Research article
The relationship between serology of hepatitis E virus with liver and kidney function in kidney transplant patients
Abbas Ali Zeraati1, Fatemeh Nazemian1, Ladan Takalloo1, Amirhossein Sahebkar2,3, Elahe Heidari4, Mohammad Ali Yaghoubi5
1Kidney transplantation Complications Research Center, Mashhad University of Medical Sciences, Mashhad, Iran2Biotechnology Research Center, Mashhad University of Medical Sciences, Mashhad, Iran
3Metabolic Research Centre, Royal Perth Hospital, School of Medicine and Pharmacology, University of Western Australia, Perth, Australia
4Department of Pediatrics, Imam Reza Hospital , Mashhad University of Medical Sciences, Mashhad, Iran
5Endocrine Research Center, Ghaem Hospital, Mashhad University of Medical Science, Mashhad, Iran
EXCLI J 2016;15:Doc343
Abstract
Although hepatitis E virus (HEV) is well known to cause acute hepatitis, there are reports showing that HEV may also be responsible for progression of acute to chronic hepatitis and liver cirrhosis in patients receiving organ transplantation. In this study, we aimed to evaluate the prevalence of HEV in patients with kidney transplantation. In this study, 110 patients with kidney transplantation were recruited, and anti-HEV IgG, creatinine, alanine transaminase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), and estimated glomerular filtration rate (eGFR) in the first, third and sixth months after renal transplantation were measured. The mean serum anti-HEV IgG titers in the study participants was 1.36 (range 0.23 to 6.3). Twenty-three patients were found to be seropositive for HEV Ab defined as anti-HEV IgG titer > 1.1. The difference in liver and renal function tests (creatinine, eGFR, AST, ALT and ALP) at different intervals was not significant between patients with HEV Ab titers higher and lower than 1.1 (p > 0.05). However, an inverse correlation was observed between HEV Ab and eGFR values in the first (p = 0.047, r = -0.21), third (p = 0.04, r = -0.20) and sixth (p = 0.04, r = -0.22) months after renal transplantation in patients with HEV Ab < 1.1 but not in the subgroup with HEV Ab > 1.1. Also, a significant correlation between age and HEV Ab levels was found in the entire study population (p = 0.001, r = 0.33). Our findings showed a high prevalence of seropositivity for anti-HEV IgG in patients receiving renal transplants. However, liver and renal functions were not found to be significantly different seropositive and seronegative patients by up to 6 months post-transplantation.
Keywords: hepatitis E, kidney transplant, creatinine, alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase
Introduction
Hepatitis E, which is generally a self-limiting disease, is caused by hepatitis E virus (HEV) (Balayan et al., 1983[7]; Gupta and Smetana, 1957[12]). This virus is a single-stranded RNA virus without envelope, and is the only member of hepevirus from the Hepeviridae family (Kane et al., 1984[18]; Koonin et al., 1992[24]). Distribution of HEV, which was previously known as non-A and non-B hepatitis, is mainly through water and feces, as is for viral hepatitis A (HAV) (Balayan et al., 1983[7]; Khuroo, 1980[21]). However, the transmission of HAV is much easier with global distribution and causes more infections whereas HEV is more distributed in industrialized countries (Clemente-Casares et al., 2003[10]; Tang et al., 1991[35]). The highest incidence of HEV infection is reported in Asia, Africa, the Middle East and Central America (Arankalle et al., 1988[5]; Gupta and Smetana, 1957[12]). Iran is one of the countries where hepatitis E is endemic, and several HEV outbreaks have occurred so far (Alavian et al., 2009[3]; Ghorbani et al., 2007[11]). Although the transmission of hepatitis E virus has been reported from animal to human in developed countries, the major route of transmission is through the fecal-oral route. It has also been found that the consumption of undercooked meat or meat products is a major risk factor for HEV infection (Legrand-Abravanel et al., 2010[26]). The most important diagnostic methods of HEV are PCR amplification of virus RNA or detection of Immunoglobulin M (IgM) antibodies against HEV. Currently, serological tests and qualitative and quantitative assessment of HEV RNA are considered as the gold standard tests for detection of HEV both for diagnostic and epidemiological purposes (Baylis et al., 2011[8]; Khudyakov and Kamili, 2011[19]; Takahashi et al., 2005[34]). Several interventions such as elimination of chronic infection after reduction or discontinuation of immunosuppressive therapy, use of oral ribavirin and pegylated interferon have been suggested to be effective in the management of HEV infection. However, there is as yet no proven treatment for acute or chronic hepatitis E in organ transplant patients (Aggarwal and Jameel, 2011[2]; Chaillon et al., 2011[9]; Kamar et al., 2011[15], 2014[16]).
