liquid passed through. However, the large pores constituting 68.4 % of the total pores were filled with free water and most of the fluid flow took place within these large pores.
The pressure drop of p(HEMA) composite cryogel column should be as low as possible for sample loading. Pressure drop experiments were carried out under water equilibration with linear flow rates from 20 to 300 cm/h. p(HEMA) composite cryogel column which has interconnected supermacropores had very low liquid flow resistance and low back pressure because of large and highly porous interconnected structure.
DNA adsorption to p(HEMA)-DAPs-Zn2+ composite cryogels in aqueous solution
The surface functional hydroxyl groups of silica and alumina are the major binding sites for incorporation of metal ions to the DAPs. Surface charges of diatomite may form by ionization of functional groups such as oxides and hydroxides. The surface is charged through [AlOH2+ / SiOH2+] and [AlO− / SiO−] ionizations. The protonation / deprotonation of hydroxyl groups along the surface of diatomite can result in negative charge depending on the pH of the solution. Caliskan et al. (2011[1]) reported that the pHpzc obtained was ∼3.70 for diatomite, thus diatomite surface may be negatively charged, providing adsorption sites for Zn2+ ions. They also stated that diatomite surface has positive charge below pHpzc and negative charge above pHpzc. Increasing the pH of the clay-aqueous system leads to form more negatively charged sites on diatomite, enhancing sorption of positively charged metal cations. Adsorption of DNA to diatomite surface is significantly higher in the presence of Zn2+ when compared to the p(HEMA)-DAPs.
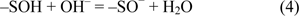

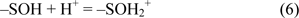
where -SOH and -SO− stand for surface hydroxyl groups and surface oxides respectively. The amount of chelated Zn2+ ions on DAPs was measured as 1.61 mg/g DA.
Here, to explain the process allowing the adsorption of DNA on the diatomite surface it can be referred to a ''salt bridge'' effect between the negatively charged diatomite surface and the negatively charged DNA that is mediated by the divalent cations (Shao et al., 1996[15]). Pretreatment of diatomite with a transition metal cation (Zn2+) appears to enhance the DNA attraction and reduces the repulsive contribution stemming from negatively charged sites.
The chelated metals (Zn2+) interaction with bases in DNA molecule could play a lesser role in DNA binding to the Zn2+-DAPs. Considering the in accessibility of aromatic nitrogen bases in DNA to the immobilized Zn2+ ions, the affinity interaction of chelated metals (Zn2+) with nucleic acid bases could be hindered (Sousa et al., 2008[16]). Murphy et al. (2003[9]) reported low IMAC binding affinity for pDNA and gDNA, single stranded nucleic acids, in contrast, showed higher chelated metal affinity. It was also reported that the preference of affinity interaction with immobilized ion metals followed the order: endotoxins>RNA>pDNA (Tan et al., 2007[21]; Sousa et al., 2008[16]).
