Review article
Natural sweetener Stevia rebaudiana: Functionalities, health benefits and potential risks
Victoria Peteliuk1, Lesia Rybchuk1, Maria Bayliak1, Kenneth B. Storey2, Oleh Lushchak1[*],3
1Department of Biochemistry and Biotechnology, Vasyl Stefanyk Precarpathian National University, 57 Shevchenko Str., Ivano-Frankivsk, 76018, Ukraine2Institute of Biochemistry, Carleton University, 1125 Colonel By Drive, Ottawa, Ontario K1S 5B6, Canada
3Research and Development University, Shota Rustaveli Str., 76000, Ivano-Frankivsk, Ukraine
EXCLI J 2021;20:Doc1412
Abstract
Stevia rebaudiana is a South American plant, the cultivation of which is increasing worldwide due to its high content of sweet compounds. Stevia sweetness is mainly due to steviol glycosides, that are ~250-300 times sweeter than sucrose. Many studies have suggested the benefits of Stevia extract over sugar and artificial sweeteners, but it is still not a very popular sugar substitute. This review summarizes current data on the biological activities of S. rebaudiana extract and its individual glycosides, including anti-hypertensive, anti-obesity, anti-diabetic, antioxidant, anti-cancer, anti-inflammatory, and antimicrobial effects and improvement of kidney function. Possible side effects and toxicity of Stevia extract are also discussed.
Keywords: Stevia rebaudiana, stevioside, rebaudioside A, sweetener, health benefits
Abbreviations
CFTR, cystic fibrosis transmembrane conductance regulator; GFR, glomerular filtration rate; LPS, lipopolysaccharide; MDCK cells, Madina-Darby canine kidney cells; NF-kB, nuclear factor kappa-light-chain-enhancer of activated B cells; Nrf2, nuclear erythroid factor 2; TRPM5, transient receptor potential cation channel subfamily melastatin member 5; TPA, 12-O-tetradecanoyl-phorbol-13-acetate.
Introduction
The genus Stevia of the Asteraceae family includes 230 species, but only one of them, Stevia rebaudiana Bertoni produces sweet steviol glycosides. It was earlier thought that S. phlebophylla also has this property, but new research denies this (Brandle and Telmer, 2007[17]; Ceunen et al., 2013[21]).
S. rebaudiana is a perennial shrub growing in South America, in particular in Brazil and Paraguay, where it is also known as “Honey Leaf”, “Sweet-Leaf” or “Sweet-Herb”. Stevia preparations are used in many forms such as fresh and dried Stevia leaves, Stevia leaf powder, extracts and liquid concentrates. Stevia extract is a great alternative for synthetic sweeteners being approximately 200 to 300 times sweeter than sugar (Singh and Rao, 2005[100]). Many studies have shown that Stevia leaf preparations as a natural non-calorie sugar substitute is not only safe for people with diabetes, high blood pressure, and obesity but also can be used for the treatment of these diseases or prevention of their complications (Carrera-Lanestosa et al., 2017[19]). Safety of the consumption of the herb by children was also shown (Aguero et al., 2014[4]).
The main compounds responsible for the sweetness of Stevia preparations are steviol diterpene glycosides (Figure 1(Fig. 1)) called stevioside and rebaudiosides, and they are used in the food industry as sweeteners. In addition to sweet compounds, Stevia leaves contain many other biologically active substances, which have beneficial effects for human health. In particular, the anti-diabetic, antihypertensive, antitumor, anti-cariogenic, anti-inflammatory and bactericidal effects of the herb have been studied, and these issues are discussed in detail below. There are also data on the protective effects of Stevia on the digestive system and skin disorders as well as on common complications associated with metabolic syndrome (Singh and Rao, 2005[100]; Sanches Lopes et al., 2016[93]; Ranjbar et al., 2020[86]).
History of Stevia in the World
Stevia has been known to people from ancient times. The Guarani Indians called Stevia “Ka-a He-e”, which means “sweet grass”, and used it to savor bitter drinks such as mate. There are some reports that Stevia was already known in Spain in the 16th century. However, other Europeans learned about the plant only in the late 19th century, after Stevia was introduced and promoted by the botanist Moises Santiago Bertoni (Ramesh et al., 2006[85]). Bertoni renamed it from Eupatorium rebaudianum to Stevia rebaudiana (Carakostas et al., 2008[18]). In 1901, Bertoni wrote that a few leaves of Stevia grass were enough to sweeten a large cup of tea. In 1920, Stevia began to be cultivated in large quantities on plantations in Brazil and Paraguay. In 1931, the French chemists Briedel and Lavieille isolated the glycoside which provide Stevia with its sweet taste. This compound was named stevioside (Barriocanal et al., 2008[13]). During World War II, Stevia began to be actively consumed in the United Kingdom due to sugar shortages and rationing of its consumption (Zaman et al., 2015[119]).
In the 1970s, Stevia was introduced in Japan and research was started to evaluate its beneficial potential for human health. Since then, the Japanese actively use this sweetener in a variety of foods and Japan is one of the major producers of Stevia now (Ramesh et al., 2006[85]). In 2013, the Coca-Cola Company started production of drinks containing Stevia instead of sugar and having 30 % less calories. These drinks are now sold in a number of countries worldwide (Heyden, 2013[41]). In addition to Japan, Brazil, and Paraguay, Stevia is commercially cultivated in many other countries around the world, including Ukraine (Midmore and Rank, 2002[64]).
Biologically Active Compounds of Stevia
The leaves of S. rebaudiana contain more than 30 different steviol glycosides, among which stevioside and rebaudioside A are present in the highest levels (Figure 1(Fig. 1)). Stevioside and rebaudioside A are also the main sweet components of Stevia. Stevioside was first isolated from Stevia in 1931 and its chemical structure was established in 1952. Stevioside is a diterpene glycoside consisting of three molecules of glucose and a glucone moiety - steviol. Stevioside content is from 4 % to 13 % of all glycosides in Stevia (Marcinek and Krejpcio, 2015[56]). Rebaudioside A is another steviol diterpene glycoside; the percentage of its sweetness fluctuates from 30 % to 40% that is ~ 180-400 times sweeter than sugar (Kaplan and Turgut, 2019[47]). The concentration of stevioside is 6.5 - 9.1 % and rebaudioside A is 2.3 - 3.8 % (Atteh et al., 2011[12]; Goyal et al., 2010[39]).
In total, the best-known compounds that have been isolated from Stevia leaves are glycosides, stevioside, several types of rebaudiosides (from A to F), steviolmonoside, rubusoside, dulcoside A, and steviolbioside (Starratt et al., 2002[102]; Savita et al., 2004[95]). Less common are penta-glucoside rebaudioside D and hexa-glucoside rebaudioside M (Olsson et al., 2016[71]). Rebaudioside D and rebaudioside M are present in dried S. rebaudiana leaves in very low quantities of 0.2 % and 0.1 %, respectively (Neuwirth, 2020[68]). Despite that, rebaudioside M is commercialized by PureCircle Limited and the Coca-Cola Company for food and beverage use (Prakash et al., 2014[81]). Rebaudioside D can also be used in the food industry as a non-caloric sweetener (Allen et al., 2013[8]).