Hepatitis E is responsible for acute hepatitis, and it was previously believed that HEV cannot be developed to chronic hepatitis. However, more recent reports show that HEV can lead to chronic hepatitis as well as liver cirrhosis in patients receiving organ transplants (Aggarwal, 2008[1]; Koning et al., 2015[23]). It has been proposed that HEV does not usually lead to chronic hepatitis, except in patients who have solid organ transplantation or those whose immune system is suppressed (Khuroo et al., 1980[22]). Patients with organ transplantation are more susceptible to diseases caused by rare infectious viruses such as HEV; thus, differential diagnosis of pathogens such as hepatitis E virus in transplant recipients is suggested to be applied according to the reports showing that there is a relatively rapid progression to cirrhosis and chronic hepatitis E in people who have had organ transplants (Ibarra et al., 1994[14]; Stefanidis et al., 2004[32]). Seroprevalence studies have shown that 6 to 15.6 % of kidney transplant patients are positive for anti-HEV IgG. In addition, studies have shown that HEV infection is commonly observed in patients with transplantation of solid organs such as the kidneys, liver and pancreas (Kamar et al., 2008[17]; Khuroo, 2008[20]). The frequency of acute HEV infection in French patients with solid organ transplantation has been reported to be between 5 and 6.5 % (Legrand-Abravanel et al., 2011[27]). Other factors that may lead to chronic HEV infection in patients with kidney transplantation include low lymphocyte (CD2, CD3 and CD4) count, low platelet count, young age < 52 years and lymphopenia (Kamar et al., 2011[15]; Legrand-Abravanel et al., 2011[27]).
Some studies have investigated the prevalence of hepatitis E in patients with kidney transplantation (Kamar et al., 2008[17]; Legrand-Abravanel et al., 2011[27]). Given the high prevalence of transaminitis in the early stages after kidney transplantation and also uncertainty about the role of hepatitis E, in this study, the serology of hepatitis E was evaluated in relation to hepatic and renal function indices in a group of patients with kidney transplantation.
Methods
Data collection and population size calculation
Demographic information of patients including age, sex, and type of organ transplant donor (living or cadaver) was collected based on medical history and questionnaire. All patients underwent standard triple immunosuppressive therapy with prednisone, cyclosporine and Mycophenolate mofetil. Samples were randomly selected from patients with kidney transplantation in the Montaserieh Hospital, Mashhad, Iran. Patients with transplant rejection or positive hepatitis B surface antigen (HbsAg), anti-hepatitis C virus (HCV), and anti-cytomegalovirus (CMV) IgM were excluded. Considering that the prevalence of HEV infection has been reported to be 7.7 % in previous studies (Kamar et al., 2008[17]; Khuroo, 2008[20]; Legrand-Abravanel et al., 2011[27]), the population size was calculated to be 110 patients using the following formula.
N = (1.96)2×p(1-p)/d2=(1.96)2×0.077(1-0.077)/(0.05)2=110
Biochemical assessment
Serum levels of anti HEV IgG, creatinine, alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), and estimated glomerular filtration rate (eGER) were measured at the first week, first month, third month, and sixth month post-transplantion. To determine anti-HEV IgG level, 2 mL of venous blood were taken and measured with ELISA after separating the serum from the blood. eGFR was calculated by the modification of diet in renal disease (MDRD) using the following equation:
MDRD = 175.0 × (Serum Creatinine)-1.154
× (Age)-0.203 × 0.742 (if female)
× 1.21 (if black)
Statistical analysis
Descriptive data are expressed as mean ± SD. Between-group comparisons were performed using independent samples t-test (for normally distributed data) or Mann-Whitney U (for non-normally distributed data) test. Pearson (for normally distributed data) or Spearman (for non-normally distributed data) correlation coefficients test were used to evaluate the correlation between serology of hepatitis E with renal and hepatic function tests. All statistical analyses were performed using SPSS 15 software, and p-values less than 0.05 were considered as statistically significant.
Results
Demographic information of the patients
Among patients with renal transplantation admitted to the Montaserieh hospital, 110 patients were randomly selected. The patients were enrolled after obtaining written informed consent. Of the enrolled participants, 59 patients (64.53 %) were male and 51 (36.46 %) were female. The mean age of patients was 36.97 years, ranging between 15 and 62 years.