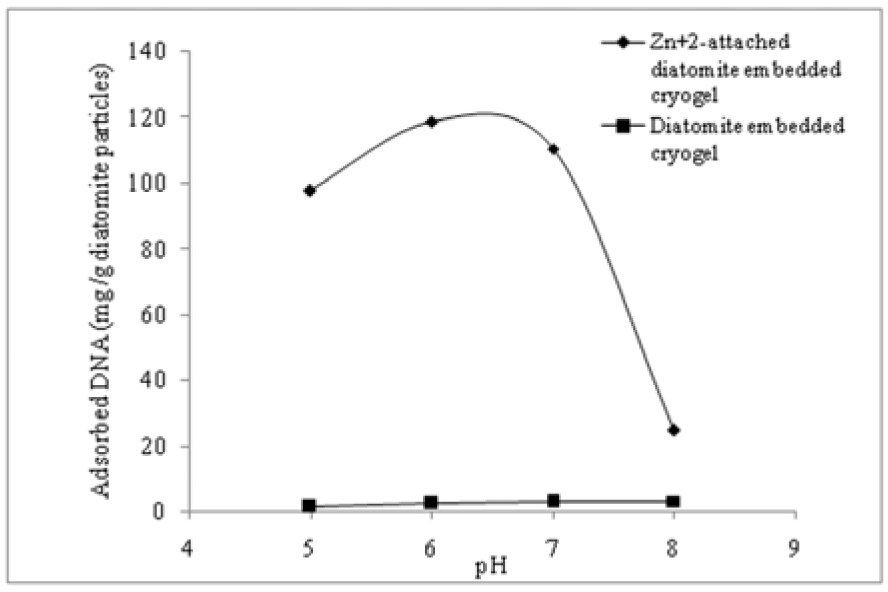
Effect of pH on DNA adsorption
DNA adsorption onto the p(HEMA)-DAPs and the p(HEMA)-DAPs-Zn2+ monolithic cryogels as a function of pH is presented in Figure 2(Fig. 2). Figure 2(Fig. 2) indicates an increase from 5.0 to 6.0 and a decrease from 6.0 to 8.0. The optimal pH for DNA adsorption on p(HEMA)-DAPs-Zn2+ supermacroporous monolithic composite cryogel is around 6.0. The DNA adsorption mechanism is mainly through chelation between metal ions and the phosphate backbone or the DNA bases and also direct bonding of phosphate group at the end of the DNA molecule to the OH- groups of Al-oxide layer (Hansma and Laney, 1996[4]). The isoelectric point of DNA is around pH 5 (Stotzky, 2000[17]), which can be stated here to explain lower DNA adsorption at pH 5.0. Below pH 5.0, the bases can accept protons which create positive charged sites in the DNA molecule. If the pH is above 5.0, DNA is overall negatively charged and can be considered as a polyanion (Trevors, 1996[22]).
It appears that increasing pH of the DNA solution to more than 5 could lead SiOH and AlOH groups on the surface of diatomite particles to be deprotonated to SiO− and AlO−. Concomitantly, the negative charges on DNA molecules could also increase with pH. Furthermore, it can be stated here that this pH dependence likely causes release of protons by bases of DNA molecule, thus lowering positively charged sites on DNA molecule. Regarding these factors, it can be assumed that more DNA is adsorbed in acidic than in alkaline conditions. It should be also noted that nonspecific adsorption (i.e. adsorption on plain DA embedded cryogel) was independent of pH and it was almost observed to be the same at all pH values studied.
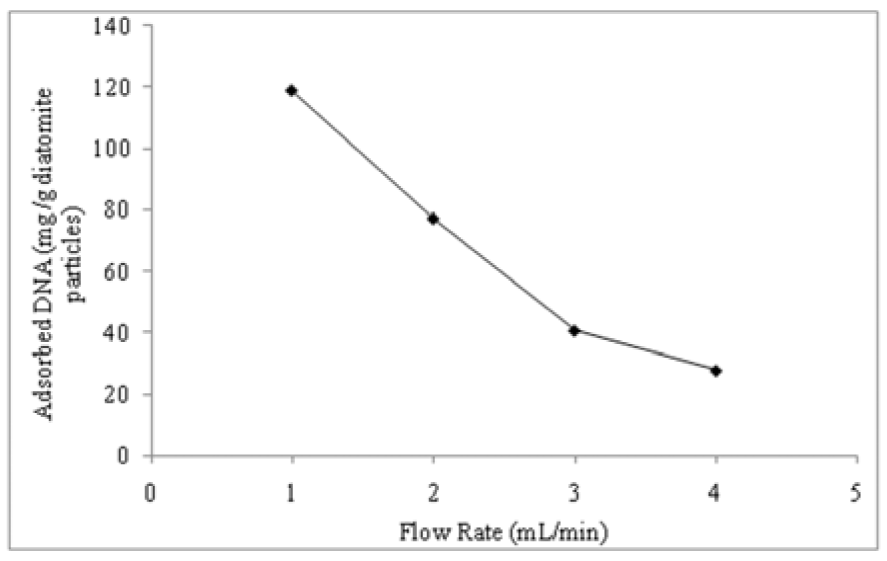
Effect of flow rate
The effect of flow rate on the adsorption of DNA in the Zn2+-DAPs embedded supermacroporous monolithic composite cryogel column was investigated by changing the flow rate from 1.0 to 4.0 mL/min. The results illustrated in the Figure 3(Fig. 3) show that DNA sorption capacity decreased with increasing flow rate. The adsorption capacity decreased considerably from 118.5 to 27.8 mg/g particle with the increase of the flow-rate from 1.0 to 4.0 mL/min. It can be concluded that decrease in flow-rate increases column efficiency. Column efficiency is approximately tripled by decreasing the flow-rate from 4.0 to 1.0 mL/min. At higher flow rates the contact time of DNA molecules with the column material is shorter. When the flow rate decreases the contact time in the column is longer. Thus, DNA molecules have more time to diffuse to the cavities of cryogel and to bind to the ligand; hence a better adsorption capacity is achieved.