Recently, other rebaudiosides have been identified in Stevia, namely rebaudioside U, T, R and S (Ibrahim et al., 2016[43]; Perera et al., 2017[76]). Steviol and isosteviol, which are products of hydrolysis of stevioside can be used for therapeutic purposes (Wang et al., 2018[116]). Most of these isolated diterpenoid glycosides have the same chemical backbone structure (steviol) but differ in the residues of carbohydrate at positions C13 and C19 (Shibata et al., 1995[97]; Purkayastha and Kwok, 2020[84]). Sweet Stevia glycosides differ both in the degree of sweetness and in quantitative content in the leaves. The most abundant glycosides, stevioside and rebaudiosides, are the sweetest compounds of Stevia and in the refined form are in 150-300 and 250-450 times sweeter than sucrose, respectively (Marcinek and Krejpcio, 2015[56]). Processed forms of Stevia leaves are generally 250-300 times sweeter than sugar.
In addition to sweet compounds, Stevia leaves contain proteins, carbohydrates, lipids, dietary fibers, oils, vitamins, and phenolic compounds. Dried Stevia leaves contain (per 100 g of dried mass) 11.2-16.0 g proteins, 61.9 g carbohydrates, 1.9-3.73 g lipids and 6.8-15.2 g dietary fiber (Abou-Arab et al., 2010[2]; Goyal et al., 2010[39]; Atteh et al., 2011[12]).
The chemical composition of preparations from Stevia differs in dry and fresh leaves and depends on the method of processing or extraction (Snehal and Madhukar, 2012[101]).
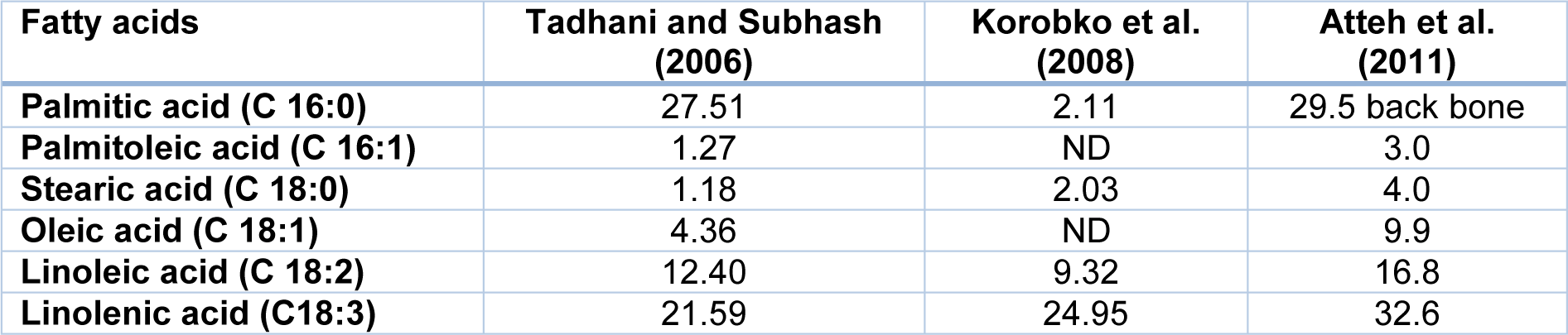
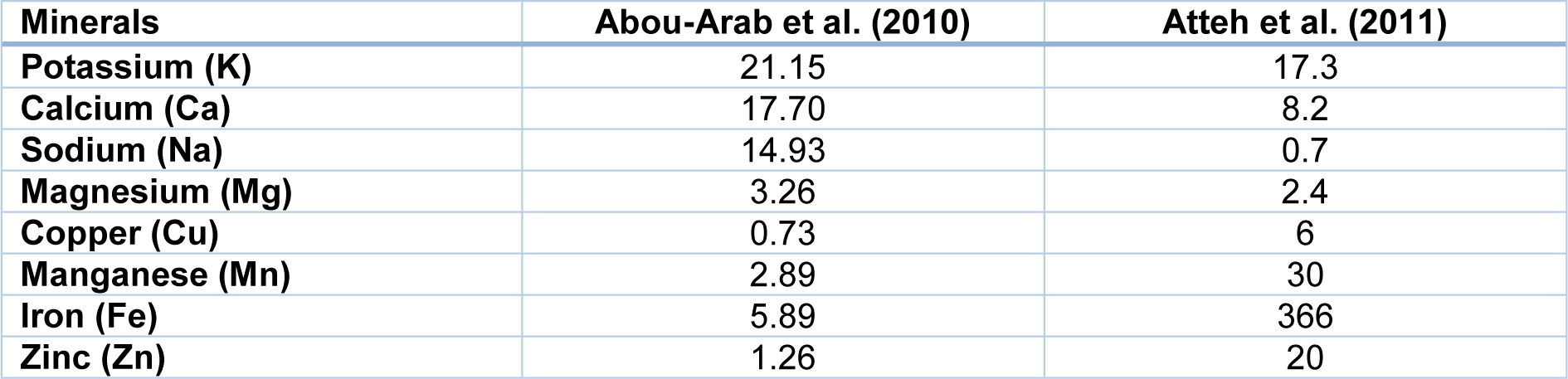
In addition, the biochemical composition of the plant depends on the geographical region of the growth (Table 1(Tab. 1)) (Khiraoui et al., 2017[48]). It was found that Stevia leaves contain a number of phenolic compounds that exhibit strong antioxidant properties. The content of total polyphenols and flavonoids in methanolic extracts was estimated as 25.18 mg/g and 21.73 mg/g, respectively (Tadhani et al., 2007[103]). This herb contains also oils, which are rich in palmitic, palmitoleic, stearic, oleic, linoleic and linolenic fatty acids (Table 2(Tab. 2)). Stevia leaves are also an important source of water-soluble vitamins including vitamin C (14.98 mg/100 g), vitamin B2 (0.43 mg/100 g), folic acid (52.18 mg/100 g) (Kim et al., 2011[49]). Moreover, the plant is rich in macro- and microelements such as Zn, Fe, Ca, K, Na, Mg and other minerals that are essential for human health (Table 3(Tab. 3)).
It should be noted, that not only Stevia contains stevial glycosides. Rubus suavissimus also known as sweet tea, contains steviol monoside, rebaudioside A, B, C, D, F, M, stevioside, steviolbioside, and rubusoside (Uhler and Yang, 2018[108]). In addition, R. suavissimus is rich in its own glycosides called suaviosides B, C, D1, D2, E, F, G, H, I and J (Ohtani et al., 1992[70]).
