Letter to the editor
Recent update on barbiturate in relation to brain disorder
Sachchidanand Pathak1, Gaurav Gupta1, Lakshmi Thangavelu2, Sachin K. Singh3, Kamal Dua4, Dinesh Kumar Chellappan5, Ritu M. Gilhotra1
1School of Pharmacy, Suresh Gyan Vihar University, Mahal Road-302017, Jagatpura, Jaipur, India
2Department of Pharmacology, Saveetha Dental College and Hospitals, Saveetha Institute of Medical and Technical Sciences, Saveetha University, Chennai, India
3School of Pharmaceutical Sciences, Lovely Professional University, Phagwara, Punjab, 144411, India
4Discipline of Pharmacy, Graduate School of Health, University of Technology Sydney, NSW 2007, Australia
5Department of Life Sciences, School of Pharmacy, International Medical University, Bukit Jalil 57000, Kuala Lumpur, Malaysia
EXCLI J 2021;20:Doc1028
Dear Editor,
Barbiturate is a potent substance which forms a quintessential part of the NDPS Act. The substance is categorized under the psychoactive groups of drugs and is essentially a drug that possesses both hypnotic and sedative properties. The precursor for barbiturate is barbituric acid which is a condensation product of malonic acid and urea. However, barbituric acid itself is not a centrally acting depressant. Diethylbarbituric acid (Veronal) is the first ever barbiturate with hypnotic properties that was used as early as 1903 (Hadjihambi et al., 2020[5]). The drug induced sleep both in human and animals. The substance was also called as barbital. Later in the year 1912, a second barbiturate drug, phenobarbitone was introduced into clinical practice which had both sedative and hypnotic properties. The phenomenal success of both these drugs announced the beginning of the barbiturate era. Their influence as the pre-eminent sedative-hypnotic agents was felt for over half a century. Although several so-called non-barbiturate drugs attempted to displace the barbiturates from their pinnacle from time to time, it was not until 1961 when a substance named chlordiazepoxide was introduced into the market that their position was seriously challenged (Velle et al., 2021[23]). Several earlier studies have reported the characteristic features and the severity of the barbiturate withdrawal syndrome. In cases of mild withdrawal syndrome, symptoms like apprehension, hyperexcitability, mild tremors, loss of appetite and piloerection were observed. An intermediate withdrawal syndrome exhibited tightness in the muscles, extreme tremors, sudden loss of body weight, altered motor activity, excessive nausea, and vomiting (Sharpe et al., 2020[16]). The hallmarks of a severe withdrawal syndrome are convulsions, delirium or hallucination and hyperthermia or unusually high fever. The severity of withdrawal syndrome has been shown to depend on the frequency of drug administration and the duration of action of the drug. We review recent research on the role of barbiturates in brain disorders in this letter (Table 1(Tab. 1); References in Table 1: Brandt et al., 2018[1]; Chakraborty and Hocker, 2019[2]; Colton et al., 2014[3]; Forsyth et al., 2003[4]; Hadjihambi et al., 2020[5]; Hocker et al., 2018[6]; Klein et al., 2015[7]; Lewis and Adams, 2021[8]; Mairinger et al., 2012[9]; Mansour et al., 2013[10]; Murphy et al., 2020[11]; Ryu et al., 2019[12]; Sakuma et al., 2020[13]; Sánchez Fernández et al., 2019[14]; Schizodimos et al., 2020[15]; Shein et al., 2016[17]; Specchio and Pietrafusa, 2020[18]; Tat et al., 2017[19]; Töllner et al., 2014[20]; Tremont-Lukats et al., 2008[21]; Velle et al., 2019[22]; Wang et al., 2018[24]; Xie et al., 2009[25]; Young et al., 2016[26]; Zhang et al., 2019[27]).
Conflict of interest
The authors declare no conflict of interest.
References
1.
Brandt C, Seja P, Töllner K, Römermann K, Hampel P, Kalesse M, et al. Bumepamine, a brain-permeant benzylamine derivative of bumetanide, does not inhibit NKCC1 but is more potent to enhance phenobarbital's anti-seizure efficacy. Neuropharmacology. 2018;143:186-204. 2.