Effect of DNA concentration and divalent cations
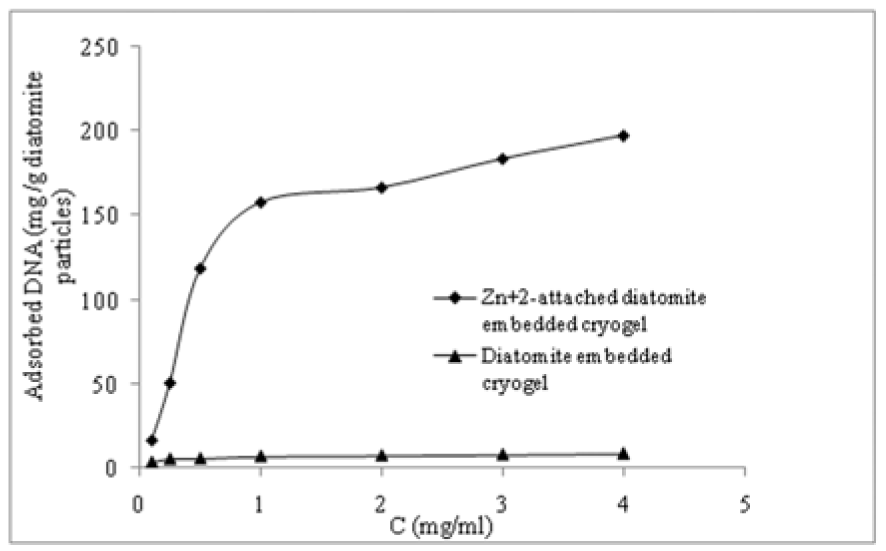
The effect of initial DNA concententration on adsorption capacity onto the p(HEMA)-DAPs and the p(HEMA)-DAPs-Zn2+ monolithic cryogels is presented in Figure 4(Fig. 4) p(HEMA)-DAPs cryogels were used as a reference to study the effect of diatomite pretreatment by divalent transition metal ion, i.e., Zn2+. Adsorption capacity is significantly higher in the presence of divalent cations (Zn2+). It appears that adsobed DNA layer is more rigid in the presence of Zn2+ due to the complexation of Zn2+ with the phosphate backbone of DNA and some negatively charged sites of diatomite particles. Also, charge screening between the surfaces and DNA molecules promoted DNA adsorption even better. As both DNA and diatomite surfaces are negatively charged, DNA approaching a surface has to overcome electrostatic repulsion (Pastrѐ et al., 2003[13]). Let us add that Zn2+ ion was preferred to alkali earth-metal ions, for binding DNA to diatomite particles since the transition metal cations coordinate strongly to the DNA bases (Rouzina and Bloomfield, 1996[14]).
The attractive hydrophobic force between the diatomite surface and DNA polyelectrolyte could be pronounced here for binding DNA to diatomite particles. Nevertheless, this force should not play a key role in the DNA adsorption regarding the DNA molecules strong dependence on the presence of divalent cations that neutralize both the diatomite surface and the DNA backbone, suggesting that the attraction force has an electrostatic origin.
DNA adsorption on p(HEMA)-DAPs cryogel was lower (about 16.1 mg/g DA particle) than the p(HEMA)-DAPs-Zn2+ monolithic composite cryogel. DNA absorption capacity of DAPs-Zn2+ monolithic composite cryogel (197.7 mg/g DA particle) was enhanced via inorganic salt bridging, i.e., Zn2+, as above mentioned. As expected, further increasing the DNA concentration, the amount of DNA adsorbed nearly reached a plateau of around 4.0 mg/mL because of saturation of the active binding sites.