Health Benefits of Stevia
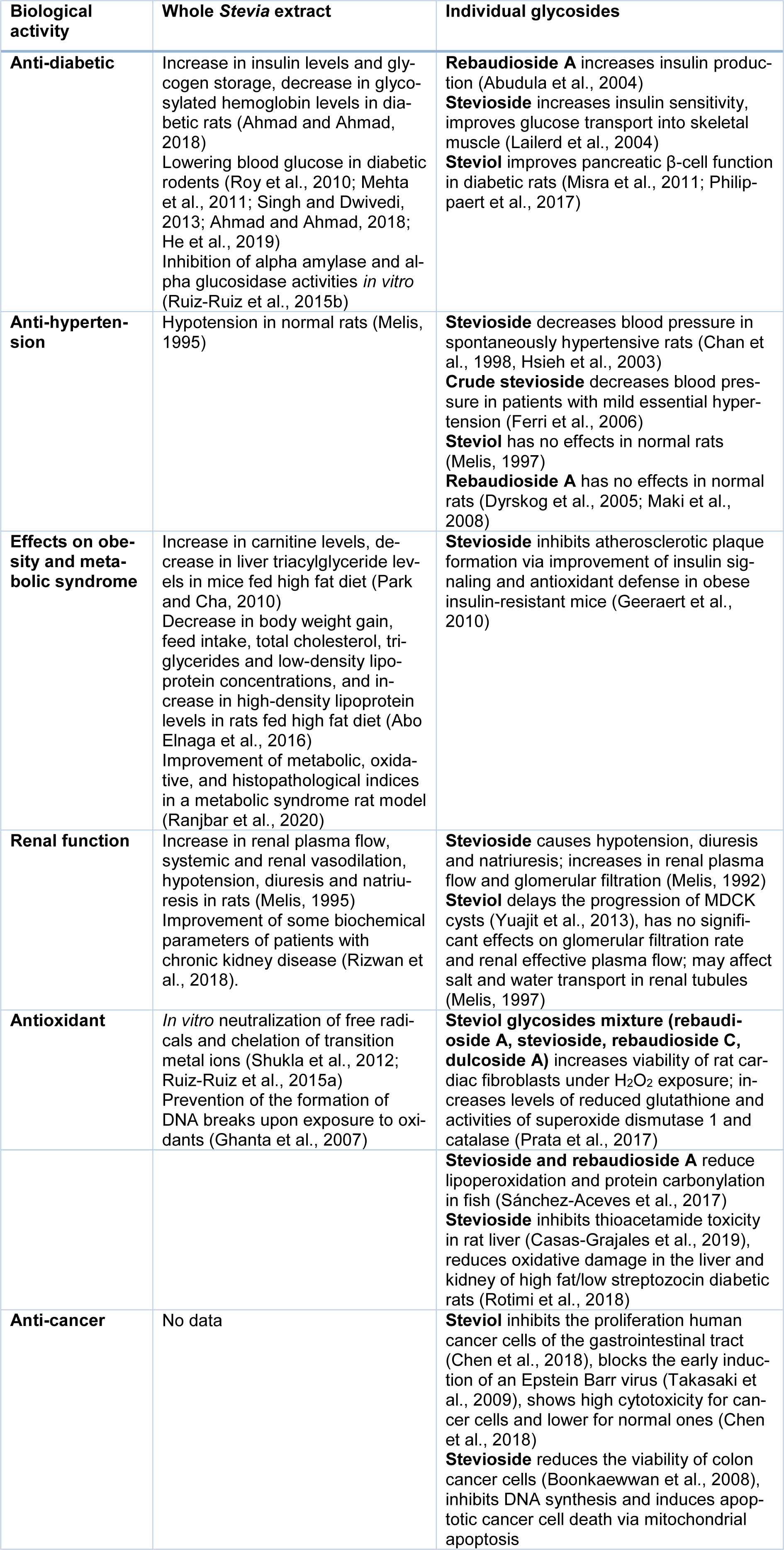
The properties of Stevia have been studied for over 100 years. Both earlier and current studies not only confirm the safety of Stevia leaf preparations but also find more and more benefits of its consumption for human health. Next, we will summarize current data on the established and potential pharmacological effects of Stevia extracts and its individual components in various medical models (Table 4(Tab. 4); References in Table 4: Abo Elnaga et al., 2016[1]; Abudula et al., 2004[3]; Ahmad and Ahmad, 2018[5]; Boonkaewwan et al., 2008[15]; Casas-Grajales et al., 2019[20]; Chan et al., 1998[22]; Chen et al., 2018[24]; Dyrskog et al., 2005[29]; Ferri et al., 2006[32]; Geeraert et al., 2010[36]; Ghanta et al., 2007[37]; He et al., 2019[40]; Hsieh et al., 2003[42]; Lailerd et al., 2004[52]; Maki et al., 2008[55]; Mehta et al., 2011[58]; Melis, 1992[62], 1995[59], 1997[61]; Misra et al., 2011[66]; Park and Cha, 2010[74]; Philippaert et al., 2017[79]; Prata et al., 2017[82]; Ranjbar et al., 2020[86]; Rizwan et al., 2018[88]; Rotimi et al., 2018[89]; Roy et al., 2010[90]; Ruiz-Ruiz et al., 2015[92][91]; Sánchez-Aceves et al., 2017[94]; Shukla et al., 2012[98]; Singh and Dwivedi, 2013[99]; Takasaki et al., 2009[105]; Yuajit et al., 2013[118], Figure 2(Fig. 2)).
Antioxidant properties of S. rebaudiana
Free radicals, which are continuously formed in any organism as result of metabolic processes or exposure of various stresses, may contribute a large number of human diseases, including cancer, obesity, diabetes, and neurodegenerative diseases (Garaschuk et al., 2018[34]; Bayliak et al., 2019[14]; Vaiserman et al., 2020[110][111]). Under physiological conditions, the capacity of endogenous antioxidant defense is sufficient to neutralize free radicals and prevent oxidative damages, but aging and excessive caloric intake are accompanied by imbalance between the production and elimination of free radicals followed by chronic oxidative stress and systemic inflammation development (Garaschuk et al., 2018[34]; Bayliak et al., 2019[14]; Vaiserman et al., 2020[111]). Therefore, using bioactive phytochemicals having antioxidant and anti-inflammatory activities is considered as a promising therapeutic approach to combat aging and associated pathological conditions (Michels et al., 2018[63]; Piskovatska et al., 2019[80]; Vaiserman et al., 2020[110][111]). Many studies showed that the antioxidant properties of S. rebaudiana might determine its ability to prevent and treat these diseases. Stevia leaves contain a number of phenolic compounds that are able to neutralize free radicals and chelate transition metal ions, thus preventing the involvement of the latter in free radical generation via the Fenton reaction (Ruiz-Ruiz et al., 2015[92]; Prata et al., 2017[82]).
Viability of rat cardiac fibroblasts pre-treated with glycosides of steviol was significantly increased under H2O2 exposure compared with the control cell group (Prata et al., 2017[82]). These cells also showed an increase in levels of reduced glutathione, activities of superoxide dismutase 1 and catalase (Prata et al., 2017[82]). One study found that Stevia aqueous leaf extract was able to reduce the levels of 1,1-diphenyl-2-picrylhydrazyl-hydroxyl radical, nitric oxide and superoxide anion radicals in vitro, although the effect was slightly lower compared to the antioxidant potential of ascorbic acid that was used as a positive control (Shukla et al., 2012[98]). Another study showed that ethyl acetate extract and crude 85 % methanolic extract from S. rebaudiana leaves prevented the formation of DNA breaks caused by free radicals (Ghanta et al., 2007[37]).
Stevia glycosides were found to reduce lipoperoxidation and decrease hydroperoxide and protein carbonyl content in Cyprinus carpio (Sánchez-Aceves et al., 2017[94]). Stevioside significantly reduced oxidative stress by decreasing the levels of lipid peroxidation and nitric oxide in the liver and kidney of diabetic rats (Rotimi et al., 2018[89]). In addition, stevioside has been shown to inhibit thioacetamide toxicity in rat liver (Casas-Grajales et al., 2019[20]). Prolonged administration of thioacetamide to rats caused damage to the liver and altered the content of nuclear erythroid factor 2 (Nrf2) that led to a decrease in antioxidant capacity of the liver. Stevioside prevented thioacetamide-induced liver damage by regulation of the level of Nrf2 protein (Casas-Grajales et al., 2019[20]).