Assessment of biochemical markers
Most of the patients (93.6 %) had hemodialysis before receiving the transplanted kidney, and 35 patients had a history of blood transfusion (31.82 %). The mean serum anti-HEV IgG titers in the study participants was 1.36 (range 0.23 to 6.3). Antibody titer for anti-HEV IgG was positive for 23 patients (20.9 %), and negative in 86 (78.18 %); in addition, antibody levels in 1 case was on the borderline (0.91 %) that was further considered as seronegative.
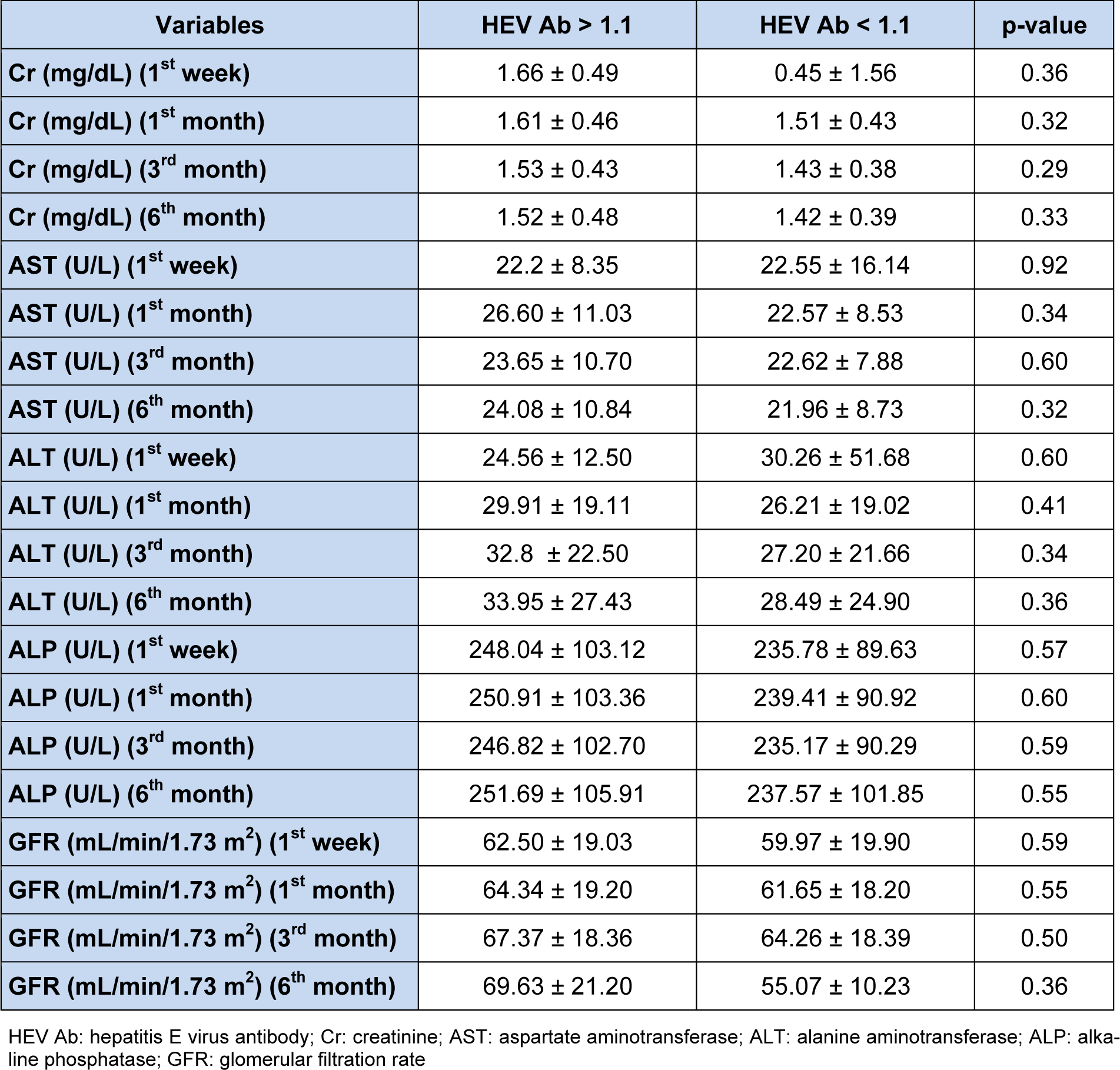
The results showed that there is no significant difference in serum concentrations of creatinine, AST, ALT, and ALP at different intervals between patients with HEV Ab titers higher and lower than 1.1. The mean value of serum ALP six months after transplantation in seropositive and seronegative groups was 91.105 ± 69.251 and 57.237 ± 85.101, respectively, that was not statistically significant (p = 0.55). Mean serum creatinine level at the sixth month after transplantation in the seropositive and seronegative groups were 1.52 ± 0.48 and 1.42 ± 0.39, respectively, indicating that the difference between seropositive and seronegative groups was not significant (p = 0.33). In the group of patients with HEV Ab lower than 1.1, a negative correlation was found between the HEV Ab and the eGFR in the first month (p = 0.047, r = -0.21), the third month (p = 0.04, r = -0.2) and the sixth month (p = 0.04, r = -0.22) after transplantation. However, no significant difference was observed in serum concentrations of creatinine, AST, ALT, and ALP at different intervals between patients with HEV Ab titers higher and lower than 1.1. Moreover, a positive correlation was found between age and serum HCV Ab titers (p = 0.001, r = 0.33) in the entire study population. Serum concentrations of creatinine, AST, ALT, ALP and eGFR at different intervals in the seropositive and seronegative subjects are demonstrated in Table 1(Tab. 1).