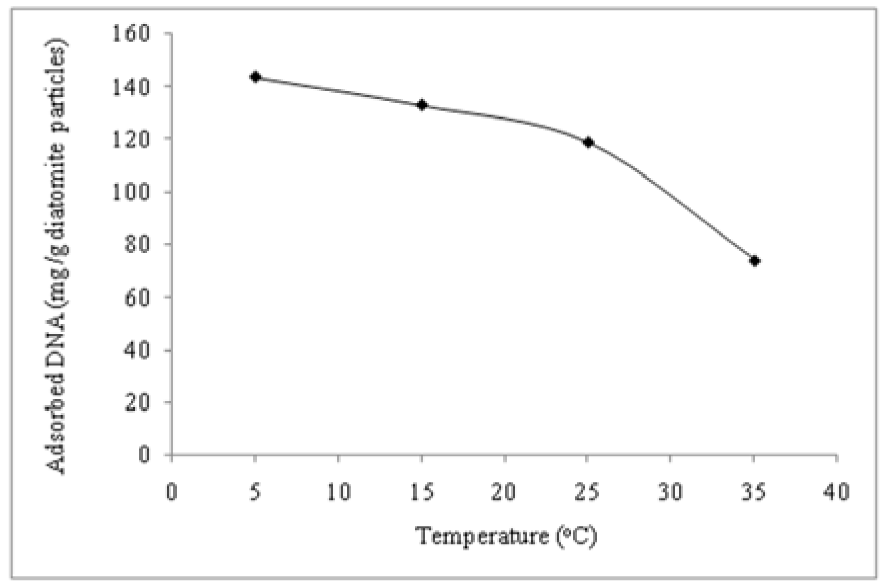
Effect of temperature
To better understand the features of DNA adsorption onto diatomite, the effect of temperature was studied in the range of 5 -35 °C (Figure 5(Fig. 5)). It appears that the DNA adsorption process is exothermic (Melzak et al., 1996[8]). It implies further that the adsorption of DNA to p(HEMA)-DAPs-Zn2+ surfaces is governed by electrostatic interactions. This could be a contributing factor for DNA adsorption as low temperatures favored the binding.
However, the temperature dependence of DNA adsorption may be due to hydrophobic interaction, macromolecular self-association or molecular diffusion (Liu et al., 2011[7]).
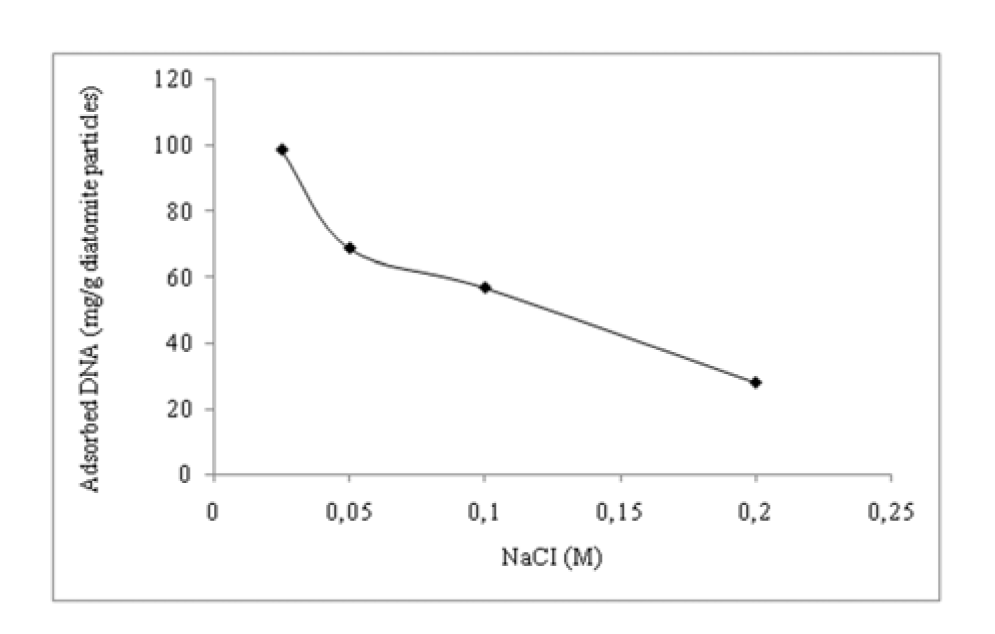
Effect of ionic strength
As presented in Figure 6(Fig. 6), DNA adsorption efficiencies were lower in the presence of Na+ ions. With the increase in ionic strength, Zn2+ ion-DNA molecule interaction was reduced. This can be attributed to electrostatic forces acting on DNA and DAPs-Zn2+ as expected from the well-known screening effect of added salt species, i.e. NaCl (Nguyen and Chen, 2007[10]). DNA molecules are highly charged poly ions and can be treated as a charged plane surface providing that ionic strength is higher than 0.1 M and is lower than 1 M (Rouzina and Bloomfield, 1996[14]; Pastrѐ et al., 2003[13]). It is likely that Na+ ions led to the charge neutralization of DNA's phosphate backbone, thus masking sorption sites. It is also possible that Na+ ions competed with Zn2+ ions, leading to repulsive electrostatic forces between like-charged Zn2+-DAPs and gDNA molecules. Hence, lower adsorption efficiencies were observed in DNA solutions containing Na+ ions.