The antioxidant properties of Stevia extracts depend on the methods of extract processing including stages of drying and extraction. Thus, the highest total content of phenols was observed in glycol-aqueous extracts from Stevia leaves, with lower phenol content in aqueous extracts and the lowest levels in ethanol extracts (Gaweł-Bęben et al., 2015[35]). Comparison of the antioxidant capacity of Stevia leaves dried by different methods (hot air drying at 100 °C, hot air drying at 180 °C, freeze drying, shade drying) showed that the best method of drying to keep high antioxidant potential was hot air drying at 180 °C. Under this regime, the content of antioxidants was 2-3 times higher than when using other drying methods (Periche et al., 2015[77]).
Anti-cancer effects
Cancer is the second leading cause of mortality in the world. The most common forms of cancers in 2017 were breast cancer, colorectal cancer, prostate cancer, and lung cancer. Therefore, the search for agents that may help in cancer treatment or prevention is extremely important (Ferlay et al., 2018[31]).
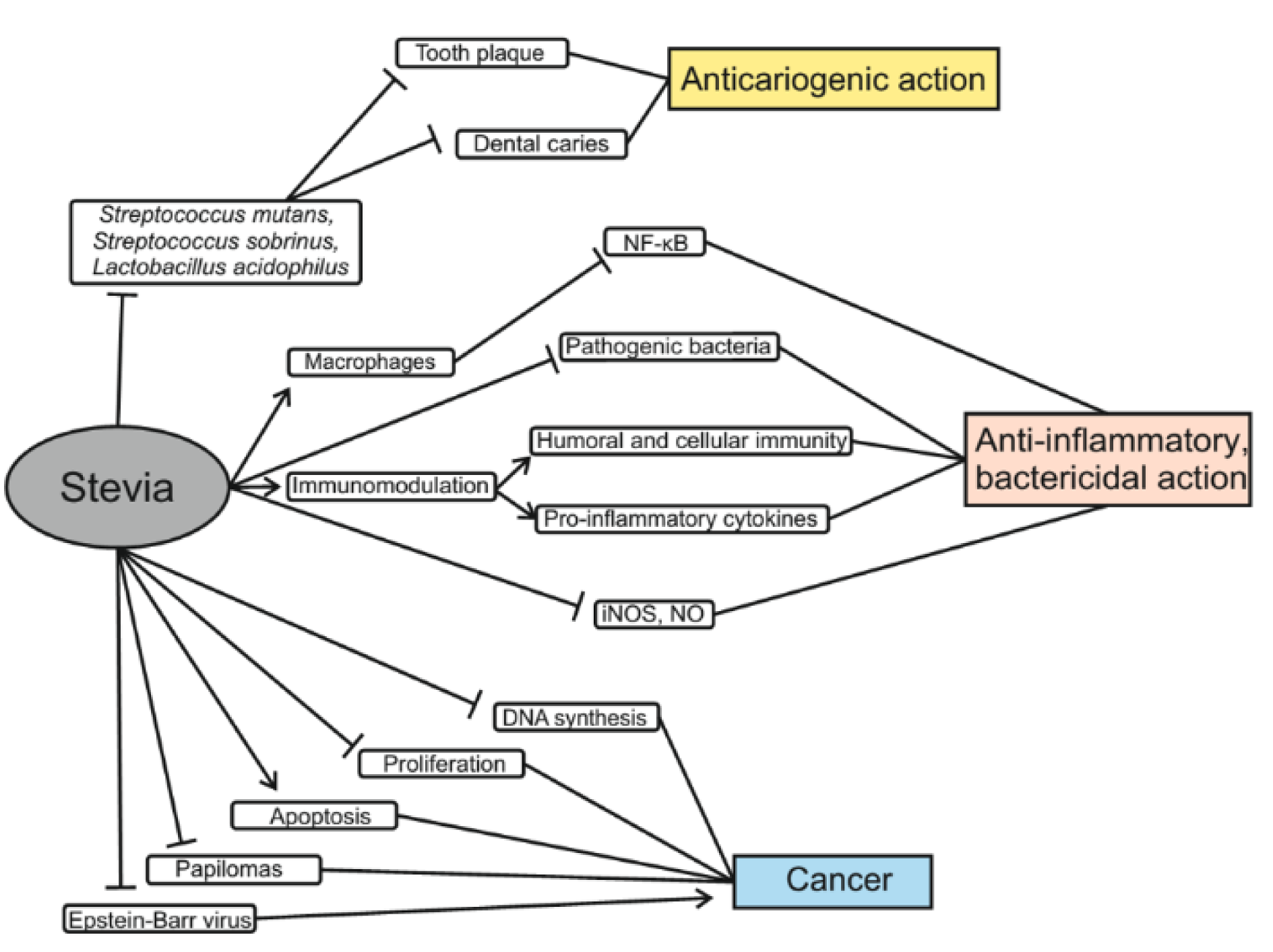
The results of one experiment showed that steviol inhibited the proliferation of six types of human cancer cells of the gastrointestinal tract (Chen et al., 2018[24]). High concentrations of stevioside were shown to reduce the viability of colon cancer cells (Boonkaewwan et al., 2008[15]). At a concentration of 100-200 μg/mL steviol acts with similar efficacy as 5-flourouracil (anti-cancer drug), and at a concentration of 250 μg/mL, it showed even greater cytotoxicity than 5-FU. Accordingly, steviol may become a potential chemotherapeutic agent for cancer treatment (Chen et al., 2018[24]). It is also worth noting that stevioside is less toxic to normal cells even at higher doses. Another study showed that stevioside could inhibit DNA synthesis and induce death of cancer cells via the mitochondrial apoptotic pathway. This is because steviol increases the expression of p21 and p53 proteins and decreases Cyclin D. As a result, it leads to up-regulation of the Bax/Bcl-2 ratio. Bax protein, in turn, leads to apoptosis associated with dysfunction of mitochondria and release of cytochrome c, which causes the activation of caspases that then cleave enzymes involved in repairing DNA and maintaining genome integrity (Paul et al., 2012[75]; Chen et al., 2018[24]). Due to this, stevioside showed a strong anti-cancer activity in cultured breast cancer MCF-7 cells (Paul et al., 2012[75]). In mice, combined treatment with stevioside and carcinogens, 7,12-dimethylbenzanthracene and 12-O-tetradecanoyl-phorbol-13-acetate (TPA), for 20 weeks, reduced papilloma formation by 94 % (Konoshima and Takasaki, 2002[50]). Similar results were obtained in another study that reported a three-fold reduction in the formation of tumors on skin cell meshes exposed to stevioside as compared with mice treated with 7,12-dimethylbenzanthracene plus TPA only (Yasukawa et al., 2002[117]). In addition, steviol, isosteviol and their metabolites block the induction of an early Epstein Barr virus antigen that inhibits tumor progression (Takasaki et al., 2009[105]). Thus, all of these studies support that effectiveness of steviol and other Stevia components in chemotherapy of cancer (Mizushina et al., 2005[67]) (Figure 3(Fig. 3)).
Anti-inflammatory and bactericidal action of Stevia. Anti-cariogenic properties
Recent studies demonstrate that S. rebaudiana is a unique plant with anti-inflammatory and bactericidal properties (Figure 3(Fig. 3)). Stevia extract inhibits the activity of many pathogenic bacteria, can be used to treat immune diseases and reduce edema (Preethi et al., 2011[83]; Jeong et al., 2010[45]; Arya et al., 2012[10]).