Chakraborty T, Hocker S. Weaning from antiseizure drugs after new onset status epilepticus. Epilepsia. 2019;60:979-85. 3.
Colton K, Yang S, Hu PF, Chen HH, Bonds B, Scalea TM, et al. Intracranial pressure response after pharmacologic treatment of intracranial hypertension. J Trauma Acute Care Surg. 2014;77:47-53.4.
Forsyth PA, Weaver S, Fulton D, Brasher PM, Sutherland G, Stewart D, et al. Prophylactic anticonvulsants in patients with brain tumour. Can J Neurol Sci. 2003;30:106-12. 5.
Hadjihambi A, Karagiannis A, Theparambil SM, Ackland GL, Gourine AV. The effect of general anaesthetics on brain lactate release. Eur J Pharmacol. 2020;881:173188. 6.
Hocker S, Clark S, Britton J. Parenteral phenobarbital in status epilepticus revisited: Mayo Clinic experience. Epilepsia. 2018;59(Suppl 2):193-7. 7.
Klein S, Bankstahl M, Löscher W. Inter-individual variation in the effect of antiepileptic drugs in the intrahippocampal kainate model of mesial temporal lobe epilepsy in mice. Neuropharmacology. 2015;90:53-62. 8.
Lewis CB, Adams N. Phenobarbital. StatPearls. Treasure Island (FL): StatPearls Publishing, 2021.9.
Mairinger S, Bankstahl JP, Kuntner C, Römermann K, Bankstahl M, Wanek T, et al. The antiepileptic drug mephobarbital is not transported by P-glycoprotein or multidrug resistance protein 1 at the blood-brain barrier: a positron emission tomography study. Epilepsy Res. 2012;100:93-103. 10.
Mansour N, deSouza RM, Sikorski C, Kahana M, Frim D. Role of barbiturate coma in the management of focally induced, severe cerebral edema in children. J Neurosurg Pediatr. 2013;12:37-43. 11.
Murphy L, Wolfer H, Hendrickson RG. Toxicologic confounders of brain death determination: A narrative review. Neurocrit Care. 2020;2020:1-18. 12.
Ryu JA, Jung W, Jung YJ, Kwon DY, Kang K, Choi H, et al. Early prediction of neurological outcome after barbiturate coma therapy in patients undergoing brain tumor surgery. PLoS One. 2019;14(4):e0215280. 13.
Sakuma H, Horino A, Kuki I. Neurocritical care and target immunotherapy for febrile infection-related epilepsy syndrome. Biomed J. 2020;43:205-10. 14.
Sánchez Fernández I, Gaínza-Lein M, Lamb N, Loddenkemper T. Meta-analysis and cost-effectiveness of second-line antiepileptic drugs for status epilepticus. Neurology. 2019;92:e2339-e48. 15.
Schizodimos T, Soulountsi V, Iasonidou C, Kapravelos N. An overview of management of intracranial hypertension in the intensive care unit. J Anesth. 2020;34:741-57. 16.
Sharpe C, Reiner GE, Davis SL, Nespeca M, Gold JJ, Rasmussen M, et al. Levetiracetam versus phenobarbital for neonatal seizures: A randomized controlled trial. Pediatrics. 2020;145(6):e20193182.17.
Shein SL, Ferguson NM, Kochanek PM, Bayir H, Clark RS, Fink EL, et al. Effectiveness of pharmacological therapies for intracranial hypertension in children with severe traumatic brain injury - results from an automated data collection system time-synched to drug administration. Pediatr Crit Care Med. 2016;17:236-45. 18.
Specchio N, Pietrafusa N. New-onset refractory status epilepticus and febrile infection-related epilepsy syndrome. Dev Med Child Neurol. 2020;62:897-905. 19.
Tat YB, Hassan W, Chuen TY, Ghani ARI. Life-threatening dyskalaemia after barbiturate coma therapy: The strategy of management. Malays J Med Sci. 2017;24:100-5. 20.