Adsorption isotherms
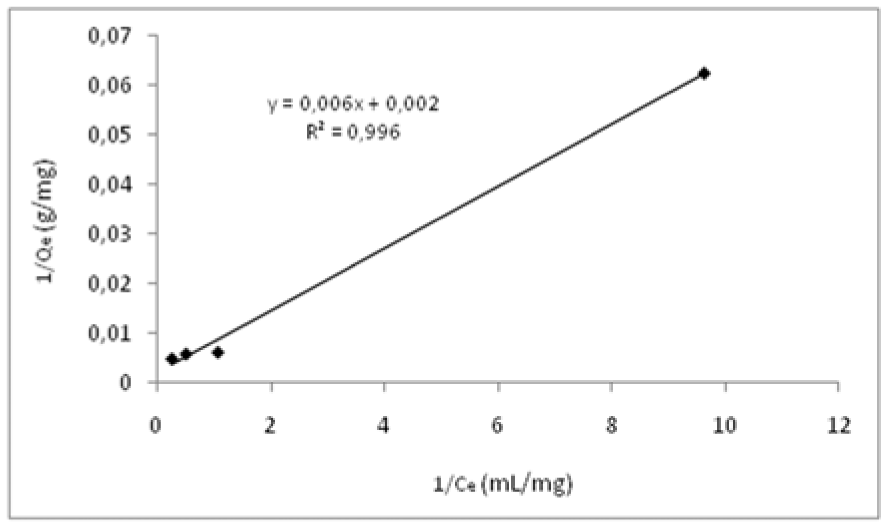
The equilibrium adsorption isotherms for DNA adsorption on Zn2+-DAPs are shown in Figure 7(Fig. 7). The adsorptive affinity of DNA to the Zn2+-DAPs was analyzed using the Langmuir equation, represented in linear form, is given by the following equation:
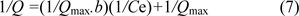
where Q is the concentration of DNA bound to the adsorbent (mg/g), Ce is the equilibrium DNA concentration in solution (mg/mL), b is the Langmuir constant (mL/mg) and Qmax is the maximum adsorption capacity (mg/g). The plots of 1/Q versus 1/Ce gave a straight line and the values of Qmax and b were calculated from the intercept and slope of the plots to be 500 mg/g and 0.317 mL/mg, respectively. The Langmuir linear regression was used for curve fitting. Evaluated experimental data and the high correlation coefficient (R2=0.9965) suggests that Langmuir curve fitting can provide a method for the comparison of the adsorption phenomena.
Conclusion
This work investigates the usability of p(HEMA)-DAPs-Zn2+ cryogels for the isolation of gDNA from aqueous media. In purification and isolation of very large biomolecules such as gDNA, porous carriers have many advantages over conventional ones. Considering the versatile properties of 3-dimensional p(HEMA) hydrogel and its large convective pores accessible to large gDNA molecules, this study opens the door for future application of p(HEMA)-diatomite composite gels in the design of functionalized adsorption columns. The adsorption study we describe here takes advantage of the of high porosity, good hydrophilicity, high chemical inertness nature of diatomite and biocompatibility of p(HEMA) based hydrogel composite columns. The production of p(HEMA)-DAPs-Zn2+ cryogels provides a fast and convenient approach for producing robust p(HEMA)-composite materials with high affinity for target biomolecules i.e. DNA in aqueous media. In recent years, there is a growing interst for developing supermacroporous monolithic cryogels as adsorbents for diverse applications including proteomic studies, protein purification and biomedical therapy for such diseases as SLE (Odabaşı and Denizli, 2001[12]). Furthermore, this present study provides a foundation for assessment of the fate of extracellular DNA molecules (excreted actively by living cells or released from lysed dead cells from various organisms) in soil environment.
References
[*] Corresponding Author:
Mahmut Erzengin, Aksaray University, Faculty of Art and Science, Chemistry Department, 68100 Aksaray, Turkey; Tel: +90-382-288-2137, Fax: +90-382-288-2125, eMail: merzengin@hotmail.com