It was found that treatment of RAW 264.7 macrophage cells with the ethyl acetate fraction from Stevia leaves significantly inhibited the NF-κB-mediated gene expression that was stimulated by bacterial lipopolysaccharide (LPS). Such inhibition of NF-κB signaling was closely associated with a decrease in levels of interleukin-6 and monocyte chemoattractant protein-1. It was suggested that Stevia could be used for treatment of immune diseases such as rheumatoid arthritis and lupus (Jeong et al., 2010[45]). Stevioside was found to exhibit immunomodulatory effects and can act by stimulation of both humoral and cellular immunity and phagocytic function (Sehar et al., 2008[96]). Stevioside and steviol attenuate LPS-induced pro-inflammatory cytokine production by affecting cytokine gene expression via the IκBα/NF-κB signaling pathway (Boonkaewwan and Burodom, 2013[16]). Austroinulin and 6-O-acetyl-austroinulin are other natural diterpenoids isolated from S. rebaudiana with anti-inflammatory activity. It was found that these diterpenoids inhibit NO production and iNOS expression by blocking the activation of STAT1, IRF3, and NF-κB in LPS-stimulated RAW264.7 macrophages (Cho et al., 2013[25]). The methanolic extract of callus culture as well as the intact plant part of Stevia significantly inhibited carrageenan-induced rat paw edema (Preethi et al., 2011[83]; Arya et al., 2012[10]) analyzed the antibacterial activity of Stevia extracts in vitro against pathogenic bacteria such as Bacillus subtilis, Klebsiella pneumonia, Proteus vulgaris, Streptococcus pneumoniae, Staphylococcus aureus and Pseudomonas fluorescence. Six extracts from the leaves and three extracts from Stevia flowers obtained using of different solvents showed good antibacterial activity against all tested microorganisms.
Stevia also has antibacterial effects in the oral cavity (Gamboa and Chaves, 2012[33]). Dental caries is a common disease that affects not only adults but also children. Tooth caries occur due to poor oral hygiene, hereditary predisposition, consumption of foods rich in carbohydrates, or low levels of calcium and phosphorus in the body. Poor oral hygiene and consumption of carbohydrates promotes active reproduction of mouth microbiota following microbial colonization and biofilm and plaque formation on teeth. In vitro studies have shown that S. rebaudiana stops the growth of Streptococcus mutans, S. sobrinus and Lactobacillus acidophilus. These bacteria are associated with the development of caries (Contreras, 2013[26]).
Gamboa and Chaves (2012[33]) also studied the activity of different Stevia extracts against microorganisms that cause tooth decay. Extracts were obtained from dried S. rebaudiana leaves using hexane, methanol, ethanol, ethyl acetate or chloroform as solvents. The antimicrobial activity of these five extracts against 16 bacterial strains of the genera Streptococcus and Lactobacillus was evaluated by the well-known diffusion method. For the four Lactobacillus species, the inhibition zones obtained between 12.3 and 17.3 mm were somewhat larger with ethyl acetate and chloroform extracts, suggesting that these bacteria were the most susceptible microorganisms. In another study, four groups of rats were fed with stevioside, rebaudioside A or sucrose, which were added to the main diet. There were significant differences in the rates of sulcal caries and Streptococcus sobrinus count between a group fed diet with 30 % sucrose and the other 3 groups. There were no differences between groups fed stevioside or rebaudioside A and groups without additives. Thus, it can be argued that neither stevioside nor rebaudioside A are cariogenic (Das et al., 1992[27]). An in vivo study in humans was performed by measuring the plaque formation after rinsing with sucrose solution and S. rebaudiana solution four times a day for 5 days. The plaque accumulation after rinsing with Stevia solution was 57.82 % less than when rinsing with sucrose, measured by the Silness-Loe index, and 10.40 % less plaques when measured via the O'Leary plaque index (De Slavutzky, 2010[28]). Caries-positive and caries-negative controls were used for another experiment. That experiment found, that Stevia extract solution reduced the number of viable microbial cells compared to sucrose, suggesting that Stevia prevents growth of bacteria in the mouth (Giacaman et al., 2013[38]). Due to observed antimicrobial activity, Stevia was proposed for use to prevent dental caries (Ма and Blanksma, 2015[54]). Moreover, Stevia was shown to have strong anti-plaque and anti-gingivitis properties (Vandana et al., 2017[115]).
Stevia in the Treatment of Diabetes
Diabetes is a group of diseases in which insulin is either not produced in sufficient quantities (type 1 diabetes) or cannot be used effectively due to insulin resistance (type 2 diabetes). As of 2019, there were 464 million people with diabetes in the world; for comparison, in 2000, 175.4 million of patients with diabetes was recorded. According to the International Diabetes Federation, by 2040 this number will increase to 642 million (Zimmet et al., 2016[120]). About 90 % of patients are diagnosed with type 2 diabetes in both developing and developed countries (Tao et al., 2015[106]; Vaiserman and Lushchak, 2019[112]). Major risk factors contributing to the development of type 2 diabetes include genetic predisposition, unhealthy dietary behaviors, and low physical activity. The pathophysiology of type 2 diabetes is characterized by impaired glucose metabolism in the liver and insulin resistance in peripheral tissues which show reduced responsiveness to normal insulin concentrations and, therefore, cannot utilize glucose. As a result, glucose levels are increased in the blood in patients with type 2 diabetes, despite increased insulin production by the pancreas (Vaiserman and Lushchak, 2019[112][113]).
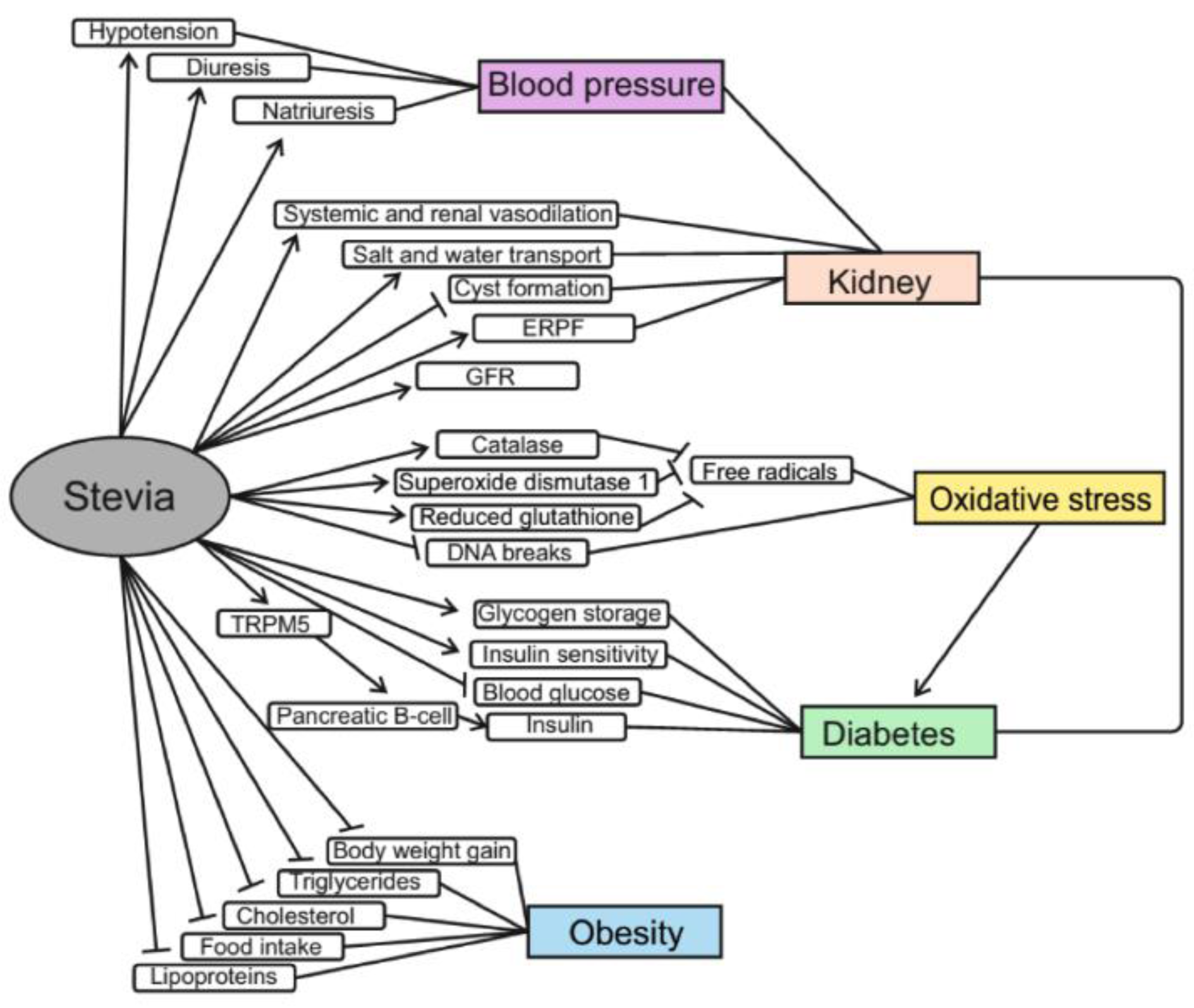