Töllner K, Brandt C, Töpfer M, Brunhofer G, Erker T, Gabriel M, et al. A novel prodrug-based strategy to increase effects of bumetanide in epilepsy. Ann Neurol. 2014;75:550-62. 21.
Tremont-Lukats IW, Ratilal BO, Armstrong T, Gilbert MR. Antiepileptic drugs for preventing seizures in people with brain tumors. Cochrane Database Syst Rev. 2008(2):Cd004424.22.
Velle F, Lewén A, Howells T, Enblad P, Nilsson P. Intracranial pressure-based barbiturate coma treatment in children with refractory intracranial hypertension due to traumatic brain injury. J Neurosurg Pediatr. 2019:1-9;epub ahead of print.23.
Velle F, Lewén A, Howells T, Nilsson P, Enblad P. Temporal effects of barbiturate coma on intracranial pressure and compensatory reserve in children with traumatic brain injury. Acta Neurochir (Wien). 2021;163:489-98. 24.
Wang SN, Gu CP, Liu GH, Lin ZZ, Zheng P, Pan SY, et al. The effectiveness of phenobarbital in patients with refractory status epilepticus undergoing therapeutic plasma exchange. Neuroreport. 2018;29:1360-4. 25.
Xie J, Li Y, Huang Y, Qiu P, Shu M, Zhu W, et al. Anesthetic pentobarbital inhibits proliferation and migration of malignant glioma cells. Cancer Lett. 2009;282:35-42. 26.
Young L, Berg M, Soll R. Prophylactic barbiturate use for the prevention of morbidity and mortality following perinatal asphyxia. Cochrane Database Syst Rev. 2016 (5):Cd001240.27.
Zhang Q, Yu Y, Lu Y, Yue H. Systematic review and meta-analysis of propofol versus barbiturates for controlling refractory status epilepticus. BMC Neurol. 2019;19(1):55.
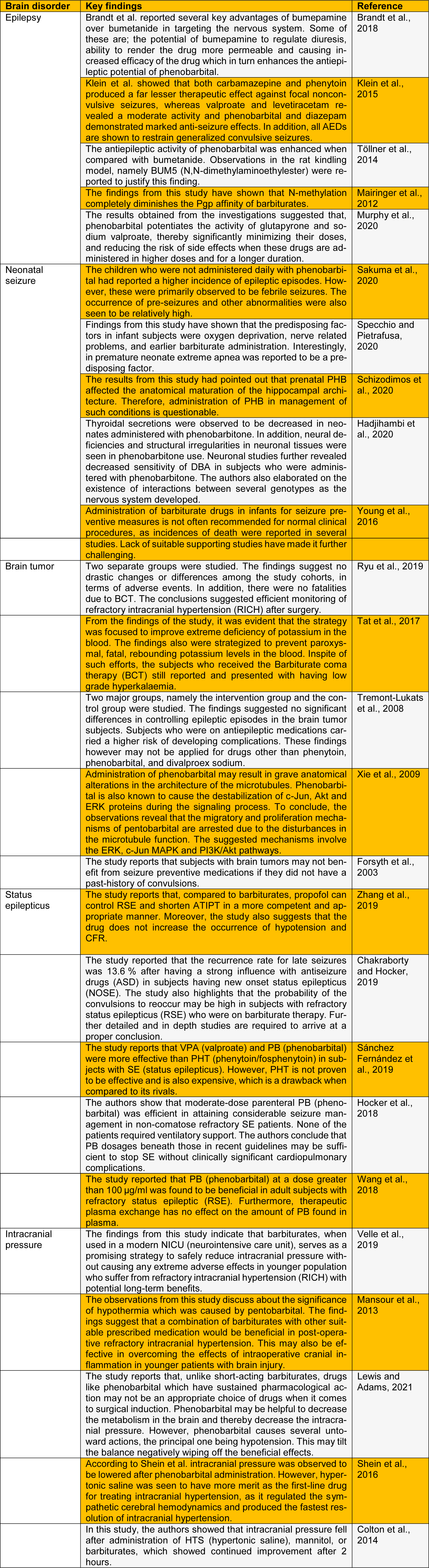
Table 1: Recent study on the role of barbiturates in brain disorders