Raw data are shown in Supplementary Table 1Sahebkar_Yaghoub_02062016_Supplementary_table_1.pdf.
Discussion
Kidney transplant recipients are known to be more susceptible to viral infections and show more severe clinical manifestation compared with healthy individuals (Quintana et al., 2005[30]; Tang et al., 1991[35]). More susceptibility to viral infections in these patients is mainly due to immunosuppression caused by immunosuppressive drugs. Although hepatitis E was once thought to be a self-limited acute infection that rarely leads to chronic infection, several recent investigations have reported chronic hepatitis and even cirrhosis related to hepatitis E in organ transplant recipients (Ibarra et al., 1994[14]; Tsega et al., 1991[37]; Yamashita et al., 2009[38]).
In a study in 2011, it was shown that there was no significant difference in serum ALT levels between patients with positive and negative anti-HEV IgG (Rostamzadeh Khameneh et al., 2011[31]). It was also shown that there is no association between hepatitis E infection and increased serum levels of ALT. Hence, it was deduced that anti-HEV IgG antibodies is significantly higher in kidney transplant patients compared with general or hemodialysis population. In the mentioned study, of the total number of patients, 28 patients were seropositive (30.8 %), whereas in our study, the prevalence of seropositivity for anti-HEV IgG was 20 %. Moreover, consistent with the above-mentioned study, our findings showed that there was no significant association between serum ALT levels and anti-HEV IgG in seropositive and seronegative groups. Nonetheless, some studies have reported a positive association between the seroprevalence of HEV and blood transfusion in patients with organ transplantation (Arankalle et al., 1995[6]; Lynch et al., 1995[29]; Sylvan, 1998[33]). In a previous study on the epidemiology of anti-HEV IgG antibodies in healthy blood donors of Iran, the prevalence of HEV infections was reported to be 7.7 % (Arabzadeh et al., 2010[4]; Hesamizadeh et al., 2016[13]). It has also been shown that in a particular group of patients, such as patients undergoing hemodialysis, HEV seroprevalence correlates with the prevalence of infections in the community (Taremi et al., 2008[36]). In different populations receiving kidney or liver transplantation, the HEV seroprevalence patients were reported between 1 % and 14.5 % (Koonin et al., 1992[24]; Yamashita et al., 2009[38]). Outbreak of hepatitis E also varies in different geographical areas. According to epidemiological studies, low prevalence of anti-HEV IgG antibodies have been reported in countries such as Israel (8.2 %) and Turkey 8.3 %, but the prevalence of antibodies is rather high in countries such as Iraq (14.8 %), Saudi Arabia (16.4 %) and Pakistan (17.5 %) (Kane et al., 1984[18]; Krawczynski and Bradley, 1989[25]; Yamashita et al., 2009[38]). In a study that was conducted on 700 patients with organ transplantation, 34 patients were found to be infected with HEV one year after transplantation, and among them, 47 % were identified with chronic HEV and accordingly the annual incidence of HEV was reported to be 3.2 % (Lu et al., 2006[28]).
In conclusion, our findings in this study indicated that the prevalence of anti-HEV IgG is rather high (20 %) in Iranian patients with renal transplantation. However, it was shown that serum levels of creatinine, AST, ALT, and ALP, and eGFR at different intervals is comparable between patients with HEV Ab titers higher and lower than 1.1.
Acknowledgement
The authors are thankful to the Research Council of the Mashhad University of Medical Sciences for the financial support of this study.
Conflict of interest
The authors declare no conflict of interest.
References
File-Attachments
- Sahebkar_Yaghoub_02062016_Supplementary_table_1.pdf (84,22 KB)
Supplementary table 1