For many years, Stevia has been used not only as a sweetener but also as a medication in the treatment of diabetes and hyperglycemia in traditional medicine in Brazil and Paraguay (Chand and Kumar, 2016[23]). The anti-diabetic properties of Stevia leaf extracts and its individual ingredients are now being actively investigated (Figure 4(Fig. 4)). One study showed that diabetic rats that consumed an aqueous extract from Stevia leaves had higher levels of insulin and glycogen than animals of the control group (Ahmad and Ahmad, 2018[5]). An increase in insulin production was also observed in isolated mouse islets of Langerhans treated with rebaudioside A (Abudula et al., 2004[3]). In addition, stevioside can increase insulin sensitivity; in particular, low amounts of stevioside improved the effect of insulin on glucose transport into skeletal muscle (Lailerd et al., 2004[52]). Glycosylated hemoglobin in experimental groups of rats was significantly decreased as compared with diabetic control groups and was near to the value for non-diabetic rats (Lailerd et al., 2004[52]). Consumption of Stevia aqueous extract for 8 weeks resulted in a significant decrease in random blood glucose by 73.2 % and fasting blood glucose by 66.1 % (Ahmad and Ahmad, 2018[5]). A number of studies have also shown significant reduction in blood glucose levels after consumption of Stevia extract (Roy et al., 2010[90]; Mehta et al., 2011[58]; Singh and Dwivedi, 2013[99]; He et al., 2019[40]). Stevia extract has been found to improve pancreatic β-cell function in diabetic rats due to the ability of steviol glycosides to increase glucose-induced Ca2 + oscillations and insulin release by pancreatic islets (Misra et al., 2011[66]) This mechanism is based on the interaction of steviols, rebaudioside A and stevioside with the transient receptor potential cation channel subfamily melastatin member 5 (TRPM5). These receptors play an important role in the transmission of bitter, sweet and umami flavors and are localized on type II taste receptor cells and pancreatic β-cells. TRPM5 channels are activated by increasing the content of intracellular calcium and increased ATP release and afferent signaling from the type II taste receptor cell. In the case of β-cells, increasing the activity of TRPM5 leads to an increase in the frequency of oscillations of Vm and [Ca2+]cyt, which promotes insulin secretion (Philippaert et al., 2017[79]). Moreover, rats with hyperglycemia, after administration of Stevia aqueous extract, consumed significantly less water compared to other hyperglycemic rats (Ahmad et al., 2018[6]). Other studies have shown that stevioside and steviol may have potential in the treatment of diabetes because they act as anti-hyperglycemic agents by directly affecting β-cells of the pancreas and their insulinotropic action stopped when blood glucose level is dropped to the normal level (Jeppesen et al., 2000[46]). The absence of insulin-stimulating effects at normal glucose levels may reduce the risk of hypoglycemia (Jeppesen et al., 2000[46]). Groups of diabetic mice that consumed the antioxidant fraction of Stevia supplements showed better glucose tolerance than the control group (Milani et al., 2017[65]). Furthermore, Stevia leaf extract was found to be able to inhibit α-amylase and α-glucosidase. This can potentially slow down carbohydrate metabolism and reduce the risk of hyperglycemia in patients with type 1 and type 2 diabetes (Ruiz-Ruiz et al., 2015[91]). However, some studies on humans found no effects of supplements of Stevia aqueous extract on glucose, insulin, and glycosylated hemoglobin levels, but also no side effects (Barriocanal et al., 2008[13]; Ajami et al., 2020[7]).
Anti-hypertension effects of Stevia
High blood pressure or hypertension is the most dangerous factor in the development of myocardial infarction and ischemic stroke. It can be a separate disease or a symptom of chronic diseases of kidney and endocrine organs, some heart defects, atherosclerosis, organic lesions of the central nervous system, etc. (Oparil et al., 2018[73]).
In one study, the effect of intravenous stevioside on blood pressure was studied in spontaneously hypertensive rats (SHR). The hypotensive effect on both systolic and diastolic blood pressure was dose-dependent for intravenous doses of 50, 100 and 200 mg/kg in conscious SHR. The hypotensive effect lasted for more than 60 min with a dose of 200 mg/kg. Thus, stevioside was effective in blood pressure reduction (Chan et al., 1998[22]). In another study, S. rebaudiana extracts were administered to rats for 20, 40, and 60 days and results showed that rats treated with Stevia extract for 20 days did not significantly differ from the control group in the arterial pressure (Melis, 1995[59]). However, after 40 or 60 days of the administration, the extract caused hypotension, diuresis and natriuresis with a constant glomerular filtration rate. Steviol treatment did not cause significant changes in mean blood pressure in rats, diuresis and natriuresis with a constant glomerular filtration rate (Melis, 1997[61]). In one study, patients took capsules containing 500 mg stevioside powder or placebo three times daily for 2 years. This study shown that in patients with mild hypertension, oral stevioside significantly decreased systolic blood pressure and diastolic blood pressure compared with placebo (Hsieh et al., 2003[42]). Consumption of 1000 mg/day of rebaudioside A caused no clinically important changes in blood pressure in healthy adults with normal and low-normal blood pressure (Maki et al., 2008[55]). Rebaudioside A did not affect blood pressure or glycemic control when Goto-Kakizaki rats consumed this glycoside (0.025 g/kg body mass/day) for eight weeks (Dyrskog et al., 2005[29]). Systolic and diastolic blood pressure decreased in humans during treatment with crude stevioside, but a similar effect was observed in the placebo group (Ferri et al., 2006[32]).
Stevia effects on obesity
At present, obesity is considered to be a serious global problem, and the number of obese and overweight people continues to increase worldwide. However, obesity is not just overweight, but it is a complex disease with many metabolic complications, such as diabetes, cancer, cardiovascular diseases, stroke, and sleep apnea. Obesity significantly increases the risk of these diseases leading eventually to impaired quality of human life (Bayliak et al., 2019[14]). Abdominal obesity is one of the factors of metabolic syndrome, that increases a risk of cardiovascular diseases (Carrera-Lanestosa et al., 2017[19]; Bayliak et al., 2019[14]). These factors include hepatic steatosis and dyslipidemia. Research shows that consumption of non-nutritive sweeteners can prevent body weight gaining by reducing energy intake (Ashwell, 2015[11]). In line with this, studies with S. rebaudiana support its anti-obesity properties (Figure 4(Fig. 4)). In one 12-week experiment on rats, it was found that oral administration of Stevia sweetener at doses of 25, 250, 500 and 1000 mg/kg body mass decreased body weight gain by 40.3, 41.4, 45.0 and 48.3 %, respectively (Abo Elnaga et al., 2016[1]). This may be due to the reduced food intake after administration of Stevia sweetener. Stevia sweetener intake also reduced total cholesterol, triglycerides and low-density lipoprotein concentrations and increased high-density lipoprotein levels in the blood of experimental rats (Abo Elnaga et al., 2016[1]). S. rebaudiana extract supplementation decreased serum and liver triacylglyceride le-vels in mice fed a high-fat diet compared to mice fed the high fat diet without adding Stevia extract (Park and Cha, 2010[74]). In addition, mice kept on a high-fat diet with Stevia extract showed an increase in carnitine levels, which plays an important role in fatty acid metabolism as it transports fatty acids from cytosol to mitochondria where they are oxidized (Park and Cha, 2010[74]).
Another human study found that the significant difference in calorie intake between the control group and the group that consumed Stevia extract was only due to the difference in calorie intake; therefore, consumption of Stevia extract did not affect the feeling of hunger and appetite (Farhat et al., 2019[30]). It also can explain why the participants in another human experiment did not consume additional food to compensate for the lack of calories under Stevia extract administration in contrast to participants who consumed sucrose (Anton et al., 2010[9]).
Obesity is frequently associated with insulin resistance and increased inflammation and oxidative stress. One study showed that stevioside treatment was associated with inhibition of atherosclerotic plaque development via improvement of insulin signaling and antioxidant defense in obese insulin-resistant mice (Geeraert et al., 2010[36]). In addition, it was shown that Stevia extract could prevent the adverse effects of high fat, high sucrose diet on lipid profiles, total antioxidant capacity and histopathologic factors in obese rats (Ranjbar et al., 2020[86]).
Renal function
The kidneys are one of the most important organs, their main function being to excrete end- and side-products of metabolism. The kidneys are also involved in regulation of blood pressure, water-salt balance and red blood cell formation (Onopiuk et al., 2015[72]). Diabetes is the leading cause of kidney disease. In turn, diabetic kidney disease (DKD) is the leading cause of kidney failure worldwide. People with diabetes most often die from renal failure (Reidy et al., 2014[87]). High glucose concentrations induce specific cellular changes, which affect many types of cells in the kidney, including endothelial cells, smooth muscle cells, mesangial cells, podocytes, cells of the tubular and collecting duct system, and inflammatory cells and myofibroblasts (Vallon and Komers, 2011[114]).
Experiments with normal and hypertensive rats showed that stevioside caused hypotension, diuresis and natriuresis in both groups (Melis, 1992[62]). The first (control) group demonstrated an increase in renal plasma flow and glomerular filtration rate constant following stevioside administration. For the group that consumed the extract for 60 days, an increase in renal plasma flow was observed. In addition, it was shown that the aqueous extract from dried Stevia leaves caused systemic and renal vasodilation, hypotension, diuresis and natriuresis (Melis, 1995[59]). Yuajit et al. (2013[118]) investigated inhibitory effects and the detailed mechanisms of action of steviol and its derivatives on cyst growth using a cyst model, the Madina-Darby canine kidney (MDCK) cell line. Taken together, the data suggest that steviol delays the progression of MDCK cysts by directly inhibiting cystic fibrosis transmembrane conductance regulator (CFTR) chloride channel activity and reducing CFTR expression, in particular by promoting proteasomal degradation of CFTR. Therefore, steviol and related compounds can be further used to treat polycystic kidney disease. Stevia extract improves some biochemical parameters of patients with chronic kidney disease; in particular, patients have improved serum creatinine, uric acid, and blood sugar before meals (Rizwan et al., 2018[88]). Steviol elicited no significant changes in glomerular filtration rate (GFR) and renal effective plasma flow (ERPF). However, the steviol infusion induced a significant increase in fractional sodium excretion, fractional potassium excretion, and urinary flow as a percentage of glomerular filtration rate (V/GFR) when compared to controls. The data suggest that steviol may affect salt and water transport in renal tubules (Melis, 1997[61]).
Toxicity and Side Effects
The acceptable daily intake of Stevia dry extract defined by the Scientific Committee on Food of the European Food Safety Authority and Food and Drug Administration is 4 mg/kg body mass (Lohner et al., 2017[53]). One animal study showed the allergenic potential of Stevia preparations. It is possible that crude Stevia extracts have a higher allergenic potential than high purity stevia-based sweeteners containing ≥ 95 % steviol glycosides, since crude extracts are more likely to contain allergenic substances inherent in the Asteraceae family (Urban et al., 2015[109]), but this issue is not well studied yet.
According to the results of one earlier study, Stevia extract reduced the fertility of rats by up to 21 % compared with control group of rats. Fertility remained reduced by 47 % even after a 50-60 day recovery period (Mazzei-Planas and Kuc, 1968[57]). In Stevia-treated rats, a decrease in the relative weight of seminal vesicle and testis was observed as well as a significant decrease in the number of spermatozoa stored (Melis, 1999[60]).
One study found that steviol is mutagenic, although other studies have not confirmed this effect (Pezzuto et al., 1985[78]). The main evidence of Stevia safety is that for over 1500 years of continuous use by Paraguayans, there have been any reports of adverse effects. Additional confirmation of the safety of Stevia consumption is the absence of reports on side effects of any kind in Japanese populations where Stevia has been consumed in large quantities in recent years (Singh and Rao, 2005[100]). In addition, most of the studies that have investigated Stevia effects on the human body have showed no side effects (Roy et al., 2010[90]; Nikiforov et al., 2013[69]; Uçar et al., 2017[107]). However, it should be noted that not all of the Stevia products sold are of high quality. In particular, in the one experiment Raman spectra of six commercial products of Stevia were measured and it was found that three of the commercial Stevia products were counterfeit products. They also contained sodium cyclamate and small amounts of sodium saccharin (Jentzsch et al., 2016[44]).
Conclusions
S. rebaudiana, a perennial plant native to Paraguay, has come to be cultivated around the world as a source of high-potency sweetener with no caloric value. Two main steviol diterpene glycosides, stevioside and rebaudioside A, that are present in high levels in Stevia leaves provide the sweet taste of the plant and are 150-450 times sweeter than sucrose to human taste buds. Pure sweet glycosides and crude Stevia extracts with 50 % of glycosides are actively used in the food market. A number of preclinical and clinical studies suggest potential therapeutic and pharmacological applications for Stevia extracts and their individual compounds because they demonstrate no toxicity in experimental trails and exhibit health-promoting activities. In addition to different glycosides, Stevia leaves contain many other compounds like flavonoids and fatty acids that together provide the diverse biological properties of the plant. Thanks to these components, Stevia products stimulate insulin production in diabetics, improve polycystic kidney disease, have chemotherapeutic action in cancer and possess powerful antibacterial, antioxidant and immunomodulating properties (Figure 2(Fig. 2)). More research is needed to elucidate which compounds are the main determinants of the known Stevia-based effects as well as their molecular mechanisms of action. In addition, mechanisms need to be established by which Stevia sweetener reduces food intake and lowers total cholesterol, triacylglycerides, and low-density lipoproteins.
Notes
Victoria Peteliuk and Lesia Rybchuk contributed equally as first authors.
Ethical statement
This is a review paper, which does not include animal or human experiments.
Funding
This work was partially supported by the Ministry of Education and Science of Ukraine (grants #0117U006426 and #0118U003477) and a Discovery grant from the Natural Sciences and Engineering Research Council of Canada (#6793).
Conflict of interest
The authors declare that they have no conflict of interest.
Authors’ contributions
Victoria Peteliuk and Lesia Rybchuk collected literature and wrote the original draft, prepared the figures and tables; Maria Bayliak performed analysis, review and edited the manuscript, helped to prepare and arrange tables and figures; Kenneth Storey performed review, editing and provided valuable discussion; Oleh Lushchak: provided funding acquisition, idea and design of the article, supervising, review, and editing. All authors read and approved the final manuscript.
References
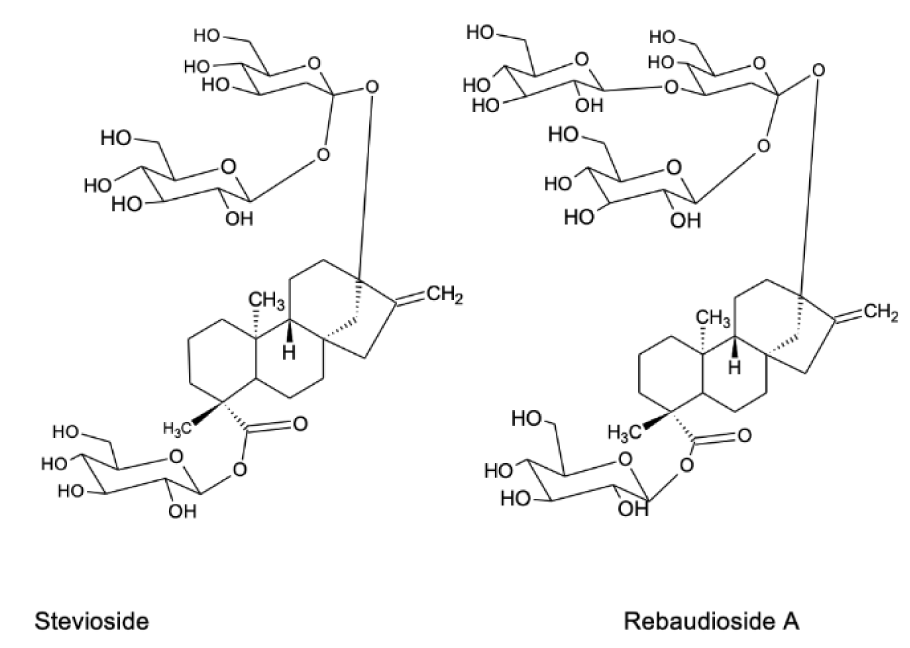
Figure 1: Chemical structure of two main compounds of Stevia leaves responsible for their sweet taste
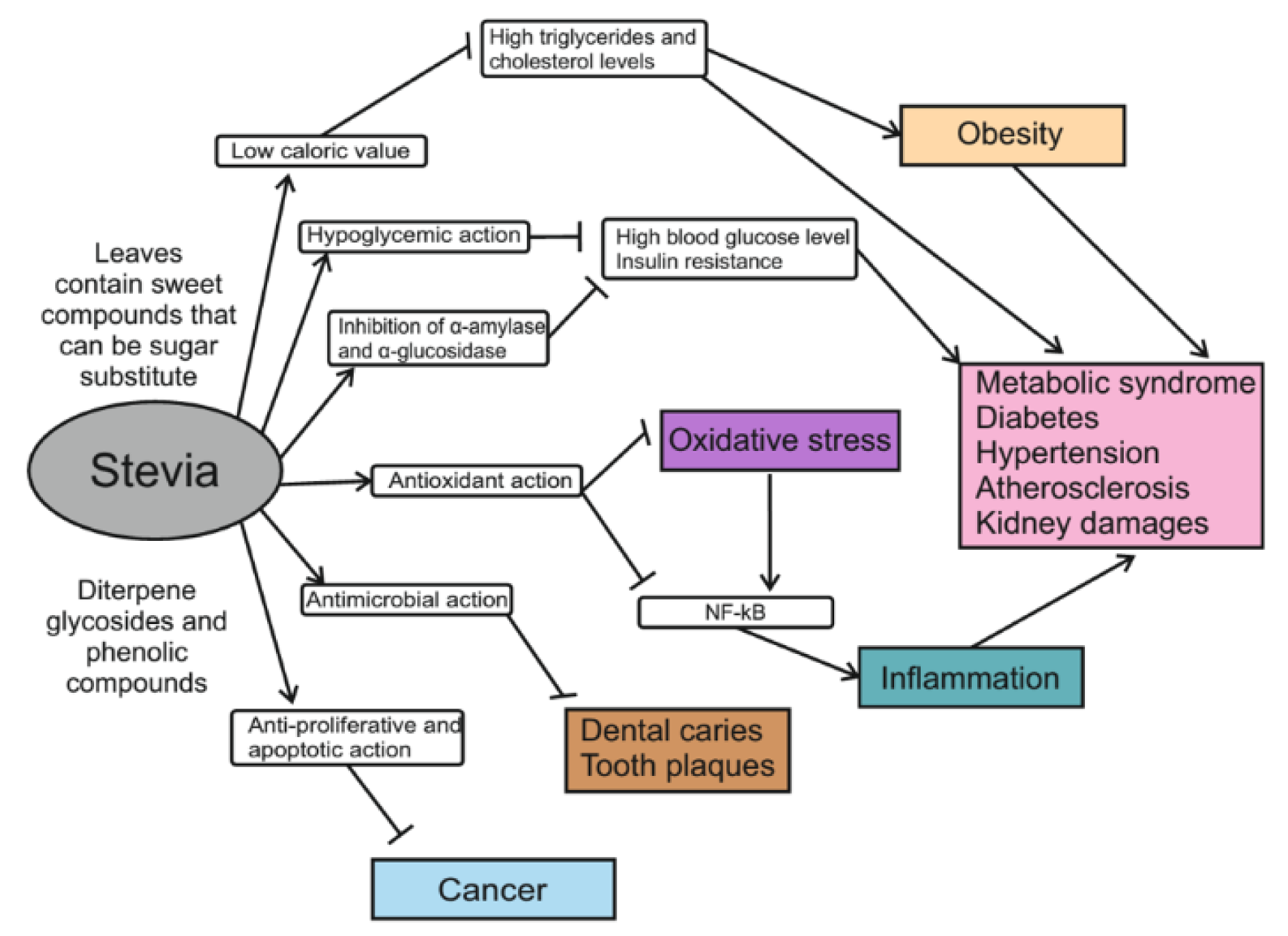
Figure 2: Schematic summarizing of health-promoting effects of Stevia leaf extract and its constituents
[*] Corresponding Author:
Oleh Lushchak, Department of Biochemistry and Biotechnology, Vasyl Stefanyk Precarpathian National University, 57 Shevchenko Str., Ivano-Frankivsk, 76018, Ukraine, eMail: oleh.lushchak@pnu.edu.ua